Overview
- Background extinction operates continuously at a low, steady rate of roughly 0.1 to 1.0 species per million species-years, while mass extinctions are rare, catastrophic events that eliminate 75 percent or more of species in geologically brief intervals and fundamentally reset the trajectory of life on Earth.
- Mass extinctions are selective in ways that differ from normal times — traits such as broad geographic range, which confer little advantage during background intervals, become critical determinants of survival during crises, meaning that the winners and losers of mass extinctions cannot be predicted from ordinary Darwinian fitness alone.
- Modern extinction rates are estimated at 100 to 1,000 times the background rate derived from the fossil record, placing the current biodiversity crisis in a category comparable to the five major mass extinctions of the Phanerozoic.
Extinction — the permanent disappearance of a species from Earth — is as fundamental to the history of life as the origination of new species. More than 99 percent of all species that have ever lived are now extinct, and the patterns of their disappearance have shaped the structure of modern biodiversity as profoundly as natural selection and speciation.1, 2 Since the pioneering work of David Raup and Jack Sepkoski in the 1980s, paleontologists have recognized that extinction operates in two qualitatively different modes: a steady trickle of species losses known as background extinction, and rare, catastrophic mass extinctions that eliminate vast fractions of life in geologically brief intervals.2 The relationship between extinction and natural selection is more complex than Darwin envisioned. While background extinction tends to remove species that are poorly adapted to their environments, mass extinctions can override the accumulated gains of millions of years of adaptive evolution, eliminating lineages for reasons largely unrelated to their ordinary ecological fitness.3, 4
Background extinction
Background extinction refers to the continuous, low-level loss of species that occurs between the major extinction events in the fossil record.
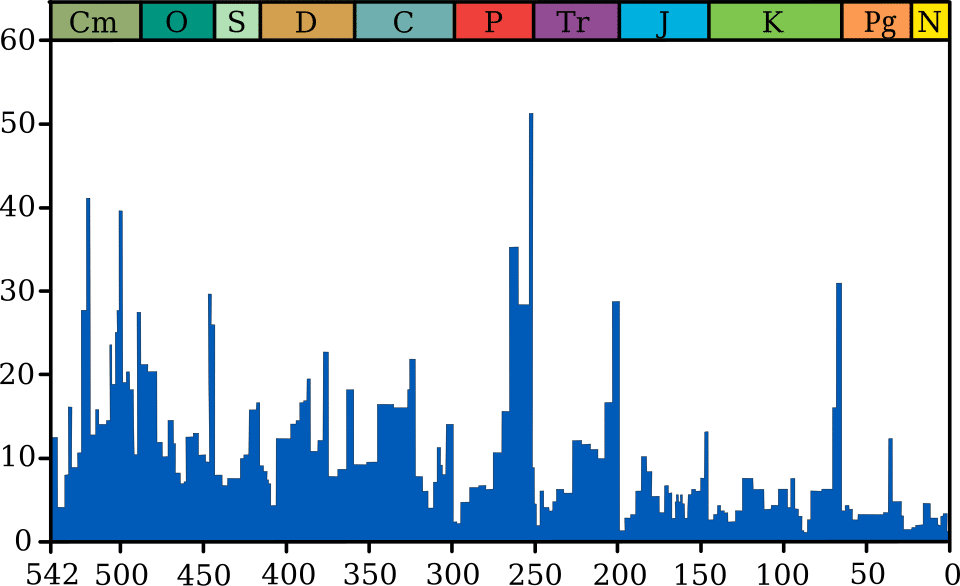
Paleontological analyses of marine invertebrate genera have established that the background extinction rate over the Phanerozoic (the past 541 million years) averages roughly 0.1 to 1.0 species per million species-years, though this rate varies among taxonomic groups and geological intervals.2, 9 Background extinction is the normal cost of doing business in the evolutionary economy: species disappear because they lose in competition with better-adapted rivals, because their habitat contracts, because their prey or hosts vanish, or because they fail to keep pace with changing environmental conditions.
