Overview
- The hominin clade — the lineage leading to modern humans after the divergence from the common ancestor with chimpanzees — encompassed a surprising diversity of species, with at least 20 to 25 named species spanning roughly seven million years, and multiple lineages frequently coexisting in the same regions and time periods.
- Ancient DNA analysis has revealed 'ghost populations' — archaic human groups known only from their genetic signatures in modern or ancient genomes rather than from fossil remains — demonstrating that hominin diversity was even greater than the fossil record alone suggests and that interbreeding among distinct lineages was a recurrent feature of human evolution.
- As recently as 50,000 to 100,000 years ago, at least five distinct hominin lineages coexisted on Earth — Homo sapiens, Neanderthals, Denisovans, Homo floresiensis, and Homo luzonensis — and the current situation of a single surviving human species is historically exceptional rather than typical.
Modern humans are the sole surviving species of a once-diverse radiation of bipedal apes. Today, Homo sapiens occupies every continent and every major ecological zone, and no other hominin exists to share the planet. This situation is historically anomalous. For most of the seven million years since the lineage leading to humans diverged from the lineage leading to chimpanzees and bonobos, multiple hominin species coexisted — sometimes on the same continent, occasionally in the same region, and in at least some cases interbreeding with one another.1, 2 The fossil record and ancient DNA together reveal a hominin clade of surprising species richness, punctuated by repeated episodes of speciation, dispersal, coexistence, and extinction, a history that the single-species present obscures.16
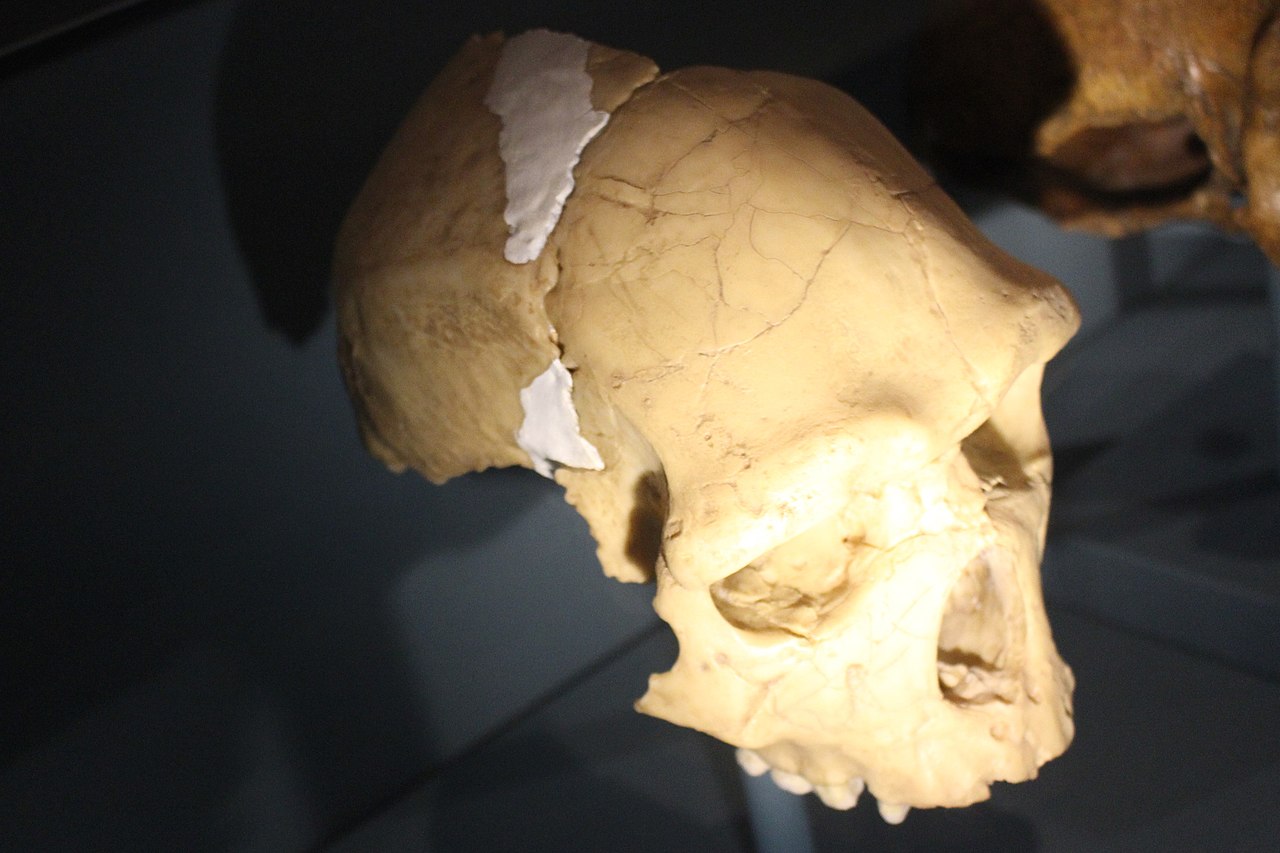
The species richness of the hominin clade
The number of recognised hominin species depends on taxonomic philosophy — splitters recognise more species than lumpers — but most palaeoanthropologists accept at least 20 to 25 named species distributed across several genera. The earliest known hominins include Sahelanthropus tchadensis (approximately 7 million years ago in Chad), Orrorin tugenensis (approximately 6 million years ago in Kenya), and Ardipithecus kadabba and Ardipithecus ramidus (approximately 5.8 to 4.4 million years ago in Ethiopia).7, 8 By the Pliocene (approximately 4 to 2.5 million years ago), multiple australopithecine species occupied eastern and southern Africa, including Australopithecus anamensis, Australopithecus afarensis, Australopithecus africanus, Australopithecus garhi, and the robust australopithecines (Paranthropus aethiopicus, Paranthropus boisei, and Paranthropus robustus).1
The genus Homo emerged roughly 2.5 to 2.8 million years ago and itself contained considerable diversity: Homo habilis, Homo rudolfensis, Homo erectus (including the variant sometimes classified as Homo ergaster), Homo antecessor, Homo heidelbergensis, Homo rhodesiensis (or Homo bodoensis, as some recent taxonomic proposals suggest), Homo neanderthalensis, Denisovans (whose taxonomic status remains debated), Homo floresiensis, Homo luzonensis, Homo naledi, and Homo sapiens.1, 2, 10, 11, 12 At numerous points in this history, multiple species coexisted. Around 1.8 million years ago, for example, at least three hominin species — Homo erectus, Paranthropus boisei, and either Homo habilis or Homo rudolfensis — occupied the same landscape around Lake Turkana in Kenya.1, 2
Late Pleistocene coexistence
The diversity of the hominin clade was not confined to the distant past. As recently as 50,000 to 100,000 years ago, at least five distinct hominin lineages coexisted on Earth. Homo sapiens had emerged in Africa by at least 300,000 years ago, as evidenced by fossils from Jebel Irhoud in Morocco and Omo Kibish in Ethiopia, and had begun dispersing into Eurasia.9 Neanderthals occupied Europe and western Asia. Denisovans, known primarily from DNA extracted from fragmentary fossils in Denisova Cave in Siberia and Baishiya Karst Cave in Tibet, ranged across eastern Eurasia.4, 5 Homo floresiensis, a diminutive species standing roughly one metre tall with a brain volume of approximately 426 cc, inhabited the Indonesian island of Flores until at least 50,000 years ago.11 And Homo luzonensis, discovered in Callao Cave in the Philippines and described in 2019, represents yet another Late Pleistocene lineage with a mosaic of primitive and derived features.10
