Overview
- Endocasts, whether natural casts preserved in sediment, physical molds, or virtual reconstructions from CT scanning, preserve the size, shape, and surface morphology of the brain's outer surface, providing the only direct window into brain evolution across millions of years of hominin history.
- Pioneering work by Ralph Holloway, Dean Falk, and Emiliano Bruner has revealed that brain reorganization, particularly frontal lobe restructuring, parietal expansion, increasing lateralization, and cerebellar growth, was at least as important as raw size increase in hominin cognitive evolution.
- Key specimens from the Taung child to Homo naledi demonstrate that brain organization and brain size could evolve on partly independent trajectories, with some small-brained hominins exhibiting Homo-like frontal morphology and modern humans distinguished by a species-specific expansion of the parietal lobes.
The brains of extinct hominins do not fossilize. Soft neural tissue decays rapidly after death, leaving no direct trace in the geological record. Yet the inner surface of the braincase faithfully records the size, overall shape, and many surface features of the brain it once housed, and under favorable circumstances this information can be recovered. An endocast is a cast of the endocranial cavity — the hollow interior of the skull that contained the brain, its meninges, and the cerebrospinal fluid surrounding them. Whether formed naturally by sediment filling a braincase, manufactured in the laboratory from latex or plaster, or digitally reconstructed from computed tomography (CT) scans, endocasts have been the primary source of evidence about brain evolution across the seven-million-year span of the hominin fossil record.1, 7 They cannot reveal the fine cellular architecture of the cortex or the density of synaptic connections, but they preserve a surprising amount of information about brain volume, lobe proportions, cortical folding patterns, vascular drainage, and hemispheric asymmetry — features that, when interpreted carefully, illuminate the evolutionary trajectory from an ape-sized brain to the organ that defines Homo sapiens.7, 15
What endocasts are
The most famous natural endocast in paleoanthropology is the one that launched the entire field of hominin paleontology. In 1924, the anatomist Raymond Dart received a box of fossils from a limestone quarry at Taung in what is now South Africa's North West Province. Among them was a small, rounded object: a natural endocast of a juvenile primate, formed when calcium-rich sediment had infiltrated and hardened inside the braincase after death, faithfully preserving the size, shape, and surface impressions of the brain.2 Dart recognized that the endocast displayed features intermediate between apes and humans, and in February 1925 he published his description of Australopithecus africanus in Nature, arguing that the Taung child represented a transitional form between living apes and the human lineage.2 The endocast was central to his case: its size (later redetermined at approximately 405 cubic centimeters for the juvenile and an estimated 440 cc for the adult by Holloway in 1970) exceeded that of similarly sized apes, and its surface morphology suggested a brain more advanced than that of any known great ape.2, 3
Natural endocasts like the Taung specimen are geological accidents, formed only when sediment infiltrates an intact braincase and lithifies before the cranial bones collapse or disintegrate. They are correspondingly rare. For the vast majority of hominin crania, the endocranial surface must be recorded by other means. Beginning in the mid-twentieth century, paleoneurologists produced physical endocasts by coating the interior of skulls with layers of latex rubber or filling them with plaster, creating detailed molds that could be measured, compared, and curated. Ralph Holloway, working at Columbia University from the 1960s onward, assembled the largest collection of such physical endocasts in the world, covering virtually every major hominin cranium then known.7, 21
The revolution in endocast methodology came with computed tomography. By the late 1990s and early 2000s, CT scanning and three-dimensional digital reconstruction made it possible to generate virtual endocasts from fossil crania without physically touching the specimen — a critical advantage for fragile or fragmentary fossils. Virtual endocasts can be rotated, measured, sectioned, and compared computationally with far greater precision than physical molds, and they permit the application of geometric morphometric methods that quantify shape differences across entire surfaces rather than relying on a limited set of linear measurements.8, 12 High-resolution synchrotron CT scanning has further enhanced the technique, revealing surface details as fine as individual meningeal vessel impressions on specimens such as the Australopithecus sediba cranium MH1.14
What endocasts preserve and what they cannot
