Overview
- In 1995, Leslie Aiello and Peter Wheeler proposed that the metabolic cost of the enlarged human brain was offset not by an increase in total metabolic rate but by a corresponding reduction in the gastrointestinal tract, another metabolically expensive organ system, yielding a net energy budget close to that predicted for a primate of human body size.
- The hypothesis rests on allometric analyses showing that the human brain is roughly three times larger than expected for a primate of our body mass while the gut is only about 60% of its expected size, and it predicts that encephalization required a dietary shift toward higher-quality, more easily digested foods such as meat, marrow, and cooked tubers.
- Subsequent critiques, particularly Navarrete et al. (2011) highlighting adipose tissue trade-offs and Pontzer et al. (2016) demonstrating elevated total energy expenditure in humans, have challenged the strict organ-for-organ trade-off but have not diminished the broader insight that brain expansion and dietary quality are deeply linked in hominin evolution.
The human brain is metabolically extraordinary. Although it constitutes only about 2% of total body mass, it consumes roughly 20–25% of the body's resting metabolic rate, a share far larger than that of any other primate relative to body size.2 In 1995, the biological anthropologist Leslie Aiello and the physiologist Peter Wheeler proposed one of the most influential explanations for how the hominin lineage could afford such a costly organ. Their expensive tissue hypothesis argued that the energetic demands of an enlarged brain were not met by raising overall metabolic rate but rather by reducing the mass of another metabolically expensive organ system: the gastrointestinal tract.1 This elegantly simple proposition linked brain evolution directly to dietary change and became one of the most cited and debated ideas in paleoanthropology. Three decades of subsequent research have refined, challenged, and extended the hypothesis, but its core insight — that encephalization in hominins cannot be understood apart from the evolution of diet — remains central to the field.
Aiello and Wheeler's 1995 formulation
The expensive tissue hypothesis was published in Current Anthropology in April 1995, accompanied by extensive commentary from other scholars.1 Aiello and Wheeler began from a well-established observation: the human brain is approximately three times larger than would be predicted for a primate of human body mass on the basis of allometric scaling relationships.1, 20 The metabolic cost of maintaining this oversized brain is substantial. Brain tissue consumes energy at a rate of approximately 11.2 watts per kilogram, making it one of the most metabolically expensive tissues in the body, comparable to the heart, kidneys, and liver and roughly 22 times more costly per unit mass than resting skeletal muscle.1, 2
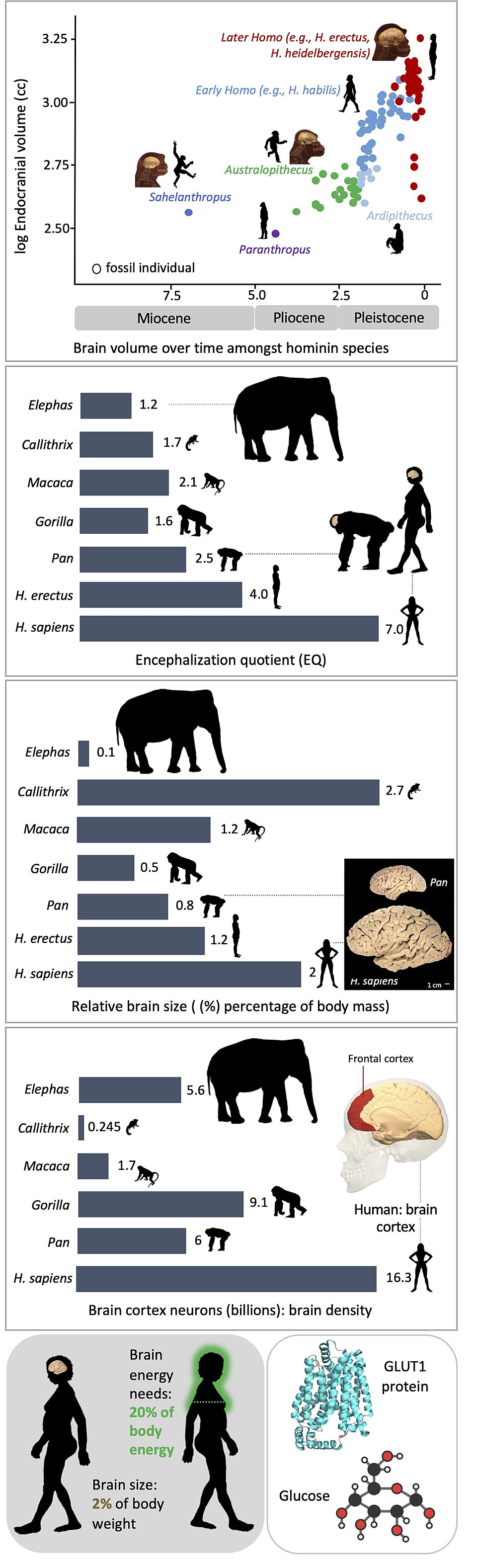
A natural expectation would be that humans, burdened with such an energy-intensive brain, should have a correspondingly elevated basal metabolic rate (BMR) compared with other primates of similar body size. Yet multiple studies had shown that human BMR falls within the range predicted for a primate of human body mass.1, 2 Aiello and Wheeler identified this as a paradox requiring explanation: if the brain demands far more energy than expected but the total energy budget is not elevated, then the excess must be offset by a reduction in the metabolic costs of other organs.
