Overview
- Over roughly 7 million years of hominin evolution, the brain tripled in size from approximately 350 cubic centimeters in early hominins to 1,400 cc in Homo sapiens, a trajectory driven by interlocking pressures from diet, sociality, and life-history change rather than any single cause.
- Endocast analysis reveals that brain reorganization — particularly expansion of the frontal and parietal lobes, asymmetric lateralization, and changes to the cerebellum — was at least as important as raw size increase, reshaping the brain's internal architecture for language, planning, and tool use.
- The enlarged human brain consumes roughly 20–25% of the body's resting energy despite comprising only 2% of body mass, a metabolic burden that required dietary upgrades through meat eating and cooking and co-evolved with fundamental changes in life history including extended childhood and delayed maturation.
The human brain is the most complex structure known in the natural world, and the story of how it came to exist is one of the central questions of paleoanthropology and evolutionary biology. Over approximately 7 million years, the lineage leading to Homo sapiens underwent a roughly threefold increase in brain volume, from the roughly 350 cubic centimeter (cc) brains of the earliest hominins to the approximately 1,350–1,400 cc brains of anatomically modern humans.1 This expansion was not gradual or uniform. It accelerated at key moments in hominin evolution, involved profound reorganization of internal brain architecture alongside simple size increase, and imposed steep metabolic costs that reshaped diet, life history, and social behavior in return.1, 3 Understanding how and why the human brain evolved requires integrating evidence from the fossil record, comparative neuroanatomy, genetics, and ecology in ways that no single discipline could achieve alone.
Comparative context: primate brains and encephalization
Among vertebrates, mammals tend to have larger brains relative to body size than fish, reptiles, or amphibians, and among mammals, primates are particularly encephalized.1 The standard measure used to compare brain sizes across species of different body sizes is the encephalization quotient (EQ), first systematized by Harry Jerison in the late 1960s. The EQ expresses a species' actual brain mass as a ratio of the brain mass expected for a mammal of that body weight, based on allometric scaling relationships.2 A species with an EQ of 1.0 has exactly the brain size predicted for its body mass; a species with an EQ of 7.5 has a brain seven and a half times larger than the mammalian average for its body weight.2
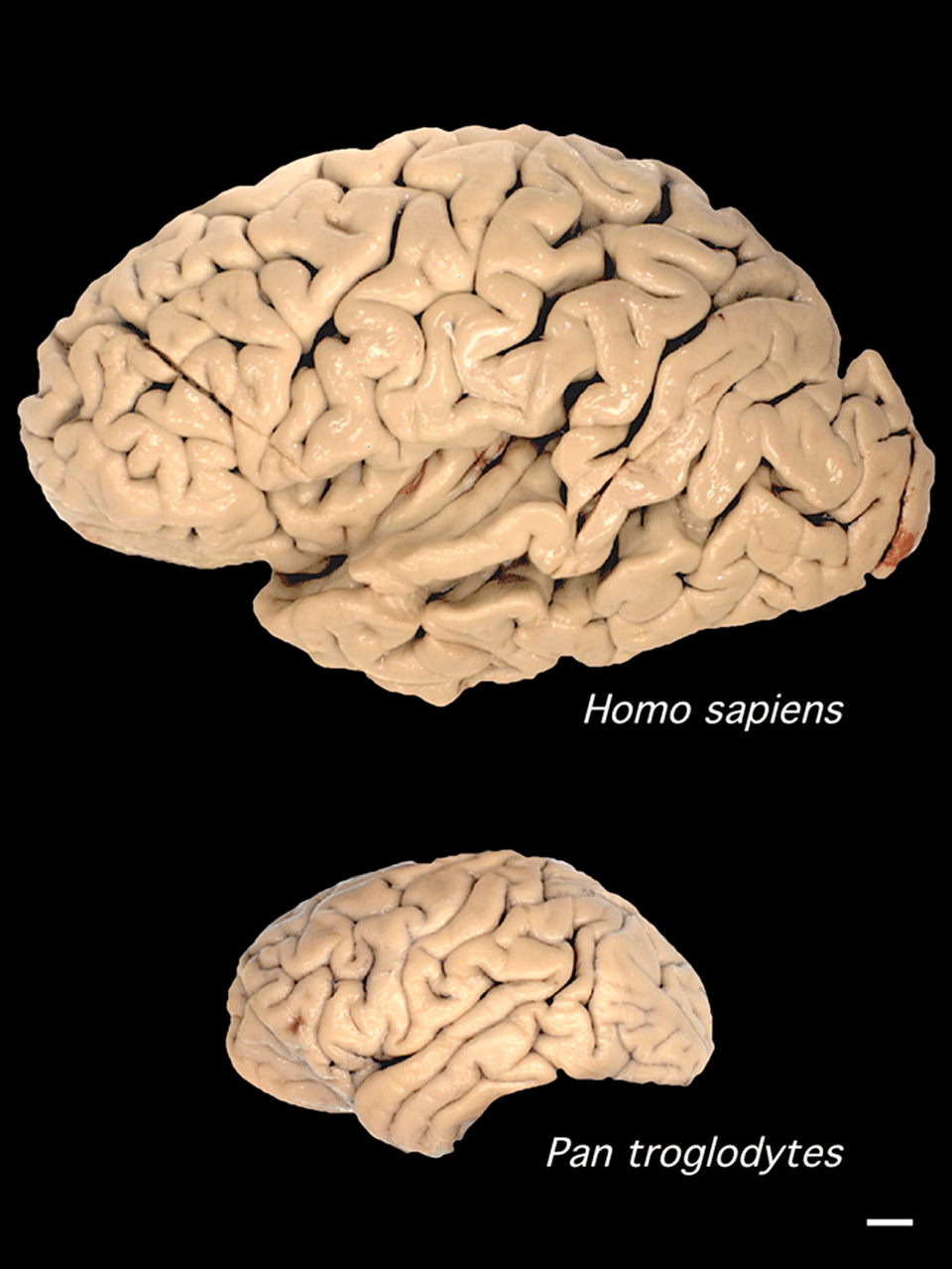
By this measure, modern humans stand apart from every other animal. With an EQ of approximately 7.4 to 7.8, humans possess brains far larger relative to body size than any other primate.1, 2 Chimpanzees, our closest living relatives, have an EQ of roughly 2.5 and a mean brain volume of approximately 400 cc, comparable to the earliest hominins in the fossil record.20 Gorillas and orangutans, despite their much larger bodies, have EQs in the range of 1.5 to 2.0. The dolphin (Tursiops) is the only non-primate with an EQ approaching that of great apes, at roughly 4.0, and even this falls well short of the modern human value.1
