Overview
- Evolution operates through four primary mechanisms—natural selection, mutation, genetic drift, and gene flow—that collectively drive the adaptation, diversification, and speciation of populations over time.
- Natural selection is the primary creative force in evolution: by systematically favoring heritable variants that improve survival and reproduction, it accumulates beneficial changes across generations to build complex adaptations without foresight or planning.
- When populations become reproductively isolated, the independent accumulation of genetic changes leads to speciation—the origin of new species—while similar selective pressures acting on unrelated lineages can produce strikingly convergent solutions to the same ecological problems.
Evolution proceeds through a set of well-characterized mechanisms that, acting together, drive the adaptation and diversification of life. Mutation generates heritable variation in DNA sequences; natural selection systematically favors variants that enhance survival and reproduction; genetic drift causes random fluctuations in allele frequencies, especially in small populations; and gene flow transfers genetic material between populations through migration and interbreeding.2 These four mechanisms, unified by the Modern Synthesis of the 1930s and 1940s, form the core of evolutionary theory and have been documented through laboratory experiments, field observations, comparative genomics, and mathematical modeling.3, 4, 7
The interplay among these mechanisms produces the two great patterns of evolutionary change: adaptation within lineages and the branching diversification that generates new species. Natural selection is the primary creative force, capable of building complex structures—from the vertebrate eye to the immune system—through the cumulative accumulation of small improvements over many generations.1, 2 When populations become geographically or ecologically isolated, they accumulate independent genetic changes that can eventually prevent interbreeding, producing distinct species. And when unrelated lineages face similar environmental challenges, natural selection can independently craft strikingly similar solutions—a phenomenon called convergent evolution that underscores the predictability of the evolutionary process.9, 10
Variation and selection
The logic of natural selection, first articulated by Charles Darwin in 1859, rests on three observable conditions: individuals within a population vary in their traits, some of that variation is heritable, and different variants differ in their rates of survival and reproduction.1 When these conditions are met, traits that improve fitness become more common in each successive generation while traits that reduce fitness become rarer. The process is non-random—it systematically favors organisms better suited to their environment—but it requires no foresight, planning, or external direction.1, 2

Mutation is the ultimate source of all genetic variation. Point mutations substitute one nucleotide for another; insertions and deletions add or remove stretches of DNA; gene duplications create extra copies of genes that can diverge in function; and whole-genome duplications, which have occurred multiple times in vertebrate and plant evolution, massively expand the raw material available for evolutionary innovation.5, 6 Most mutations are selectively neutral—they neither help nor harm the organism—and a smaller fraction are deleterious. But a significant minority are beneficial, and these are the mutations that natural selection amplifies across generations.6 Because mutation, recombination, and gene duplication continually replenish the pool of genetic variation, selection has a perpetually renewed supply of raw material on which to act. This is what makes cumulative selection possible: each beneficial change provides the platform on which the next improvement can build.2, 5
Genetic drift—the random fluctuation of allele frequencies due to chance events in reproduction—is especially influential in small populations, where sampling effects can cause alleles to be lost or fixed regardless of their fitness consequences.3 Gene flow, the movement of alleles between populations through migration, can introduce novel variation into a population or counteract the divergence produced by selection and drift in isolated populations. The Modern Synthesis, achieved by R. A. Fisher, Sewall Wright, J. B. S. Haldane, Theodosius Dobzhansky, and Ernst Mayr among others, integrated these mechanisms with Mendelian genetics into the theoretical framework that remains the foundation of evolutionary biology.3, 4
Speciation and divergence