To test this proposition, Aiello and Wheeler identified five major metabolically expensive organs — the brain, heart, kidneys, liver, and gastrointestinal tract — and compared their observed masses in a reference 65-kilogram human male with the masses predicted by allometric equations derived from other primates.1 The results were striking. The human heart and kidneys were approximately the size expected for a primate of human body mass, differing by only trivial amounts from allometric predictions. The liver was slightly smaller than expected, falling roughly 8% below the predicted value. The gastrointestinal tract, however, was dramatically reduced: at approximately 1,100 grams, the human gut was only about 60% of the approximately 1,880 grams predicted for a primate of human body mass, a shortfall of roughly 780 grams.1, 18 Meanwhile, the human brain, at approximately 1,300 grams, exceeded its predicted mass of roughly 450 grams by about 850 grams.1 The critical finding was that the combined mass of all five expensive organs in humans was almost exactly what would be predicted for a primate of human body size: approximately 4,400 grams observed versus approximately 4,450 grams expected. The brain's excess was nearly perfectly compensated by the gut's deficit.1
Human organ masses compared to allometric predictions for a primate of human body size1
The metabolic budget framework
The logic of the expensive tissue hypothesis is rooted in a broader understanding of how organisms allocate metabolic energy. Across mammals, basal metabolic rate scales predictably with body mass according to Kleiber's law, which holds that BMR is proportional to body mass raised to the 0.75 power.2 This relationship imposes a relatively fixed energy envelope within which individual organ systems must compete for metabolic resources. The five metabolically expensive organs identified by Aiello and Wheeler — brain, heart, kidneys, liver, and gastrointestinal tract — collectively account for 60–70% of the body's basal metabolic rate while constituting less than 7% of total body mass.1, 3 The remaining 30–40% of BMR is distributed across skeletal muscle (which comprises roughly 42% of body mass but contributes only about 15% of BMR at rest), adipose tissue, bone, skin, and other structures.1
Within this framework, a dramatic enlargement of any one expensive organ must be accommodated by one of three mechanisms: an increase in total metabolic rate, a reduction in the size of one or more other expensive organs, or some combination of both.4 Aiello and Wheeler argued that the first option — simply raising total BMR — was empirically excluded by the observation that human BMR is not elevated relative to other primates of similar body mass. This left organ trade-off as the primary mechanism, and the data pointed specifically to the gastrointestinal tract as the organ that had been reduced to finance brain expansion.1
William Leonard and Marcia Robertson had independently established complementary findings in 1994. Working with data from primates and other mammals, they demonstrated that the relative proportion of resting metabolic energy allocated to the brain is positively correlated with dietary quality across primate species.2 Species that consume higher-quality diets — those richer in animal protein, lipids, and easily digestible carbohydrates — tend to have relatively larger brains. Humans occupy the extreme positive end of this relationship, combining the highest relative brain size with the highest-quality diet of any living primate.2, 3 Leonard and Robertson's metabolic analysis thus provided an independent empirical basis for the dietary prediction embedded in Aiello and Wheeler's hypothesis.
