Overview
- Baraminology is a creationist pseudoscience that attempts to define discrete “kinds” (baramins) of organisms that God created separately, within which evolution is permitted but between which it is not.
- Its central methods — the hybridization criterion and statistical baraminology — lack theoretical grounding and produce results that contradict each other and mainstream taxonomy alike.
- Young Earth creationism’s own Ark model requires thousands of “kinds” to have radiated into millions of species in under 4,500 years, demanding rates of evolution far exceeding anything observed or proposed by mainstream biology.
Overview
Baraminology is a creationist research program that attempts to classify living organisms into discrete groups called baramins — a portmanteau of the Hebrew words bara (created) and min (kind) — on the premise that God created each such group independently and that genuine evolutionary change cannot cross the boundary between them. Proponents present baraminology as a scientific alternative to Linnaean taxonomy and phylogenetic systematics, though it operates within a prior theological commitment that predetermined its conclusions before any evidence was gathered. The field emerged within Young Earth creationist (YEC) institutions in the mid-twentieth century and has been elaborated primarily by American researchers working outside mainstream peer-reviewed biology. Its significance for the broader creationism debate lies not only in what it claims but in the internal contradictions it generates: the YEC model of Noah’s Ark and the post-flood world requires the very rapid, large-scale evolutionary change that baraminologists simultaneously insist is impossible.1, 2, 6
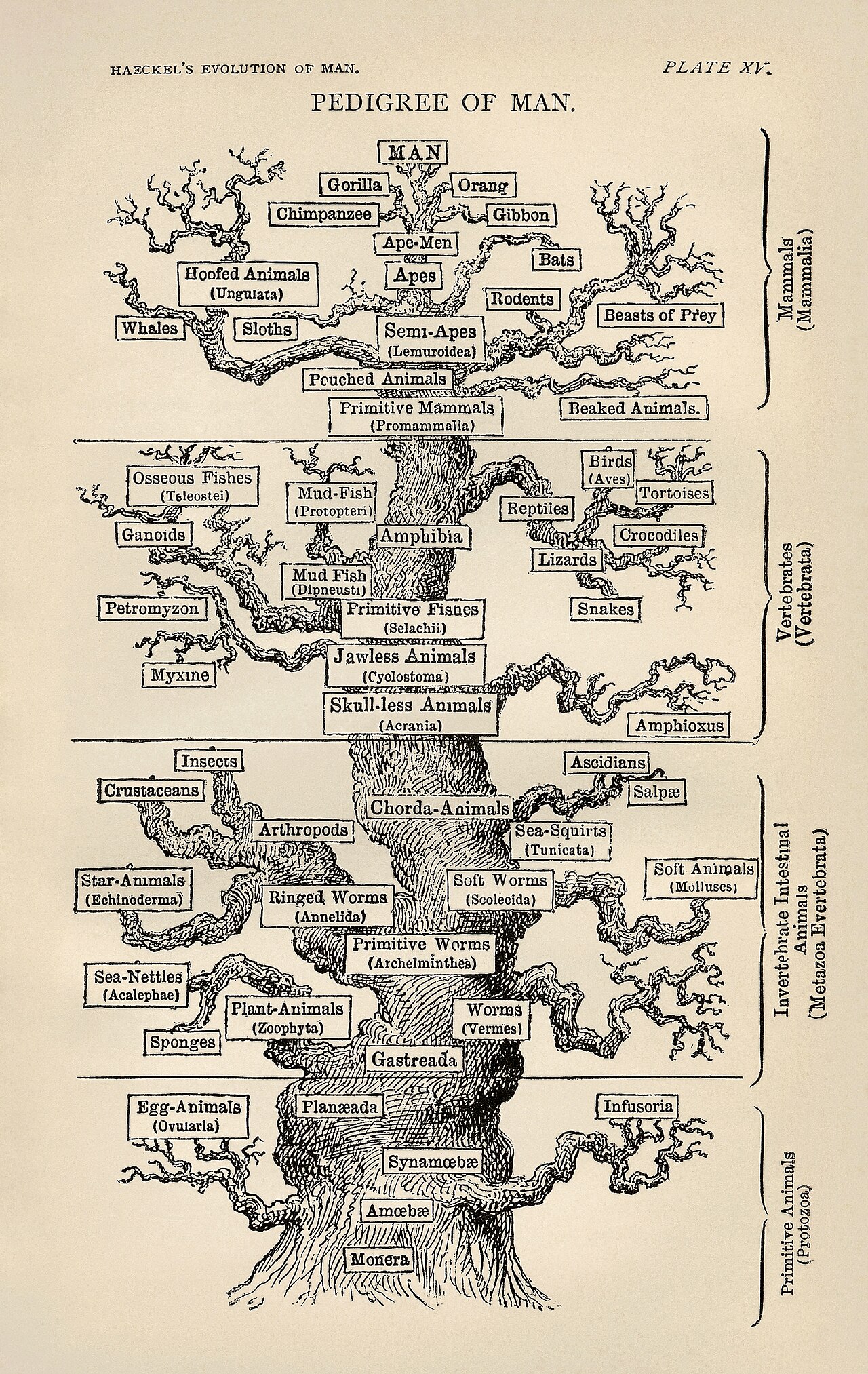
Origins and development
The modern concept of created kinds was given its first systematic treatment by Frank Lewis Marsh, a Seventh-day Adventist biologist, in his 1941 book Fundamental Biology.1 Marsh coined the term baramin and proposed that each created kind corresponded roughly to what Linnaeus had called a species, though he acknowledged that the correspondence was imperfect. Marsh was attempting to reconcile observed biological variation — including the production of new varieties through selective breeding — with a literal reading of Genesis. He argued that God had created immutable types and that all variation since creation occurred within those types, never producing genuinely new ones.