The scaling of brain size with body size follows a power law across mammals, with brain mass scaling roughly to body mass raised to the 0.75 power.2 This means that larger animals do have larger absolute brains, but not proportionally larger brains. Primates, and hominins in particular, deviate dramatically from this relationship, suggesting that selection specifically for enhanced neural processing capacity, rather than simply for larger bodies, drove the evolution of our unusually large brains.1, 20 Comparative neuroimaging studies of living primates have confirmed that the human brain is not simply a scaled-up chimpanzee brain; certain regions, particularly the prefrontal cortex, parietal lobes, and cerebellum, have expanded disproportionately relative to overall brain volume, while others, such as the primary visual cortex, are relatively smaller than expected.20
Brain size through hominin evolution
The paleoanthropological record of brain size is reconstructed primarily from endocranial volume measurements, either of the fossilized bony braincase itself or of latex or digital endocasts that capture the internal morphology of the skull. The earliest unambiguous hominins, dating to between 6 and 7 million years ago in central and eastern Africa, had brain volumes indistinguishable from those of chimpanzees. The skull of Sahelanthropus tchadensis, dated to approximately 6–7 million years ago, has an estimated endocranial volume of about 340–360 cc, similar to a small chimpanzee.8 The australopiths, the radiation of bipedal hominins that dominated Africa between roughly 4 and 2 million years ago, showed only modest brain size increases. Australopithecus afarensis, the species that includes the famous "Lucy" skeleton, had a mean cranial capacity of approximately 438 cc across multiple specimens, while Australopithecus africanus averaged around 450–460 cc.8, 22
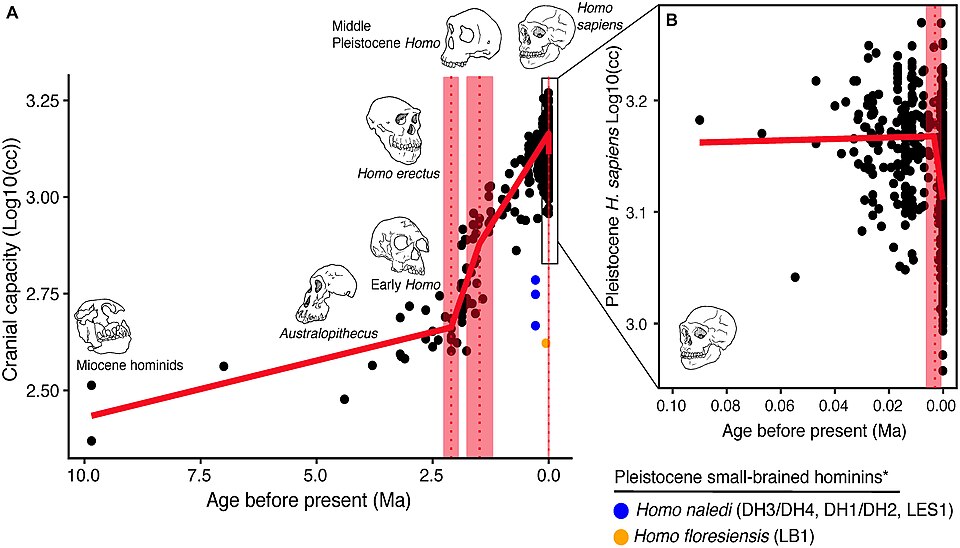
The first substantial increase in brain size is associated with the emergence of the genus Homo around 2 to 2.5 million years ago. Homo habilis, the earliest well-documented member of the genus from East Africa, had cranial capacities ranging from approximately 510 to 680 cc, with a mean around 610–640 cc, representing a clear and statistically significant increase over australopith values.8 A more dramatic acceleration occurred with the appearance of Homo erectus approximately 1.9 million years ago. The earliest H. erectus specimens, including the Dmanisi fossils from Georgia dated to 1.77 million years ago, already had brains in the range of 546–730 cc, and the species' average brain size increased steadily over its nearly two-million-year tenure, with the latest surviving populations at Ngandong in Java achieving cranial capacities up to 1,251 cc.8 By the Middle Pleistocene, archaic members of the lineage leading to modern humans, including Homo heidelbergensis, averaged around 1,200 cc or more.