The gut-brain trade-off
The inverse relationship between relative brain size and relative gut size that Aiello and Wheeler documented in humans was not unique to our species. Across a sample of higher primates, they found a statistically significant negative correlation between relative brain mass and relative gut mass, with species that had larger-than-expected brains tending to have smaller-than-expected guts and vice versa.1 The folivorous howler monkey (Alouatta), for example, has a relatively small brain but a very large, complex digestive system adapted for the prolonged fermentation of fibrous leaves. Fruit-eating spider monkeys (Ateles), by contrast, have relatively large brains and proportionally smaller guts, consistent with a higher-quality diet that requires less digestive processing.1, 7
The mechanistic logic connecting gut size to diet quality is straightforward. A large gastrointestinal tract, particularly an expanded colon and caecum, is necessary for the efficient extraction of nutrients from low-quality plant foods through bacterial fermentation, a slow process that requires extended retention times.1 Reduction of the gut is only viable if an organism shifts to foods that can be digested more quickly and completely in the small intestine, without the need for extensive hind-gut fermentation. Such high-quality foods include ripe fruits, animal tissues (meat, marrow, brain), underground storage organs (tubers, corms), and, ultimately, cooked foods of any kind.1, 8 The expensive tissue hypothesis therefore predicts that hominin encephalization should track dietary shifts toward higher-quality food sources across the archaeological and fossil record.
Jennifer Fish and Charles Lockwood provided a formal phylogenetic test of this prediction in 2003 using independent contrasts methods that controlled for the confounding effects of shared evolutionary ancestry. Their analysis of primates confirmed that evolutionary changes in diet quality were positively correlated with evolutionary changes in relative brain mass, as the expensive tissue hypothesis predicted, in both phylogenetic and non-phylogenetic analyses.7 This result lent strong support to the hypothesis by demonstrating that the brain-gut relationship was not merely a statistical artifact of cross-sectional comparisons but reflected genuine evolutionary co-variation between diet and brain size across the primate phylogeny.
Dietary implications and the archaeological record
If the expensive tissue hypothesis is correct, the major episodes of brain expansion in hominin evolution should be associated with detectable shifts in dietary quality. The archaeological, isotopic, and dental evidence broadly supports this prediction, although the timing and nature of dietary change remain subjects of active investigation.3, 15

The earliest currently known evidence of hominin interaction with animal carcasses comes from the site of Dikika in the Afar region of Ethiopia, where cut marks and percussion marks on mammalian bones have been dated to approximately 3.4 million years ago and attributed to Australopithecus afarensis, the only hominin known from the area at that time.10 These marks indicate that hominins were using stone tools to strip flesh from bones and to access marrow at least 800,000 years before the earliest known deliberately manufactured stone tools of the Oldowan tradition. The regularity and significance of this early meat acquisition remain debated, however, and it is unclear whether it represented a substantial dietary contribution or an opportunistic supplement.10
By approximately 2 million years ago, coinciding with the emergence of the genus Homo and the first substantial increases in brain size beyond the australopith range, the evidence for systematic animal food consumption becomes considerably stronger. At sites in the Koobi Fora formation in East Turkana, Kenya, dated to approximately 1.95 million years ago, David Braun and colleagues documented faunal assemblages with clear butchery marks associated with Oldowan stone tools, including evidence for the processing of diverse terrestrial and aquatic animals such as mammals, turtles, crocodiles, and fish.11 The consumption of aquatic animals is particularly significant because fish and shellfish are rich in long-chain polyunsaturated fatty acids, especially docosahexaenoic acid (DHA), which is a critical structural component of neural tissue and may have been important for supporting brain growth.11
Stable isotope analyses of hominin tooth enamel provide further evidence for dietary divergence. Carbon isotope ratios in the enamel of Paranthropus robustus from South African cave sites show surprising variability, indicating that this robust australopith consumed foods from both C3 (woodland fruits, leaves) and C4 (grasses, sedges, or animals that ate C4 plants) sources, rather than the narrow herbivorous diet that its massive jaws and molars might suggest.12 Early Homo specimens from the same time period show isotopic signatures consistent with a broader and potentially higher-quality dietary niche, although the interpretation of stable isotope data in terms of specific food items is inherently indirect.12