An endocast is not a replica of the brain. It is a cast of the endocranial cavity, which means that it records the outer surface of the brain only insofar as that surface makes contact with the inner table of the skull, mediated by the dural membranes (meninges) that envelop the brain. In humans and other primates, the correspondence between cortical surface features and endocranial surface features varies by region. In the frontal and temporal lobes, the cortical sulci and gyri often leave clear impressions on the endocranial surface, and these impressions are typically well reproduced on endocasts. In the parietal and occipital regions, by contrast, the dural membranes are thicker and more loosely adherent to the cortex, and cortical detail is correspondingly attenuated or absent on the endocranial surface.10, 15
Endocasts reliably record several categories of information. Endocranial volume, the most commonly cited metric, can be measured to within approximately 5% accuracy on well-preserved crania and serves as a proxy for brain mass.7, 17 Overall brain shape — the relative proportions of the frontal, parietal, temporal, and occipital lobes — is preserved in the gross morphology of the endocast. Major sulci, particularly the central sulcus (separating the frontal and parietal lobes), the lateral sulcus (Sylvian fissure), and the lunate sulcus (marking the anterior boundary of the primary visual cortex), can often be identified, although their interpretation has been the subject of prolonged debate.5, 10 Meningeal vessels, particularly the middle meningeal artery, leave branching impressions on the endocranial surface that record the vascular supply to the dural membranes. The venous sinuses — large drainage channels within the dura — are recorded as grooves or ridges and have been used to reconstruct patterns of cranial venous drainage in extinct hominins.6
What endocasts emphatically cannot reveal is the microstructural architecture of the cortex: the number and density of neurons, the extent of dendritic arborization, the thickness and laminar organization of cortical layers, and the connectivity between brain regions. These features are invisible on the endocranial surface. Endocasts therefore provide information about brain size and gross morphology but not about the detailed neuronal circuitry that underlies cognition.15 This limitation is fundamental and imposes a ceiling on what paleoneurology can contribute to understanding the cognitive capacities of extinct species. As Holloway and colleagues cautioned in their comprehensive review, endocasts are necessary but not sufficient evidence for reconstructing the evolution of the mind.1, 15
Ralph Holloway and the reorganization hypothesis
No single researcher shaped the field of paleoneurology more profoundly than Ralph Holloway, who spent six decades at Columbia University studying hominin endocasts. In a series of publications beginning in 1966, Holloway challenged the then-prevailing assumption that brain size was the most important variable in human brain evolution. He argued that the critical changes in hominin brain evolution were organizational rather than merely volumetric: the internal restructuring of the brain, including the expansion of association cortex at the expense of primary sensory cortex, the elaboration of the frontal lobes, and the development of hemispheric asymmetry, mattered as much as or more than the overall increase in cranial capacity.4
Holloway's 1966 paper in the American Anthropologist laid out this position with characteristic directness. He argued that attempts to infer cognitive ability from raw endocranial volume were misguided because they overlooked reorganization — the shifting quantitative relationships among brain subsystems — as a driver of behavioral evolution.4 A brain of a given volume could vary enormously in its functional capacities depending on how that volume was allocated among cortical regions. Two species with identical endocranial volumes might differ dramatically in their cognitive abilities if one had a larger proportion of its cortex devoted to higher-order association areas and a smaller proportion devoted to primary visual or olfactory processing.1, 4
Holloway applied this framework directly to the australopithecine endocasts. In his 1970 redetermination of the Taung endocast volume, he established that the juvenile specimen had an endocranial capacity of approximately 405 cc, implying an adult volume of approximately 440 cc — modestly larger than the average chimpanzee brain but well below the range of later Homo.3 More consequentially, he argued that even at this small volume, the australopithecine brain showed evidence of reorganization in the posterior region: specifically, he contended that the lunate sulcus, which marks the anterior boundary of the primary visual cortex (area V1), was positioned more posteriorly in australopithecines than in extant great apes, implying that the visual cortex had been reduced in relative size and that the adjacent parietal association cortex had expanded.1, 3 This claim became one of the longest-running debates in paleoanthropology, with Dean Falk arguing the opposing position that the lunate sulcus in australopithecines occupied a more anterior, ape-like position.5
Holloway's monumental 2004 volume, The Human Fossil Record, Volume 3: Brain Endocasts, co-authored with Douglas Broadfield and Michael Yuan, provided the first comprehensive illustrated catalogue of every major hominin endocast then known, complete with standardized measurements, photographs, and morphological descriptions.7 This reference work remains the foundation of comparative paleoneurology and is cited in virtually every subsequent study of hominin brain evolution.