Speciation—the process by which one species splits into two or more reproductively isolated lineages—is the mechanism that generates the branching pattern of the tree of life. The most common route is allopatric speciation, in which geographic barriers such as mountain ranges, rivers, or oceanic expanses physically separate populations and prevent gene flow between them. In isolation, the separated populations accumulate independent genetic changes through mutation, selection, and drift. Over time, these changes can produce reproductive incompatibilities—behavioral, mechanical, or genetic barriers that prevent successful interbreeding even if the populations come back into contact.9
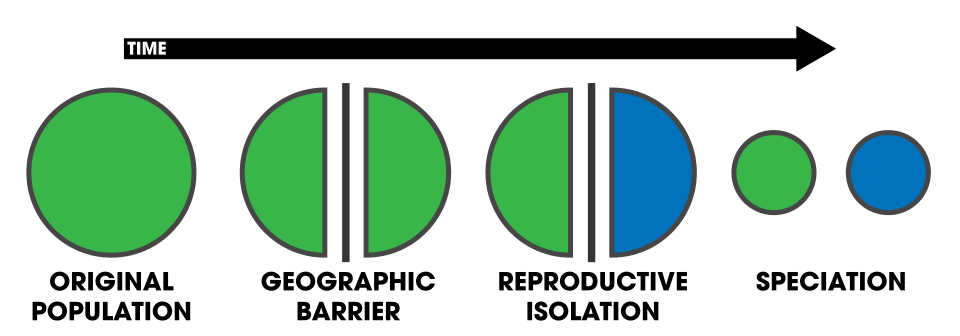
Speciation can also occur without geographic isolation. Sympatric speciation, in which new species arise within a single population, has been documented in cases where strong ecological selection or chromosomal changes create reproductive barriers. In plants, polyploidy—the duplication of the entire genome—can produce a new species in a single generation, because the polyploid offspring are reproductively incompatible with the diploid parent population. Estimates suggest that 15% of angiosperm speciation events and 31% of fern speciation events involve polyploidy.2, 9 Speciation has been directly observed in laboratory populations of bacteria evolving novel metabolic capabilities and in natural plant populations producing new species through genome duplication.8
Convergence and constraint
When unrelated lineages face similar ecological challenges, natural selection can independently produce remarkably similar adaptations—a pattern called convergent evolution. The camera eye has evolved independently at least 40 times across the animal kingdom; echolocation arose independently in bats and toothed whales; powered flight evolved separately in insects, pterosaurs, birds, and bats; and the streamlined body plan of fast-swimming predators has been reinvented by sharks, ichthyosaurs, and dolphins.10 These convergences are not coincidental. They reflect the fact that physics, chemistry, and ecology constrain the space of viable solutions to common survival problems, channeling evolution along a limited number of pathways.2, 10

Convergent evolution is significant for two reasons. First, it demonstrates that natural selection is a powerful and repeatable force, capable of independently arriving at the same functional solutions in lineages separated by hundreds of millions of years of independent evolution. Second, it shows that evolution, while unguided, is not formless: the physical and biological constraints under which organisms evolve impose a deep structure on the diversity of life, making some outcomes far more probable than others.10
Neutral evolution and the role of chance
While natural selection is the primary driver of adaptive change, a substantial proportion of evolutionary change at the molecular level is selectively neutral. Motoo Kimura's neutral theory of molecular evolution, first articulated in 1968 and developed more fully in subsequent decades, demonstrated that the majority of nucleotide substitutions that become fixed in populations are neither beneficial nor harmful but are instead neutral variants that spread through genetic drift alone.11 Kimura showed mathematically that the rate of neutral substitution is equal to the neutral mutation rate, independent of population size—a prediction that has been broadly confirmed by comparative genomic analyses across diverse taxa.11, 13
The neutral theory does not deny the importance of natural selection for adaptation. Rather, it provides the null expectation against which selection is detected: a molecular change is inferred to be under selection only if it deviates significantly from the neutral rate. The ratio of nonsynonymous to synonymous substitutions (dN/dS) in protein-coding genes has become the standard test for this deviation. Genes under strong purifying selection show dN/dS ratios well below 1.0, reflecting the removal of most amino acid changes. Genes under positive selection show elevated ratios, sometimes exceeding 1.0, indicating that amino acid changes are being fixed more rapidly than expected by chance. This framework, integrating neutral drift and selective forces, is central to modern molecular evolution.2, 13
The interplay between drift and selection has practical consequences for how evolution proceeds in different populations. In large populations, natural selection is highly efficient: even slightly beneficial mutations have a high probability of spreading, and slightly deleterious mutations are reliably purged. In small populations, the stochastic effects of drift overwhelm weak selection, allowing mildly harmful mutations to accumulate and potentially beneficial ones to be lost by chance. This population-size dependence helps explain why organisms with consistently small effective population sizes, such as obligate parasites and island endemics, tend to accumulate slightly deleterious mutations and show genome degradation over evolutionary time.3, 12
References
Intense Natural Selection in a Population of Darwin's Finches (Geospizinae) in the Galápagos