During intervals of background extinction, the rules of Darwinian natural selection appear to apply in a relatively straightforward manner. Species with larger population sizes, broader ecological tolerances, and effective antipredator or competitive strategies tend to persist longer in the fossil record.3, 5 The traits that confer high fitness at the organismal level — efficient resource acquisition, successful reproduction, evasion of enemies — translate reasonably well into greater species longevity. This correspondence between microevolutionary fitness and macroevolutionary persistence is the regime in which natural selection operates most predictably, and it has dominated the majority of geological time.3
Mass extinctions and the Big Five
Mass extinctions are defined as intervals in which extinction rates rise dramatically above the background level, eliminating 75 percent or more of species within a span of roughly one to three million years or less. Raup and Sepkoski's landmark 1982 analysis of marine animal families identified five major mass extinctions in the Phanerozoic, now universally known as the Big Five: the end-Ordovician (approximately 445 million years ago), the Late Devonian (approximately 372 million years ago), the end-Permian (approximately 252 million years ago), the end-Triassic (approximately 201 million years ago), and the end-Cretaceous (approximately 66 million years ago).2 The end-Permian event was by far the most severe, eliminating an estimated 81 percent of marine species and fundamentally restructuring marine ecosystems.1, 9

Each of the Big Five was caused by environmental perturbations of extraordinary magnitude — massive volcanism, asteroid impact, rapid climate change, ocean anoxia, or some combination of these factors — that overwhelmed the adaptive capacities of most organisms.1, 15 The critical insight from decades of paleontological research is that mass extinctions are not simply amplified versions of background extinction. They represent qualitatively different selective regimes in which the rules governing survival change abruptly and profoundly.4, 6
Estimated species loss during the Big Five mass extinctions2, 9
Selectivity during mass extinctions
One of the most important discoveries in extinction research is that mass extinctions are selective — but selective in ways that differ fundamentally from background intervals. During normal times, traits that promote species-level survival tend to mirror those that promote individual fitness: species with large populations, broad diets, and effective competitive abilities persist longer.3 During mass extinctions, these correlations break down. The traits that determine which lineages survive a catastrophe are often decoupled from, or even orthogonal to, the traits that confer success during the intervening millions of years of background evolution.4, 6
David Jablonski's extensive studies of end-Cretaceous marine bivalve and gastropod extinctions provide the clearest demonstration of this principle. During background intervals, broad geographic range at the species level predicted longer species duration, as expected. During the end-Cretaceous mass extinction, however, species-level geographic range lost its protective effect. Instead, survival depended on the geographic range of the entire genus: genera that were widespread across multiple biogeographic provinces survived preferentially, regardless of whether their individual constituent species were widespread or narrowly endemic.5, 14 This finding means that the unit of selection effectively shifted upward in the taxonomic hierarchy during the crisis — from the species to the clade.
Body size is another trait whose relationship to extinction risk changes between background and mass extinction intervals. During normal times, larger-bodied species of marine invertebrates tend to have somewhat longer durations in the fossil record.7 During mass extinctions, the relationship becomes complex and variable. Some mass extinctions preferentially eliminate large-bodied taxa, while others show no consistent body-size selectivity, suggesting that the environmental mechanisms driving each crisis interact with body size in different ways.7, 22 Metabolic rate, trophic level, and habitat specificity have also been identified as factors influencing survival during various mass extinction events, though no single trait profile consistently predicts survival across all five of the major crises.22
Extinction as a macroevolutionary force
The recognition that mass extinctions impose selective regimes qualitatively different from background conditions led Jablonski and others to argue that extinction is not merely a destructive process that prunes the tree of life but a creative macroevolutionary force that shapes its long-term architecture.4, 6 By eliminating dominant groups and breaking apart established ecological relationships, mass extinctions create vacated ecospace that surviving lineages can subsequently exploit through adaptive radiation.