Neanderthals, the close relatives of modern humans who inhabited Europe and western Asia from roughly 400,000 to 40,000 years ago, had brains that equaled or slightly exceeded those of modern humans in absolute volume, with mean cranial capacities of approximately 1,400–1,600 cc in most large samples.27 Early anatomically modern Homo sapiens similarly averaged close to 1,400 cc, with considerable individual variation. The available fossil evidence therefore documents a roughly fourfold increase in absolute brain volume from the earliest hominins to the modern human range, and a roughly threefold increase in EQ over the same interval.1, 8
Mean endocranial volume across hominin species (cubic centimeters)1, 8, 27
Endocasts and brain reorganization
Measuring cranial volume captures only one dimension of brain evolution. Equally significant are changes in the internal organization of the brain — shifts in the relative size, shape, and connectivity of different regions — that are partially preserved in the morphology of endocranial casts. The study of fossil endocasts has been pursued most systematically by paleoneurologists including Ralph Holloway, Dean Falk, and Emiliano Bruner, whose decades of work have established several key patterns in the reorganization of the hominin brain.6, 10, 11
One of the earliest and most debated shifts is the asymmetric development of Broca's area, a region in the left inferior frontal gyrus of the modern human brain critically involved in speech production and complex sequential processing. Holloway and colleagues have argued from endocast morphology that a derived, human-like pattern of frontal lobe organization, including an imprint consistent with Broca's area, appeared as early as Homo habilis, suggesting that the reorganization of language-relevant circuitry preceded substantial brain size increase.11 This interpretation remains contested, as endocasts preserve only the external surface of the brain, and the correspondence between surface imprints and underlying cytoarchitectural regions is imperfect. Nevertheless, an asymmetric pattern of frontal lobe organization is not present in chimpanzees or gorillas, making its presence or absence in fossil hominins diagnostically significant.6, 11
The parietal lobes, which integrate multimodal sensory information and are critically involved in spatial reasoning, numerical cognition, and tool use, underwent particularly pronounced expansion in the Homo lineage. Emiliano Bruner's geometric morphometric analyses of endocasts have documented a progressive expansion of the superior parietal lobule from australopiths through H. erectus to modern humans, with the most dramatic changes occurring in the lineage leading to H. sapiens and paralleled independently in Neanderthals.21 The parietal expansion is thought to underlie the enhanced capacity for visuospatial processing and mental imagery that characterizes both modern humans and Neanderthals relative to earlier hominins.21