For several decades Marsh’s framework remained a minority interest even within creationism. It was not until the 1990s that a more technical research program began to coalesce around the concept, driven principally by geologist Kurt Wise and biologist Todd Charles Wood. Wise, who holds a doctorate in paleontology from Harvard and studied under Stephen Jay Gould, argued that baraminology could be placed on a more rigorous empirical footing by identifying the features that reliably mark kind boundaries.2 Wood subsequently developed what he called statistical baraminology, applying multivariate statistical analyses to morphological and molecular character matrices in an effort to detect discontinuities in biological data that would correspond to baramin boundaries.3, 4 These researchers published primarily in the Creation Research Society Quarterly, the Answers Research Journal, and the occasional papers of the Baraminology Study Group — venues that operate outside the standard peer-review infrastructure of professional biology.
Methods used in baraminological classification
Baraminologists employ two principal methods for assigning organisms to kinds. The first and historically prior method is the hybridization criterion: if two organisms can produce viable hybrids, they are presumed to belong to the same baramin. This criterion has an intuitive appeal, since reproductive compatibility is also central to the biological species concept used in mainstream biology. However, its application in baraminology differs fundamentally. In mainstream biology, reproductive isolation marks the boundary between species, not between higher-level groups; hybridization across very different organisms is treated as evidence of evolutionary relatedness, not of shared created identity. Baraminologists, by contrast, use hybridization to lump organisms together at a level that can encompass what mainstream taxonomy regards as entire genera, families, or even orders, on the grounds that any demonstrated hybridization — even among distantly related organisms — signals common creation rather than common descent.11
The second method, statistical baraminology, applies techniques such as cluster analysis, principal coordinates analysis, and a custom metric called BARCLAY (baraminic distance) to matrices of organismal characters. The intent is to identify natural groupings — clusters of organisms with many shared characters separated by zones of apparent discontinuity from other clusters — that would correspond to original created kinds.4 Wood has applied this approach to the great apes and humans, concluding that Homo floresiensis and other fossil hominins fall within a single human holobaramin that excludes chimpanzees and gorillas — a conclusion that requires ignoring the overwhelming molecular and morphological evidence for human-ape common ancestry.5 Practitioner outputs depend heavily on which characters are selected for inclusion, and no principled, theory-independent criterion exists for making that selection within the baraminological framework.