The extinction of these non-sapiens lineages between roughly 50,000 and 15,000 years ago — through some combination of competitive exclusion, environmental change, and absorption through interbreeding — left Homo sapiens as the sole surviving hominin. This outcome was not inevitable: for most of hominin history, multi-species coexistence was the norm, not the exception.1, 2
Ghost populations from ancient DNA
The revolution in ancient DNA analysis, pioneered by Svante Paabo and colleagues, has revealed that the true diversity of archaic hominins was even greater than the fossil record suggests. The sequencing of the Neanderthal genome in 2010 demonstrated that non-Africans carry approximately 1 to 4 percent Neanderthal DNA, proving that Homo sapiens and Neanderthals interbred when they encountered each other after the out-of-Africa dispersal.3 The discovery of Denisovans was itself a triumph of ancient DNA: the first Denisovan individual was identified not from morphological analysis of a fossil but from DNA extracted from a finger bone fragment too small to provide meaningful anatomical information. Subsequent analysis revealed that modern Melanesians and Aboriginal Australians carry approximately 3 to 6 percent Denisovan DNA, while some Southeast Asian populations carry smaller amounts from a distinct Denisovan lineage, suggesting at least two separate episodes of Denisovan-sapiens admixture.4, 14
Perhaps most remarkably, genomic analysis has identified ghost populations — archaic hominin groups that left genetic traces in modern or ancient genomes but have no known fossil representation. Durvasula and Sankararaman (2020) identified signals of introgression from an unknown archaic hominin in West African populations, dating the admixture to approximately 43,000 years ago and estimating that the ghost population diverged from the ancestor of humans and Neanderthals roughly 625,000 years ago.17 Chen and colleagues (2020) independently found evidence of a previously unrecognised archaic lineage contributing DNA to African populations, with some of this genetic material also detected in Neanderthals, suggesting a complex web of gene flow among multiple hominin lineages.18
Additional ghost populations have been inferred from ancient DNA evidence in other regions. Analysis of Denisovan DNA segments in modern Southeast Asian populations suggests admixture with at least two genetically distinct Denisovan-like groups, one of which may represent a previously unknown archaic population that diverged from known Denisovans hundreds of thousands of years ago.14, 16 These findings indicate that the Late Pleistocene hominin landscape was populated by more distinct lineages than the handful represented by named fossil species, and that the boundaries between species were permeable enough to allow gene flow when populations came into contact.15, 16
Interbreeding and reticulate evolution
The evidence for pervasive interbreeding among archaic hominin lineages has fundamentally altered the conceptual framework of human evolution. The traditional model of a branching tree, in which lineages diverge and never reconnect, has given way to a model of reticulate evolution — a braided stream or network in which lineages split, diverge, and periodically rejoin through hybridisation. An early modern human from Pestera cu Oase in Romania, dated to approximately 40,000 years ago, carried 6 to 9 percent Neanderthal DNA, indicating that he had a Neanderthal ancestor within the previous four to six generations.13 The high-coverage genome from the Denisova Cave revealed that the Denisovan individual had received gene flow from Neanderthals and from an unknown archaic population, demonstrating that gene flow was not limited to sapiens-Neanderthal contact but occurred among multiple hominin lineages.5