Dean Falk and the radiator hypothesis
Dean Falk, working initially at the University of Puerto Rico and later at Florida State University, became the second towering figure in modern paleoneurology. Her early work focused on the same australopithecine endocasts that Holloway had studied, but she reached different conclusions about their surface morphology. In 1980, Falk published a detailed reanalysis of the South African australopithecine endocasts, comparing their sulcal patterns to those of humans, gorillas, and chimpanzees.5 She identified the lunate sulcus of the Taung endocast in a position she interpreted as ape-like, placing it within one standard deviation of the chimpanzee mean, and argued that the australopithecine brain had not undergone the posterior reorganization that Holloway claimed.5 The ensuing debate between Holloway and Falk over the position of the lunate sulcus on the Taung endocast persisted for decades and became one of the most prominent controversies in physical anthropology, with each researcher publishing multiple rebuttals and counter-rebuttals through the 1980s and 1990s.10, 16
Falk's most distinctive contribution, however, was the radiator hypothesis, published in Behavioral and Brain Sciences in 1990.6 She observed that hominin endocasts preserve not only cortical surface impressions but also the impressions of meningeal vessels and venous sinuses — the drainage channels through which blood exits the cranium. By studying the patterns of these vascular impressions across hominin evolution, Falk identified a striking difference between robust australopithecines (such as Paranthropus) and the lineage leading to Homo. The robust australopithecines had prominent occipital and marginal sinuses, a venous drainage pattern shared with great apes, in which blood from the brain drained primarily through large sinuses at the back and sides of the cranium. The gracile australopithecines and early Homo, by contrast, showed an increasingly elaborate network of small emissary veins connecting the intracranial venous sinuses to the external venous plexus of the scalp.6
Falk proposed that this expanded network of emissary veins functioned as a cranial radiator, selectively cooling the brain by allowing heat to dissipate through the scalp veins when body temperature rose during physical exertion in hot environments. She argued that this cooling system was a prerequisite for brain expansion: without an effective mechanism for dissipating the metabolic heat generated by a large brain, selection for increased brain size would have been constrained. The evolution of the cranial venous radiator in the gracile australopithecine-to-Homo lineage thus served as a "prime releaser" that removed a thermal constraint on brain expansion, enabling the dramatic encephalization that characterizes the genus Homo.6 While the radiator hypothesis has been debated and its precise mechanism questioned, it drew attention to a category of endocast evidence — vascular impressions — that had been largely neglected in previous paleoneurological analyses.