Why baraminology fails scientifically
The most fundamental objection to baraminology is that it posits arbitrary discontinuities in a biological world that does not exhibit them at the level required. Mainstream taxonomy, from Linnaeus onward, recognized that organisms form a nested hierarchy of similarities: species group into genera, genera into families, families into orders, and so on, with each level sharing a progressively smaller set of derived characters with its relatives. This pattern is precisely what common descent with modification predicts and precisely what special creation does not obviously predict.9 If God created each kind independently, there is no particular reason to expect that cats and dogs should share more features with each other than either shares with frogs, or that all vertebrates should share a basic body plan absent from insects. The nested hierarchy is, in the language of phylogenetics, a signal of shared ancestry. Baraminology must treat this hierarchy as a meaningless artifact or explain it away without offering a mechanistic account of why an independent creator would produce it.9
Molecular phylogenetics has deepened this problem considerably since Marsh’s era. The sequencing of entire genomes has revealed not merely sequence similarities but the shared presence of functionless or nearly functionless elements — endogenous retroviruses, pseudogenes, and conserved non-coding regions — at identical positions across related organisms.8, 12 The probability that two independently created lineages would independently acquire the same nonfunctional retroviral insertion at the same chromosomal locus is vanishingly small. Yet humans and other great apes share dozens of such insertions. These molecular signatures are precisely what a phylogenetic model predicts and what a baraminological model of independent creation cannot coherently account for. Baraminologists have not offered a mechanistic explanation for this pattern; they can only note that God could theoretically have created organisms with similar sequences, which converts the empirical question into an unfalsifiable theological one.
The hybridization criterion generates its own internal problems. Hybridization in nature is far more widespread than early baraminologists acknowledged, occurring across organisms that mainstream biology considers members of distinct genera, families, and in some plant lineages even orders.11 If successful hybridization is taken as definitive evidence of shared baramin membership, the logical consequence is that enormous swaths of the tree of life must be collapsed into single kinds — an outcome that severely undermines the notion that kinds are meaningfully distinct. Conversely, many organisms that share overwhelming morphological and molecular similarity cannot hybridize because of chromosome number differences or geographic isolation, which would place them in separate kinds despite every other line of evidence pointing to close common ancestry.
Ring species present a particularly clear challenge to the concept of fixed kind boundaries. In a ring species complex, such as the Larus gulls of the circumpolar region or the Ensatina salamanders of California, a chain of interbreeding populations forms a geographic ring such that the two terminal populations, which overlap in the same location, are reproductively isolated from each other.7 There is no non-arbitrary point in the ring at which a kind boundary can be drawn: each adjacent pair of populations can interbreed, yet the endpoints cannot. This is precisely the pattern that gradual speciation predicts, and it is the pattern that a model of fixed created kinds cannot accommodate without circularity.
The Ark problem and the hyper-evolution paradox
The most damaging internal contradiction within Young Earth creationism as a whole — and the one that baraminology both creates and cannot resolve — concerns the logistics of Noah’s Ark and the subsequent diversification of life. Current estimates place the number of described animal species on Earth at approximately 8.7 million, with the true number likely higher when undescribed species are included.14 Mainstream YEC Ark models, including those developed by Answers in Genesis for the Ark Encounter theme park in Kentucky, address this problem by arguing that the Ark carried not individual species but representative pairs of each kind. Lightner and colleagues estimated that the Ark needed to carry approximately 1,400 mammalian kinds and a total of perhaps 7,000 animal kinds across all taxa.6
This solution immediately produces a catastrophic arithmetic problem. If 7,000 kinds disembarked from Ararat roughly 4,500 years ago and have since diversified into the millions of species alive today, the average rate of new species formation required is staggering — on the order of hundreds of new species per kind per millennium, sustained continuously from the post-flood period to the present.10 For comparison, mainstream evolutionary biology, working with timescales of millions to tens of millions of years, documents speciation as a relatively slow process driven by the gradual accumulation of genetic differences under natural selection, genetic drift, and reproductive isolation. The rates required by the YEC post-flood diversification model exceed even the most rapid documented speciation events by orders of magnitude.13
Wood, to his credit, has acknowledged this problem directly and has argued for what he calls "post-flood diversification" driven by mechanisms that baraminologists have not specified in any mechanistic detail.10 The irony is stark and frequently noted by critics: Young Earth creationism, which markets itself as an alternative to evolutionary theory and insists that macroevolution is impossible, must in fact posit a rate and scope of evolutionary change that dwarfs anything proposed by mainstream evolutionary biology. The YEC model requires not less evolution but far more of it, compressed into a tiny fraction of the time, driven by mechanisms that have never been identified, tested, or even coherently described. This is not a peripheral difficulty but a structural contradiction at the heart of the research program.