Dean Falk's "radiator hypothesis" proposed a different but complementary perspective on endocast evidence.10, 28 Falk observed that australopiths and early hominins had a pattern of cranial venous drainage (the occipital/marginal sinus system) that is common in other primates but rare in modern humans. As brain size increased, hominins progressively shifted toward a different venous drainage pattern (the transverse/sigmoid sinus system) that Falk argued was more efficient at dissipating heat from a metabolically expensive brain in an upright, bipedal animal exposed to intense solar radiation on the African savanna.28 This "radiator" interpretation has been discussed as one possible anatomical correlate of the metabolic demands imposed by brain expansion.
The cerebellum has received increasing attention as its role in cognition, beyond purely motor coordination, has been recognized. In modern humans, the cerebellum constitutes roughly 10–15% of total brain volume but contains approximately 80% of all neurons in the brain, and it is involved in language, working memory, and procedural learning.20 Comparative neuroimaging studies have shown that human cerebella are significantly larger relative to total brain volume than those of chimpanzees, even after correcting for overall brain size.20 Endocast analyses suggest that this cerebellar expansion was a feature of the modern human lineage rather than a universal hominin trend, potentially linked to the emergence of complex, rapid sequential motor behaviors such as speech articulation.6
A striking case study in the decoupling of brain size and organization is provided by Homo naledi. This small-brained hominin, with cranial capacities of 465–610 cc comparable to small australopiths, survived in southern Africa until as recently as 236,000–335,000 years ago. Holloway and colleagues' 2018 endocast analysis revealed that despite its small volume, the H. naledi brain showed frontal lobe morphology more similar to members of the genus Homo than to australopiths, suggesting that fundamental reorganization of frontally mediated cognitive systems had occurred independently of the dramatic size increases seen in the main line of human evolution.19 This finding reinforced the growing consensus that brain reorganization and brain expansion were at least partially dissociable evolutionary events in the hominin lineage.19
The metabolic cost of a large brain
The human brain is metabolically extravagant. Although it constitutes only about 2% of total body mass, it accounts for approximately 20–25% of the body's resting metabolic rate in adults, and as much as 60–65% of resting metabolism in newborns and infants, when the brain is growing most rapidly.3 Glucose is the brain's primary fuel, and the human brain consumes roughly 120 grams of glucose per day.3 By the standards of mammalian organ energetics, the brain is the most expensive tissue in the body per unit mass, consuming approximately ten times more energy than an equivalent mass of skeletal muscle at rest.3 This metabolic load posed a profound challenge to evolving hominins: a larger brain required either more total food intake, or a reduction in the energetic demands of other metabolically expensive tissues, or both.