Dental evidence adds another dimension. Peter Ungar's analyses of dental microwear and gross dental morphology in early Homo species have demonstrated that Homo erectus had significantly smaller teeth with thinner enamel and reduced chewing surfaces compared with australopiths, consistent with a shift toward softer, more easily processed foods.15 Katherine Zink and Daniel Lieberman's experimental work further showed that the combination of simple Lower Palaeolithic food-processing techniques (slicing with stone flakes) and the inclusion of meat in the diet would have reduced the number of chewing cycles required per year by approximately 17% and the total chewing force by approximately 26% compared with a raw, plant-based diet, providing a plausible selective mechanism for the gracilization of the masticatory apparatus observed in early Homo.14
Allometric predictions across primates
The quantitative foundation of the expensive tissue hypothesis rests on allometric analyses that predict expected organ masses from body mass across primates. Allometric scaling describes how biological variables change with body size, typically following a power-law relationship of the form Y = aMb, where Y is the variable of interest, M is body mass, a is a scaling coefficient, and b is the scaling exponent.1 For the brain, the expected mass for a primate of human body size (approximately 65 kilograms) was computed by Aiello and Wheeler using reduced-major-axis regression on brain mass and body mass data from higher primates, yielding an expected brain mass of roughly 450 grams — less than half the observed human brain mass of approximately 1,300 grams.1
For the other expensive organs, Aiello and Wheeler relied on least-squares regression equations derived from primate organ dissection data compiled by Walter Stahl and others.1 These allometric predictions indicated that the human heart (approximately 300 grams) and kidneys (approximately 300 grams combined) were close to their expected values, that the liver (approximately 1,400 grams) was modestly smaller than the predicted 1,530 grams, and that the gastrointestinal tract (approximately 1,100 grams) was dramatically below its expected mass of roughly 1,880 grams.1, 18 The net result was that the total mass of expensive tissue in humans — approximately 4,400 grams — was virtually identical to the total predicted for a primate of human body mass, at approximately 4,450 grams. Aiello and Wheeler characterized this as a nearly exact trade-off: what humans gained in brain, they lost in gut.1
In her 1997 review, Aiello noted that this pattern extended beyond a simple cross-sectional comparison. Across primates generally, species with relatively large brains tended to have relatively small guts, and species with relatively large guts tended to have relatively small brains.18 The negative correlation between the two organ systems was statistically robust and held when body mass was controlled for, suggesting that it reflected a genuine energetic constraint rather than a coincidental scaling artifact. However, the sample sizes available from primate dissection studies were modest, and the regression equations carried substantial uncertainty, points that subsequent critics would exploit.5
Critiques and alternatives
The expensive tissue hypothesis attracted critical scrutiny almost immediately upon publication and has been the subject of increasingly rigorous quantitative tests in the decades since. Two challenges in particular have reshaped the debate.
The first major critique came from Ana Navarrete, Carel van Schaik, and Karin Isler, who published a broad comparative analysis in Nature in 2011 using organ mass data from 100 mammalian species, including 23 primates.5 When they controlled for fat-free body mass rather than total body mass, Navarrete and colleagues found no significant negative correlation between brain size and the mass of the digestive tract or any other individual organ across their expanded sample.5 Instead, they found a significant negative correlation between relative brain size and the size of adipose (fat) depots: species with relatively larger brains tended to carry less body fat, and vice versa. Navarrete and colleagues argued that the critical trade-off enabling encephalization was not between the brain and the gut but between the brain and stored body fat, suggesting that encephalization and fat storage represent alternative strategies for buffering organisms against periods of energy scarcity.5 This "fat-brain" trade-off offered a fundamentally different energetic explanation for brain size evolution, though it also raised questions about the human case specifically, since modern humans are notable among primates for their relatively high body fat percentages.