In a comprehensive 2014 review, Falk synthesized decades of work on sulcal interpretation in hominin endocasts, acknowledging the difficulty of identifying sulci on specimens where meningeal impressions, venous channels, and cortical folding patterns can be confused, and calling for more rigorous methodological standards in the field.10
Emiliano Bruner and the parietal expansion trajectory
Beginning in the early 2000s, the Spanish paleoneurologist Emiliano Bruner brought a new methodological approach to the field: geometric morphometrics, a set of statistical techniques that quantify the shape of biological structures using coordinates of anatomical landmarks and the deformations required to transform one shape into another. Applied to endocasts, geometric morphometrics offered a way to move beyond the measurement of individual dimensions or the subjective identification of sulci toward a holistic, quantitative characterization of brain shape variation across species.8
Bruner's landmark 2004 study in the Journal of Human Evolution applied two-dimensional geometric morphometrics to the lateral profiles of endocasts from multiple species of the genus Homo. The analysis revealed a clear bimodal pattern of variation: an "archaic" pole, encompassing Homo erectus, H. heidelbergensis, and Neanderthals, was characterized by relatively elongated, low-vaulted endocasts with prominent occipital poles, while a "modern" pole, corresponding to Homo sapiens, was defined by a distinctive globular shape with pronounced bulging of the parietal region and a relatively shortened occipital pole.8 The parietal bulging that distinguished the modern human endocast was not merely a consequence of overall brain expansion; it represented a species-specific change in brain proportions, with the superior parietal lobule and the adjacent precuneus region expanding disproportionately relative to other brain regions.8, 9
Bruner subsequently developed the concept of "parietal humans" to describe the uniquely expanded parietal cortex of Homo sapiens. In a series of publications culminating in a major review in Brain, Behavior and Evolution in 2018, he documented that the parietal expansion visible on endocasts correlated with functional regions of the brain involved in visuospatial integration, body awareness, tool use planning, and working memory.9, 19 Neanderthals and modern humans had similar overall brain volumes, but they differed markedly in brain shape: Neanderthals retained the elongated endocranial morphology of archaic Homo, while modern humans displayed the characteristic parietal bulging and occipital retraction.8, 12, 19 This finding suggested that brain reorganization continued well after maximum brain volume had been achieved, and that the cognitive differences between Neanderthals and modern humans may have been rooted in differences in parietal lobe proportions and the cognitive functions they subserve rather than in differences in overall brain size.9
Key specimens and discoveries
The history of paleoneurology can be traced through a succession of key specimens, each of which advanced understanding of how the hominin brain changed over evolutionary time.

Timeline of major endocast discoveries and milestones2, 4, 6, 8, 11, 14
The Taung child endocast, at approximately 405 cc for the juvenile specimen, provided the foundation for all subsequent work on australopithecine brain morphology.2, 3 KNM-ER 1470, a cranium discovered by Bernard Ngeneo at Koobi Fora in 1972 and attributed to Homo rudolfensis, had an endocranial volume of approximately 750 cc, documenting the first major brain size increase in the genus Homo and providing critical evidence for the expansion of the frontal lobes beyond the australopithecine range.7, 17 KNM-WT 15000, the "Turkana Boy," a remarkably complete Homo erectus skeleton from West Turkana dated to approximately 1.5 million years ago, preserved a cranium with an endocranial volume of approximately 880 cc, establishing that brain size continued to increase substantially within H. erectus.7 The Dmanisi crania from the Republic of Georgia, dated to approximately 1.8 million years ago and representing among the earliest hominin dispersals out of Africa, showed endocranial volumes ranging from approximately 545 to 730 cc, demonstrating that early Homo migrated across continents with brains well below the later H. erectus average.7, 17
Frontal lobe reorganization and the Broca’s cap debate