The end-Cretaceous extinction provides the canonical example. The disappearance of non-avian dinosaurs, marine reptiles, ammonites, and numerous other groups removed the ecological dominants of the Mesozoic world and opened opportunities for mammals, birds, teleost fishes, and flowering plants to diversify into niches that had been occupied or suppressed for tens of millions of years.16, 17 Mammals, which had existed as small-bodied, ecologically marginal forms for over 100 million years during the Mesozoic, underwent an explosive radiation in the Paleocene and Eocene, diversifying into bats, whales, primates, carnivores, and dozens of other orders within approximately 10 to 15 million years of the extinction event.17 Without the end-Cretaceous catastrophe, there is no reason to believe that mammals would have achieved ecological dominance.
Similarly, the end-Permian extinction, the most severe of the Big Five, eliminated the dominant Paleozoic marine fauna — including trilobites, rugose corals, and most brachiopod lineages — and paved the way for the Modern Evolutionary Fauna characterized by gastropods, bivalves, echinoids, and bony fishes that dominate marine ecosystems today.9, 10 The Cambrian radiation of animal body plans was itself preceded by the Ediacaran extinctions and environmental upheavals that cleared ecological space for the ancestors of modern animal phyla.18
Microevolutionary fitness versus macroevolutionary survivorship
The distinction between traits that promote success during background intervals and traits that promote survival during mass extinctions has profound implications for evolutionary theory. In the standard Darwinian framework, natural selection operating on individual organisms within populations is the primary engine of evolutionary change. Organisms with heritable traits that enhance survival and reproduction leave more descendants, and their traits spread through populations over generations. This process, operating continuously over millions of years, is expected to produce organisms that are progressively better adapted to their environments.3
Mass extinctions disrupt this expectation. A lineage may accumulate adaptive refinements over tens of millions of years of background selection — becoming superbly adapted to its ecological role — only to be eliminated in a geologically instantaneous catastrophe for reasons entirely unrelated to its competitive ability or ecological fitness.4, 15 Raup memorably described mass extinction as a process in which "bad genes kill you, but bad luck can also kill you." The organisms that survive a mass extinction are not necessarily the best adapted in any Darwinian sense; they are the ones that happen to possess traits — broad geographic range, physiological tolerance of the specific stressor, ecological generalism — that are relevant to surviving the particular catastrophe at hand.6, 15
Jablonski has formalized this insight by distinguishing between "constructive" selectivity, in which mass extinctions actively favour certain trait combinations, and "destructive" selectivity, in which mass extinctions simply reset the evolutionary playing field by eliminating incumbents and removing competitive hierarchies established during background times.4, 8 Both modes have been documented in the fossil record. Some clades survive mass extinctions because they possess genuinely advantageous traits (constructive selectivity), while others simply endure as remnant populations that neither recover their former diversity nor assume new ecological roles — what Jablonski has termed "dead clades walking."8 These zombie lineages persist for millions of years after a mass extinction at reduced diversity before eventually succumbing to background extinction, never having exploited the ecological opportunities created by the crisis.
Recovery and adaptive radiation
The aftermath of a mass extinction typically involves three overlapping phases: a survival interval dominated by ecological generalists and disaster taxa, a recovery interval in which diversity begins to rebuild, and a radiation interval in which new clades diversify to fill vacated and newly created ecological roles.