The most influential framework for understanding how hominins met this challenge was proposed by Leslie Aiello and Peter Wheeler in 1995 in what they termed the expensive tissue hypothesis.3 Aiello and Wheeler observed that across a sample of primate species, total metabolic rate scales predictably with body mass, leaving little room for individual organ systems to deviate widely from expected values without compensatory adjustments elsewhere. When they compared human organ masses with those of other primates of similar body size, they found that the human brain was dramatically larger than expected, but this excess was precisely offset by a gut substantially smaller than predicted for a mammal of human body size.3 The human small intestine, colon, and stomach are all markedly reduced relative to primate allometric predictions, implying that the energetic savings from a reduced gut helped finance the metabolic costs of a larger brain.3
This trade-off carries important dietary implications. A reduced gut cannot efficiently process large quantities of raw plant material, which requires extensive fermentation and long retention times for maximal caloric extraction. The expensive tissue hypothesis therefore predicts that encephalization in hominins was associated with a shift toward higher-quality, more digestible food sources: meat, marrow, underground storage organs, and ultimately cooked foods.3, 9 Archaeological and isotopic evidence largely supports this prediction. The butchery marks on animal bones at sites such as Dikika in Ethiopia, dated to approximately 3.4 million years ago, indicate that even pre-Homo hominins were accessing animal carcasses, though the significance and regularity of early meat consumption remain debated.9 By the time of Homo erectus, the archaeological record contains abundant evidence of systematic large-game hunting and carcass processing, consistent with a diet substantially richer in animal protein and fat than that of australopiths.9
The cooking hypothesis
Richard Wrangham of Harvard University has argued that cooking represents not merely a useful technological invention but the single most transformative dietary transition in human evolutionary history, one that directly drove the anatomical and neurological differences between Homo erectus and its predecessors.5 Wrangham's cooking hypothesis, elaborated at length in his 2009 book Catching Fire: How Cooking Made Us Human, rests on several lines of evidence and reasoning.5, 16

Cooking dramatically increases the net caloric yield of both plant and animal foods. Experiments by Wrangham and colleagues have demonstrated that cooking starch-rich tubers and roots can increase their digestible energy content by 30–78%, while cooking meat can increase protein digestibility and reduce the energetic cost of digestion.16 These are not trivial margins: for an animal operating close to metabolic limits, access to reliably cooked food could represent the energetic surplus necessary to sustain a dramatically expanded brain.5 The hypothesis predicts that cooking should have been adopted at or near the transition to Homo erectus, roughly 1.9 million years ago, and indeed the anatomical changes associated with that transition, including a smaller gut, smaller teeth and jaws, and an enlarged brain, are all consistent with a shift to softer, more digestible, cooked foods.5
The direct archaeological evidence for controlled fire use at this early date is sparse and contested. While the 1-million-year-old evidence from Wonderwerk Cave in South Africa is relatively secure, and hearths at Gesher Benot Ya'aqov in Israel are well-documented at 790,000 years ago, unambiguous evidence of habitual fire control before 1 million years ago has proven elusive.5, 16 Wrangham has argued that this may reflect preservation bias rather than absence, as open-air fires and ephemeral hearths leave little durable trace. Critics of the cooking hypothesis have pointed to this evidentiary gap as a reason to question the proposed timing of the transition, while acknowledging the plausibility of cooking as an important driver of later brain expansion.5
The social brain hypothesis
An influential alternative and complementary explanation for hominin encephalization emphasizes the cognitive demands of complex social life rather than dietary pressures. The social brain hypothesis, most fully developed by Robin Dunbar of the University of Oxford, proposes that the primary selective pressure driving neocortex enlargement in primates was the need to track, predict, and manipulate the behavior of other individuals in increasingly large and complex social groups.4, 24
Dunbar's foundational observation was a remarkably tight correlation, across 36 primate genera, between the ratio of neocortex volume to total brain volume and the mean social group size characteristic of each species.4 Species that live in large, complex social groups tend to have disproportionately large neocortices relative to the rest of their brains, while more solitary species have smaller neocortices.4 This relationship held controlling for body size, diet, and several other ecological variables, and Dunbar argued it reflected a genuine cognitive constraint: the computational demands of maintaining stable social relationships, tracking alliances and rivalries, engaging in strategic deception, and cooperating in coalitions all require substantial neural resources concentrated in the prefrontal and temporal cortices.4, 24