Among the most consequential features that endocasts preserve are the surface impressions of the inferior frontal gyrus, the region that in modern humans contains Broca's area — a critical cortical region involved in speech production, syntactic processing, and certain aspects of motor planning. On well-preserved endocasts, the third frontal convolution can leave a visible prominence, sometimes called the "cap" of Broca's area, that has been used to infer the presence or absence of language-related cortical architecture in extinct hominins.1, 10

Holloway was among the first to identify what he interpreted as a Broca's cap on australopithecine and early Homo endocasts, arguing that the morphology of this region in specimens such as KNM-ER 1470 showed a Homo-like configuration distinct from the pattern seen in great apes.1, 7 The inference was tantalizing: if early Homo already possessed a Broca's area configuration comparable to that of modern humans, it might indicate that the neural substrate for language was present far earlier than the archaeological evidence for symbolic behavior would suggest. However, this interpretation has been vigorously debated. The correspondence between a surface prominence on an endocast and the underlying cytoarchitectonic boundaries of Broca's area (Brodmann areas 44 and 45) is imprecise, and the boundary between what constitutes an ape-like versus a human-like configuration of the inferior frontal gyrus on an endocast is not clearly defined.10, 15
More recently, the endocast evidence from Australopithecus sediba has complicated the picture further. Carlson and colleagues' 2011 analysis of the MH1 endocast, generated from high-resolution synchrotron CT data, revealed that this small-brained australopith (approximately 420 cc) displayed frontal lobe morphology with features foreshadowing the human condition, including a posterior repositioning of the olfactory bulbs and an orbitofrontal configuration that aligned more closely with Homo endocasts than with those of other australopithecines.14 This finding suggested that neural reorganization in the frontal lobes — particularly changes in the orbitofrontal region involved in decision-making and social behavior — may have begun before significant brain enlargement, in lineages close to the origin of the genus Homo.14
Hemispheric asymmetry is another frontal lobe feature visible on endocasts. Modern humans typically display a left-occipital, right-frontal petalial pattern (the "Yakovlevian torque"), in which the right frontal lobe protrudes slightly beyond the left and the left occipital lobe protrudes slightly beyond the right. This asymmetry is associated with handedness and hemispheric lateralization for language. Holloway identified similar patterns of petalial asymmetry on several early Homo endocasts, interpreting them as evidence that hemispheric specialization for language and handedness was established early in the evolution of the genus Homo.1, 7, 21
Parietal expansion across the genus Homo
While much of the early paleoneurological debate focused on the frontal lobes and the lunate sulcus, Bruner's geometric morphometric work redirected attention to the parietal lobes as the region of the brain that most distinctively differentiates modern humans from all other hominins, including Neanderthals. His analyses showed that the superior parietal lobule, a region of the cortex involved in integrating sensory information, spatial orientation, attention, and the planning of complex hand movements, underwent a disproportionate expansion in the Homo sapiens lineage that is not seen in any other hominin species.8, 9, 19
The trajectory of parietal expansion can be traced across the endocranial record. Australopithecine endocasts show relatively small, flat parietal regions, consistent with the ape-like proportions of their brains. Early Homo endocasts, such as KNM-ER 1470, display modest parietal enlargement accompanying the general increase in brain size. Homo erectus and H. heidelbergensis endocasts show further expansion, but retain the elongated, low-vaulted shape characteristic of archaic Homo. Neanderthal endocasts, despite their large absolute volumes (averaging approximately 1,400 cc), preserve this archaic shape with wide, laterally expanded but relatively flat parietal regions.8, 19 It is only in Homo sapiens that the parietal lobes undergo the characteristic dorsal expansion and midsagittal bulging that produces the globular endocranial shape distinctive of our species.8, 12