The pattern of post-extinction radiation is not random. Clades that survived a mass extinction with relatively high diversity and broad geographic distribution tend to dominate the subsequent recovery, but the ecological roles they assume are often dramatically different from those they occupied before the crisis.4, 17 The post-Cretaceous radiation of mammals is instructive: the mammalian lineages that survived the extinction were small-bodied, ecologically unspecialized forms, but their descendants radiated into an extraordinary range of body sizes, habitats, and ecological strategies within a few million years. The ecospace vacated by dinosaurs was not filled by dinosaur-like mammals but by entirely novel adaptive solutions.17
This pattern has led paleontologists to recognize that mass extinctions do not simply reset the evolutionary clock to an earlier state. They redirect the course of evolution along new trajectories that would have been inaccessible without the removal of incumbent competitors. In this sense, mass extinctions are genuinely creative events, though their creativity is retrospective — apparent only in the subsequent radiation — rather than prospective.4, 6
Extinction and the shape of biodiversity
The interplay between extinction and origination over the Phanerozoic has produced the characteristic diversity trajectory visible in the fossil record: a long-term increase in marine animal diversity punctuated by sharp declines at each of the Big Five mass extinctions.10 Analyses of the Paleobiology Database, a comprehensive compilation of fossil occurrence data, have confirmed that this trajectory reflects real biological pattern rather than sampling artefact, though the precise magnitude of diversity changes is sensitive to methods used to correct for uneven preservation and sampling intensity.10
Mass extinctions have been particularly important in reshaping the taxonomic composition of the biosphere. The end-Permian event marks a fundamental transition from the Paleozoic Evolutionary Fauna, dominated by brachiopods, crinoids, rugose and tabulate corals, and trilobites, to the Modern Evolutionary Fauna, dominated by gastropods, bivalves, echinoids, malacostracans, and teleost fishes.9, 10 This transition was not driven by gradual competitive displacement of one fauna by another; rather, the catastrophic elimination of the Paleozoic dominants created the ecological opportunity for the Modern fauna to diversify. Without the end-Permian crisis, the taxonomic structure of today's oceans might look entirely different.
Extinction also shapes biodiversity at the level of evolutionary history. Purvis and colleagues demonstrated that extinction is not distributed randomly across the tree of life but tends to be phylogenetically clustered — closely related species tend to share vulnerability to the same extinction drivers. The loss of an entire clade eliminates not just species but the unique evolutionary history and morphological diversity (disparity) that those species represent, an irreversible loss that origination of new species in other clades cannot replace on any human timescale.21
Modern extinction rates and the sixth extinction
Estimating the current rate of species extinction and comparing it to the background rate derived from the fossil record has become one of the most consequential applications of paleontological data. Pimm and colleagues estimated the pre-human background extinction rate for mammals at approximately 0.1 to 1.0 extinctions per million species-years, based on fossil first and last appearances.13 Against this baseline, current mammalian extinction rates are estimated to be 100 to 1,000 times elevated, even using conservative assumptions that count only documented extinctions and exclude species that are critically endangered but not yet confirmed extinct.12, 13
Barnosky and colleagues compared the severity and rate of current species losses with the Big Five mass extinctions and concluded that, while the current crisis has not yet reached Big Five magnitudes in terms of total species lost, the rate of loss is consistent with a mass extinction trajectory. If currently threatened species (those listed as critically endangered, endangered, or vulnerable by the IUCN) were to go extinct within the next few centuries, the resulting loss would rival or exceed the Big Five in magnitude.11 Ceballos and colleagues reached a similar conclusion using vertebrate species data, finding that the number of species that went extinct in the twentieth century alone would have required between 800 and 10,000 years to disappear at the background rate.12
The comparison between modern and ancient extinction rates requires caution. The fossil record predominantly preserves marine invertebrates with durable skeletons, whereas modern extinction data are most complete for terrestrial vertebrates. The timescales are also different: fossil-derived background rates integrate over millions of years, while modern rates are measured over decades to centuries, making direct comparison methodologically challenging.19, 20 Nevertheless, the convergence of evidence from multiple independent analyses using different taxa, methods, and assumptions strongly supports the conclusion that modern extinction rates are far above the Phanerozoic background and that, without intervention, the current biodiversity crisis will qualify as the sixth mass extinction in Earth's history.11, 12, 13
References
The biodiversity of species and their rates of extinction, distribution, and protection