Extrapolating from the primate neocortex-group-size relationship to the hominin fossil record using reconstructed neocortex volumes, Dunbar proposed that the expected social group size for Homo sapiens with a neocortex ratio appropriate to our brain size would be approximately 150 individuals.24 This figure, now widely known as "Dunbar's number," has been argued to correspond to the typical size of hunter-gatherer bands, village communities, military units, and other natural human groupings across diverse cultures, though the empirical grounding of this specific figure has been subject to considerable debate.24 The broader claim of the social brain hypothesis, that the demands of complex sociality were a primary driver of primate and hominin brain expansion, has received substantial empirical support and remains one of the most productive frameworks in evolutionary cognitive science.4, 24
The social and dietary hypotheses are not mutually exclusive and may have operated as complementary selection pressures at different stages of hominin evolution. Dietary improvements from meat eating and cooking provided the energetic resources for brain expansion, while selection from complex social environments provided much of the adaptive incentive for that expansion. The two forces are also linked through their implications for life history: longer-lived, slower-developing animals with extended juvenile periods have more time to learn complex social rules and acquire dietary skills, and a larger brain may be required both to store the cognitive maps of social relationships and to accumulate the ecological knowledge that makes a high-quality diet possible.26
Life history changes and extended childhood
The evolution of a large, metabolically expensive brain is inseparable from profound changes in hominin life history — the timing and scheduling of growth, development, reproduction, and senescence. Among living mammals, brain size correlates strongly with life history pace: species with large brains relative to body size tend to have longer gestation periods, are born more helpless, grow more slowly, reach sexual maturity later, and live longer than small-brained species of similar body mass.26 Humans exemplify this pattern in the extreme. Compared to other great apes, human infants are born at an earlier stage of brain development (a consequence of the obstetric constraints imposed by our narrow, bipedally adapted pelvis), grow more slowly through an extended childhood and adolescence, and reach reproductive maturity years later.18, 26
Barry Bogin of the University of Michigan identified the distinctively human pattern of childhood as a derived life-history stage not found in other primates.18 In most mammals, weaning marks the beginning of nutritional independence. Human children are typically weaned between two and four years of age but remain nutritionally and socially dependent on adults for many more years, a period during which the brain continues its postnatal growth trajectory and the child acquires language, cultural knowledge, and social competencies.18 This extended juvenile period requires substantial parental and alloparental investment, which in turn favors the cooperative, pair-bonded, and kin-organized social systems characteristic of human societies.18
The birth of a neurologically immature infant with a large brain-to-be represents a distinct evolutionary compromise. The human brain at birth is approximately 30% of its adult volume, compared to about 40% in chimpanzees, and it reaches adult size only around age 5–7, with myelination and prefrontal development continuing into the mid-twenties.23 Paleoanthropological evidence suggests that some aspects of this extended developmental pattern had already appeared in Homo erectus. Analysis of dental development rates in early Homo specimens indicates a prolongation of the juvenile period relative to australopiths, consistent with a brain that required more time to develop to functional maturity.27 Studies of Neanderthal brain growth and development using virtual endocasts of infant and juvenile fossils have shown that Neanderthal brain development followed a trajectory distinct from modern humans, with a more rapid early postnatal expansion of the parietal lobes in humans compared to Neanderthals.27
Genetics of brain evolution
The molecular genetic basis of human brain evolution has become one of the most active frontiers in evolutionary biology since the completion of the human genome sequence in the early 2000s. Comparisons between the human and chimpanzee genomes have identified hundreds of genes showing signatures of accelerated evolution in the human lineage, and a subset of these are expressed specifically in the developing brain.17 Several genes and genomic regions deserve particular attention for what they reveal about the mechanistic underpinnings of brain size increase and cortical reorganization.