Neubauer, Hublin, and Gunz provided the most detailed temporal framework for this process in their 2018 Science Advances study. Using virtual endocasts from 20 Homo sapiens crania spanning the last 300,000 years, they demonstrated that the earliest H. sapiens already possessed brain volumes within the modern range but retained the elongated endocranial shape of archaic Homo. The globular shape characteristic of living humans, driven primarily by parietal and cerebellar bulging, emerged gradually over the subsequent 200,000 years, reaching the modern condition only within the last 35,000 to 100,000 years.12 This finding indicated that brain shape and brain size evolved on different timescales, with the distinctive globularity of the modern human brain representing a relatively recent evolutionary acquisition that was not a simple by-product of reaching a given brain volume.12
Cerebellar evidence
The cerebellum has historically received less attention than the cerebral cortex in paleoneurological studies, in part because the cerebellar fossa on endocasts is often poorly preserved and in part because the cerebellum was long regarded primarily as a motor coordination structure with limited relevance to higher cognition. This assessment has changed dramatically. Neuroscience research in the past two decades has established that the cerebellum is involved in a wide range of cognitive functions, including language, working memory, planning, and social cognition, and that its posterior lobes have extensive connections with prefrontal and parietal association cortex.13, 18
Barton and Venditti's 2014 study in Current Biology provided the most compelling evolutionary evidence for cerebellar importance. Using phylogenetic comparative methods to detect rate changes in brain component evolution across the primate phylogeny, they showed that the cerebellum underwent rapid size increase throughout the evolution of apes, expanding significantly faster than predicted by the corresponding change in neocortex size.13 Humans and other great apes deviated significantly from the general primate trend, in which neocortex and cerebellum tend to scale in tandem, by having relatively larger cerebella than expected for their neocortex size. In humans specifically, the cerebellum contains approximately four times more neurons than the neocortex, and the great ape lineage shows accelerated cerebellar expansion that parallels but is partly independent of neocortical expansion.13
On endocasts, cerebellar expansion is reflected in the size and projection of the cerebellar fossa, which Neubauer and colleagues identified as one of the two principal shape changes (along with parietal bulging) that drove the evolution of modern human brain globularity over the last 300,000 years.12 The gradual increase in cerebellar projection visible in the H. sapiens endocranial record aligns with Barton and Venditti's phylogenetic finding that cerebellar evolution in apes was rapid and functionally significant, and suggests that the cognitive evolution of modern humans involved coordinated changes in both cortical and cerebellar structures.12, 13
The Homo naledi case
Perhaps no recent specimen has challenged conventional thinking about the relationship between brain size and brain organization more forcefully than Homo naledi. Described in 2015 from the Rising Star cave system in South Africa and dated to between 236,000 and 335,000 years ago, H. naledi had an endocranial volume of only 465 to 610 cc across the known specimens — within the range of australopithecines and well below that of contemporaneous Homo sapiens.20, 11
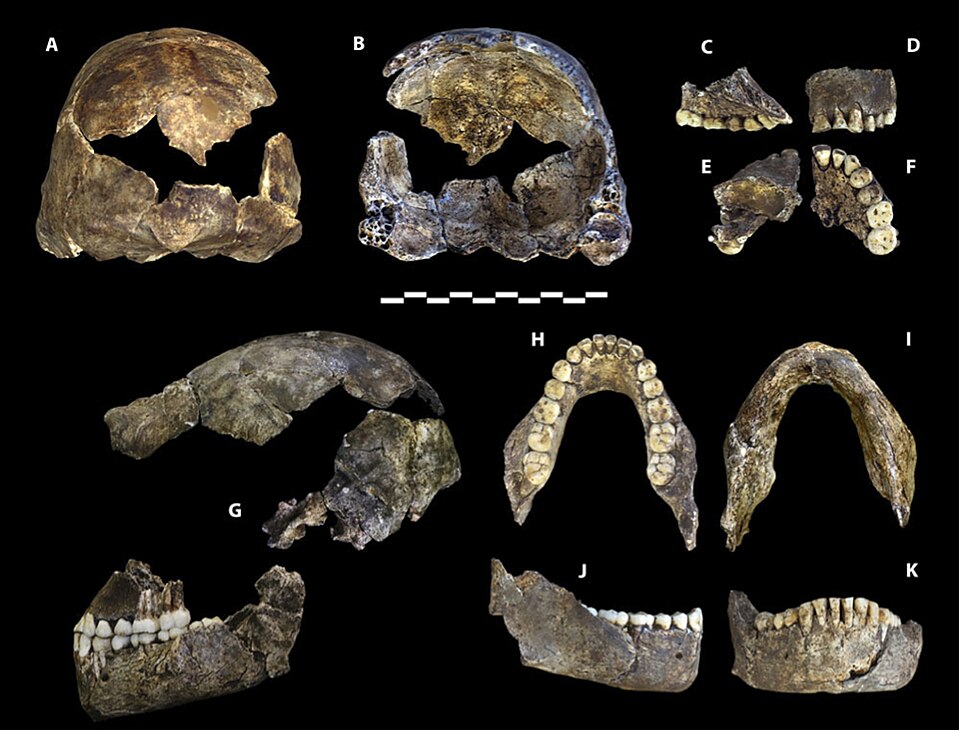
In 2018, Holloway and colleagues published a detailed analysis of the H. naledi endocasts from the Dinaledi Chamber. Despite their small size, the endocasts revealed frontal lobe morphology more similar to that of other Homo species than to australopithecines. The third frontal convolution, the region containing Broca's area in modern humans, displayed a configuration that Holloway and colleagues characterized as derived and Homo-like, with a prominent orbital cap and a distinct pattern of convolutions in the inferior frontal gyrus.11 The overall shape of the frontal lobes, while scaled to a much smaller volume, was not simply a miniaturized australopithecine brain but rather a small brain with Homo-like organizational features.11
The H. naledi endocasts also showed evidence of left-occipital, right-frontal petalial asymmetry, the pattern associated with hemispheric lateralization in modern humans. This asymmetry, combined with the Homo-like frontal morphology, suggested that brain reorganization toward the Homo pattern could proceed without the large brain size increases that traditionally define the genus.11 The implication was profound: brain organization and brain volume could evolve on at least partly independent trajectories, and the cognitive adaptations that distinguish Homo from earlier hominins may have been driven as much by how the brain was internally reorganized as by how large it grew.11, 21