The Sima de los Huesos fossils from Atapuerca, Spain, dating to approximately 430,000 years ago, illustrate the complexity further. These fossils have Neanderthal-like morphology but yielded mitochondrial DNA more closely related to Denisovans, while nuclear DNA analysis confirmed they are on the Neanderthal lineage.6 This discordance between mitochondrial and nuclear ancestry is a hallmark of past hybridisation events, suggesting that the Middle Pleistocene hominin population of Europe was already a product of complex gene flow among divergent lineages.6, 16
Implications for understanding human evolution
The recognition of archaic hominin diversity and pervasive interbreeding has several important implications. First, it demonstrates that Homo sapiens did not evolve in isolation but emerged from and interacted with a diverse community of related species, acquiring adaptive genetic variants — such as the Denisovan-derived EPAS1 allele that aids Tibetan high-altitude adaptation — through hybridisation.4, 16 Second, it challenges the concept of sharp species boundaries in the hominin clade, since species that had been diverging for hundreds of thousands of years were nonetheless capable of producing fertile offspring. Third, it underscores that the modern condition of a single human species is a recent anomaly: for most of the past seven million years, the planet supported multiple coexisting hominin species, and the causes of the others' extinction remain among the most consequential questions in palaeoanthropology.1, 2, 16
The hominin clade was not a ladder leading inevitably to modern humans but a bush of diverse, coexisting lineages, most of which went extinct without issue. The survival of Homo sapiens as the sole representative of this once-rich radiation is an outcome that demands explanation, not an inevitable trajectory. Understanding why we are alone requires understanding a past in which we were not.1, 2
Causes of archaic extinction
The disappearance of all non-sapiens hominin lineages within a geologically brief window — roughly 50,000 to 15,000 years ago — remains one of the most consequential and debated events in human evolutionary history. Several hypotheses have been proposed, and the available evidence suggests that no single factor was responsible. Climate change, particularly the extreme oscillations of the Late Pleistocene glacial-interglacial cycles, may have stressed archaic populations that had adapted to more stable conditions. Competitive exclusion by expanding Homo sapiens populations, who may have possessed superior technology, more extensive trade networks, or more flexible social organization, likely contributed as well.1, 2
The genomic evidence for interbreeding complicates simple replacement models: rather than being entirely replaced, archaic lineages were partially absorbed into expanding sapiens populations through hybridisation, with their genetic legacy persisting in modern genomes even as their distinct populations ceased to exist as independent entities. Disease transmission from incoming sapiens populations, analogous to the devastating epidemics that accompanied colonial encounters in the historical period, has also been proposed as a contributing factor, though direct evidence remains limited.3, 16 The most likely scenario is that multiple factors — climate, competition, assimilation, and possibly disease — operated in combination, with different factors predominating in different regions and for different archaic lineages.1, 16
References
Denisova admixture and the first modern human dispersals into Southeast Asia and Oceania
The age of the hominin fossils from Jebel Irhoud, Morocco, and the origins of the Middle Stone Age
Insights into human genetic variation and population history from 929 diverse genomes