ASPM (abnormal spindle-like microcephaly associated) and microcephalin are genes whose loss-of-function mutations in humans cause primary microcephaly, a condition in which the brain forms with dramatically reduced volume, often less than 400 cc, while the overall body is relatively normal in size.12, 13 Both genes are expressed during neural progenitor cell divisions in the developing cortex, and their normal function promotes the production of neurons by extending the proliferative phase of cortical neurogenesis.12 Comparative genomic analyses published in 2002 and 2005 demonstrated that both ASPM and microcephalin show elevated rates of nonsynonymous substitution (amino acid-changing mutations) in the primate lineage leading to humans, with the most rapid evolution occurring in the human and great ape clade.12, 13 In 2005, Patric Evans and colleagues reported that the derived haplotype of microcephalin present in most modern humans showed population-genetic signatures consistent with a recent selective sweep, suggesting that this variant rose rapidly to high frequency because it conferred a fitness advantage, though this interpretation has been debated in subsequent work.13
FOXP2 is a transcription factor gene first implicated in human language through the study of the KE family, in which a heterozygous point mutation causes severe impairments in articulation, grammatical processing, and orofacial motor control.15 In 2002, Wolfgang Enard and colleagues demonstrated that the human version of FOXP2 differs from that of chimpanzees at two amino acid positions, and that both substitutions occurred on the human lineage after the split from the chimpanzee ancestor, with evidence of a recent selective sweep suggesting strong positive selection on the human FOXP2 variant.15 Ancient DNA analysis has subsequently shown that Neanderthals carried the same derived human FOXP2 variants, raising the possibility that some aspects of language capacity may have been shared between H. sapiens and Neanderthals.15 FOXP2 is expressed in the developing cortex, striatum, and cerebellum, and its downstream targets include genes involved in the development of neural circuits for complex motor sequencing, consistent with a role in both speech articulation and other forms of sequential learning.15
Among the most compelling genomic evidence for human-specific brain evolution are the human accelerated regions (HARs), first systematically identified by Katherine Pollard and colleagues in 2006.14 HARs are segments of the genome that are highly conserved across vertebrates, meaning they have changed very little over hundreds of millions of years of evolution, but that show an anomalously high rate of substitution specifically in the human lineage.14 The most striking example, designated HAR1, spans approximately 118 base pairs and contains 18 human-specific substitutions, more than any comparable region of the genome, despite being virtually identical in sequence across other mammals from mice to chimpanzees.14 HAR1 overlaps with a long non-coding RNA gene, HAR1F, that is expressed specifically and exclusively in Cajal-Retzius neurons of the developing human neocortex during weeks 7 to 19 of gestation, a critical period for the organization of cortical layering and folding.14 The function of HAR1F and the other HARs in human brain development remains an active area of investigation, but their pattern of conservation and accelerated human evolution suggests they may encode regulatory elements or non-coding RNAs that fine-tune the expression of cortical development genes in human-specific ways.14, 23
Brain lateralization and language
Modern human brains are notably asymmetric, with the left hemisphere typically dominant for language processing in both right-handers and most left-handers, and with measurable anatomical asymmetries in several cortical regions.25 The planum temporale, a region of the temporal lobe involved in auditory language processing, is on average significantly larger in the left hemisphere than the right in modern humans, an asymmetry that is reduced or absent in chimpanzees and other non-human primates.25 The Broca's area region of the left inferior frontal gyrus similarly shows morphological specializations in modern humans not present in other primates, consistent with its role in speech production and complex syntactic processing.6, 25
The antiquity of cerebral lateralization in hominins has been inferred from endocast morphology, tool handedness, and comparisons with living great apes. While chimpanzees show a slight population-level bias toward right-handedness (about 65% in some studies), this is far weaker than the approximately 90% right-handedness found in human populations across cultures.25 Archaeological evidence for right-handedness in early hominins comes from the preferential direction of cut marks on bones, the orientation of grip marks on tools, and the asymmetric wear patterns on dentition used as a third hand during food preparation. Such evidence suggests a right-hand predominance in stone toolmakers as far back as Homo habilis, consistent with left-hemisphere dominance for the sequential motor planning required by tool manufacture.25 Endocast studies by Holloway and colleagues have identified asymmetric impressions of the sylvian fissure and Broca's region in some australopith and early Homo specimens, though the interpretation of these subtle morphological differences remains a matter of ongoing debate in paleoneurology.11