Classification in practice: arbitrary outcomes
When baraminological methods are applied to actual organisms, the results reveal the arbitrariness of the framework rather than confirming it. Different baraminologists, applying the same nominal criteria to the same organisms, frequently reach different conclusions about which organisms belong to the same kind. The feline family (Felidae) has been classified by some baraminologists as a single kind and by others as multiple kinds, depending on which characters are emphasized and how the hybridization data are weighted. The canid family (Canidae) presents similar ambiguities. These disagreements are not resolved by appeal to additional data but by appeal to interpretive choices that are themselves determined by prior theological commitments rather than by the data.3
The fossil record compounds the problem. Baraminology must account not only for living species but for the vast diversity of extinct organisms documented in the paleontological record. The YEC model holds that most fossil organisms were buried in the flood, meaning that a wide range of morphologically distinct fossil forms must be assigned to the same kinds as living organisms, or to kinds that went entirely extinct. As the morphological gap between a living kind and its presumed fossil relatives widens, the baraminological assignment becomes increasingly difficult to justify on empirical grounds. In the case of human evolution, baraminologists assign Australopithecus, Homo habilis, and Homo erectus either to the human kind or to an ape kind, depending on the researcher, with the assignment tracking theological preferences rather than morphological evidence.5
Academic standing and scientific reception
Baraminology has not achieved recognition within mainstream biological science. Its publications appear almost exclusively in creationist-affiliated journals that either lack peer review or apply peer review only within a community of researchers who share the same theological starting assumptions. The methods employed — statistical cluster analysis, character matrices — are borrowed from mainstream systematics, but they are applied without the theoretical framework that gives those methods their explanatory power. In mainstream phylogenetics, character matrices are analyzed to reconstruct evolutionary histories, and the methods have been extensively validated against molecular data and the fossil record. In baraminology, the same tools are used to search for discontinuities that the researcher is committed in advance to finding, and the results are not subject to the independent replication and cross-validation that characterize productive scientific programs.9
Todd Wood, the most technically sophisticated of contemporary baraminologists, has himself written candidly that "evolution is not a theory in crisis" and that the evidence for common descent is substantial and not easily dismissed.4 His acknowledgment reflects an intellectual honesty unusual within YEC literature, but it also highlights the tension between the empirical evidence and the theological commitment that baraminology is designed to preserve. Wood continues to pursue baraminology not because the evidence compels it but because his reading of scripture requires it — a transparently faith-first methodology that disqualifies the enterprise from the category of empirical science.
Assessment
Baraminology represents an attempt to give scientific form to a theological conviction that predates any of its evidence. The conviction — that God created distinct, bounded kinds of organisms that cannot give rise to genuinely new kinds — was read out of the Genesis text centuries before modern genetics, molecular biology, or phylogenetics existed. The methods Marsh, Wise, and Wood developed were constructed not to test whether the conviction is correct but to find a way of maintaining it in the face of mounting evidence to the contrary. The result is a framework that is unfalsifiable at its core (any evidence of change is attributed to variation within a kind; any apparent boundary is taken as confirmation of a kind boundary), methodologically inconsistent (the hybridization criterion collapses the distinctions baraminology needs to maintain; statistical baraminology produces researcher-dependent results), and internally self-defeating (the Ark model requires more evolutionary change, more rapidly, than mainstream biology has ever proposed).
The nested hierarchy of life, the shared molecular signatures of common ancestry including endogenous retroviruses and pseudogenes, the continuous gradations documented in ring species and the fossil record, and the demonstrated capacity of populations to accumulate reproductive isolation through ordinary genetic mechanisms all point to a single explanatory framework: descent with modification from common ancestors, operating over billions of years. Baraminology cannot account for these patterns without abandoning either its methods or its conclusions. It survives not as a scientific research program but as a theological apologetic dressed in the vocabulary of science.