The H. naledi findings resonated with earlier observations from Australopithecus sediba, where Carlson and colleagues had similarly found frontal lobe features foreshadowing the Homo condition in a brain of only 420 cc.14 Taken together, these two specimens suggested that the neural reorganization of the frontal lobes toward a more Homo-like configuration was not a consequence of brain expansion but rather preceded or accompanied it, potentially representing an early step in the evolutionary differentiation of the Homo lineage from australopithecine ancestors.11, 14
Limitations and future directions
Endocasts remain indispensable tools for studying brain evolution, but the field faces persistent methodological challenges. The identification of sulci on endocasts is inherently subjective: what one researcher interprets as the impression of a cortical sulcus, another may read as a meningeal vessel trace or a post-mortem artifact. The lunate sulcus debate between Holloway and Falk persisted for decades in part because the endocranial surface evidence was genuinely ambiguous, and advances in CT resolution have not fully resolved such interpretive difficulties.10, 16 Inter-observer reliability in sulcal identification remains a concern, and few studies have systematically tested how well different researchers agree in their readings of the same specimens.10
The relationship between endocast morphology and the underlying brain is also more complex than early paleoneurologists assumed. The meninges, cerebrospinal fluid, and arachnoid granulations all occupy space between the cortical surface and the endocranial wall, and the degree of cortical-to-endocranial correspondence varies with age, pathology, and brain region.15 Modern comparative neuroimaging, which can directly compare brain MRI scans with CT-derived endocasts in living primates, is beginning to calibrate these relationships empirically, providing a firmer foundation for interpreting the fossil record.18
Geometric morphometrics has dramatically improved the quantitative rigor of endocast comparisons, but the available fossil sample remains small: the total number of hominin crania sufficiently preserved to yield reliable endocasts numbers in the low hundreds across all species and all time periods.7, 22 Statistical power for detecting subtle shape differences between species or tracking shape change within lineages is therefore limited, and the addition of even a single new well-preserved cranium can substantially alter existing interpretations. New discoveries, improved scanning technologies, and the growing application of machine learning to endocast analysis are likely to expand the analytical toolkit of paleoneurology in the coming decades, but the fundamental constraint — the rarity of well-preserved hominin braincases — will remain.15
Despite these limitations, the endocranial record has established several findings of lasting significance. Brain reorganization, not merely brain expansion, has been a defining feature of hominin evolution since at least the australopithecine grade.4, 14 The frontal lobes underwent restructuring in the direction of the Homo pattern before brain size increased dramatically.11, 14 The parietal lobes expanded disproportionately and specifically in Homo sapiens, producing a brain shape distinct from all other hominins including Neanderthals.8, 9, 12 The cerebellum underwent rapid evolutionary expansion in apes and humans that was not a simple by-product of neocortical growth.13 And brain size and brain organization could evolve on partly decoupled timescales, as the cases of A. sediba and H. naledi demonstrate.11, 14 Together, these findings have transformed the study of human brain evolution from a narrative centered on the progressive enlargement of the cranial vault into a more nuanced account of how the internal architecture of the brain was reshaped, region by region and function by function, across the long arc of hominin history.
References
Cranial capacity, neural reorganization, and hominid evolution: a search for more suitable parameters
Endocasts: possibilities and limitations for the interpretation of human brain evolution
New high-resolution computed tomography data of the Taung partial cranium and endocast and their bearing on metopism and hominin brain evolution
Morphological differences in the parietal lobes within the human genus: a neurofunctional perspective