The co-evolution of lateralization, language, and stone tool manufacture represents one of the most intriguing themes in human brain evolution. The left-hemisphere circuitry for both tool-making and language involves overlapping regions, including the inferior frontal gyrus and the premotor cortex, and the two systems share requirements for hierarchical sequential planning: selecting subgoals, ordering operations, and embedding one action sequence within another.7, 25 Some researchers have proposed that the neural substrate for language may have been co-opted, or "exapted," from circuitry originally selected for complex manual action sequences, a hypothesis that would explain the anatomical overlap and the deep antiquity of lateralization relative to the emergence of fully modern language.7
Reorganization versus enlargement
A central theme in the contemporary study of human brain evolution is the recognition that simple size increase does not adequately characterize what distinguished the hominin brain from those of our ancestors and closest living relatives. The human brain underwent qualitative reorganization, shifts in the proportional size, connectivity, and functional organization of specific regions, that were at least as important as the volumetric expansion documented in the fossil record.20, 23
Comparative neuroimaging of humans and chimpanzees has revealed numerous differences beyond raw size. The human prefrontal cortex, long thought to be disproportionately large, is actually not significantly larger than expected for an ape brain of human size when corrected by modern volumetric methods; however, its white matter connectivity, the density and organization of long-range axonal connections linking prefrontal areas to posterior cortical and subcortical regions, differs substantially from that of chimpanzees.20 The human temporal lobe contains regions such as the angular gyrus and the supramarginal gyrus that are significantly enlarged relative to chimpanzees and that are implicated in language comprehension, reading, and abstract conceptual processing.20 Gene expression studies have shown that thousands of genes are differentially expressed between human and chimpanzee brains, with many human-upregulated genes concentrated in the neocortex and many involved in synaptic plasticity, neurotransmitter metabolism, and the regulation of neuronal activity.17
The developmental mechanisms underlying these reorganizations are beginning to be understood at the molecular level. Key processes include changes in the duration and rate of cortical neurogenesis (how long neural progenitor cells continue dividing), alterations in the timing of neuronal migration and cortical layer formation, and modifications to the pruning and strengthening of synaptic connections during postnatal brain maturation.23 Several of the genes identified as candidates for human-specific brain evolution, including ASPM, microcephalin, and the targets of HAR1, are expressed during exactly these developmental processes, suggesting a coherent molecular narrative in which regulatory changes in development gave rise to the enlarged and reorganized human neocortex.12, 14, 23
The case of Neanderthals provides a particularly instructive perspective on the relationship between brain size and cognitive organization. With absolute brain volumes equal to or exceeding those of modern humans, Neanderthals nonetheless differ from H. sapiens in endocast shape, with relatively smaller cerebellar hemispheres and a different parietal expansion trajectory.27 Whether these shape differences translated into meaningful cognitive differences between Neanderthals and modern humans, and if so which behavioral domains were affected, remains one of the most actively debated questions in paleoanthropology. The archaeological record, which shows sophisticated Neanderthal technology, personal ornamentation, and possible symbolic behavior, does not suggest a simple cognitive inferiority relative to H. sapiens, but whether the behavioral repertoires of the two groups were fully equivalent is unresolved.27
Taken together, the evidence from the fossil record, comparative neuroanatomy, developmental biology, and evolutionary genomics converges on a picture of human brain evolution as a multidimensional and multilevel process. Brain size, brain shape, white matter connectivity, gene expression, synaptic organization, and developmental timing all evolved in concert over millions of years, driven by selection pressures ranging from dietary quality and metabolic capacity to social complexity and the demands of cultural transmission. The human brain as it exists today is the product of this sustained, multivariate evolutionary process, one that operated not on a single trait but on an entire developmental system whose components were shaped, reshaped, and elaborated across the full sweep of hominin history.1, 23
References
The expensive-tissue hypothesis: the brain and the digestive system in human and primate evolution
Endocranial morphology of early Homo from Africa and its implications for cognitive evolution
Evolution of hominid locomotion: new evidence from the early Pleistocene hominid record
Microcephalin, a gene regulating brain size, continues to evolve adaptively in humans
A gene expressed specifically in human neocortex and highly enriched in human brain evolution (HAR1)
Neanderthal brain size at birth provides insights into the evolution of human life history