The second major challenge came from Herman Pontzer and colleagues, whose 2016 study in Nature measured total daily energy expenditure (TEE) in humans, chimpanzees, bonobos, gorillas, and orangutans using the doubly labelled water method, the gold standard for measuring energy expenditure in free-living organisms.6 Pontzer and colleagues found that human TEE was substantially elevated relative to all other great apes, exceeding that of chimpanzees and bonobos by approximately 400 kilocalories per day, gorillas by approximately 635 kilocalories per day, and orangutans by approximately 820 kilocalories per day, even after controlling for body size and physical activity level.6 Much of this elevation was attributable to a higher basal metabolic rate in humans, indicating increased organ metabolic activity. This finding directly challenged Aiello and Wheeler's foundational premise that human BMR was not elevated relative to other primates. If humans do in fact expend significantly more total energy than other apes, then the metabolic cost of an enlarged brain could be met in part through an absolute increase in energy intake rather than solely through inter-organ trade-offs.6
Pontzer and colleagues did not argue that the expensive tissue hypothesis was entirely wrong. Rather, they proposed that human brain evolution was enabled by a combination of increased total energy throughput and changes in energy allocation, with both mechanisms operating simultaneously.6 The shift toward higher-quality diets that Aiello and Wheeler identified remained an important part of the story, but it operated alongside a genuine increase in total metabolic rate rather than within a fixed metabolic budget. This reframing moved the debate from a strict organ-for-organ trade-off toward a more complex model of metabolic evolution.
The expensive brain framework
In parallel with these critiques, Karin Isler and Carel van Schaik proposed a broader theoretical framework in 2009 that they termed the "expensive brain" hypothesis.4 This framework subsumed Aiello and Wheeler's gut-brain trade-off as one of several possible mechanisms by which an organism could meet the costs of an enlarged brain. In the expensive brain framework, the metabolic demands of a relatively large brain can be accommodated by any combination of four strategies: increasing total energy intake (and thus total metabolic rate), reducing energy allocation to the digestive system, reducing energy allocation to locomotion, or reducing energy allocation to production (meaning growth and reproduction).4

The expensive brain framework made testable predictions across multiple domains. It predicted a positive correlation between brain size and basal metabolic rate (since species with larger brains should tend to have higher total energy throughput), a negative correlation between brain size and measures of reproductive output (since energy diverted to the brain should reduce the energy available for gestation, lactation, and growth), and a set of life-history changes including slower development and longer lifespans in species with relatively larger brains.4 Empirical tests across a broad range of mammals have supported several of these predictions, particularly the links between encephalization and reduced reproductive rate, extended juvenile periods, and altered life-history schedules.4, 17
Experimental evidence from non-primate species has also shed light on the mechanisms of brain-gut trade-offs. In a landmark study, Alexander Kotrschal and colleagues used artificial selection in guppies (Poecilia reticulata) to breed lines with relatively large and relatively small brains over multiple generations.16 The large-brained guppy lines evolved significantly smaller guts than the small-brained lines, providing direct experimental evidence that brain size and gut size are subject to an evolutionary trade-off, at least in this vertebrate system. The large-brained lines also produced fewer offspring, consistent with the expensive brain framework's prediction that encephalization carries reproductive costs.16 While the relevance of guppy evolution to hominin paleoanthropology is necessarily indirect, this study provided the first experimental demonstration that the organ trade-off at the heart of the expensive tissue hypothesis can emerge through selection on brain size alone.
Integration with cooking and sociality
The expensive tissue hypothesis does not operate in isolation but intersects with two other major frameworks for understanding human brain evolution: the cooking hypothesis and the social brain hypothesis. In each case, the expensive tissue hypothesis provides the metabolic foundation upon which these other explanatory frameworks build.8, 13
Richard Wrangham and colleagues proposed in 1999 that the adoption of cooking was a transformative event in hominin evolution, one that dramatically increased the net caloric yield of foods and thereby made possible the reduction in gut size and the expansion of the brain that characterize Homo erectus and its descendants.8 Cooking gelatinizes starches and denatures proteins, making them more accessible to digestive enzymes and reducing the energetic cost of digestion. Rachel Carmody and Wrangham systematically reviewed the energetic consequences of thermal food processing in 2009, documenting that cooking can increase the usable energy in starchy plant foods by 30–78% and in meat by a more modest but still significant margin.9 If the expensive tissue hypothesis is correct that gut reduction enabled brain expansion, then cooking may have been the dietary innovation that made gut reduction possible by compensating for the loss of digestive capacity with externalized, pre-digestive food processing.
The archaeological evidence for controlled fire use in the Lower Palaeolithic remains contested. The earliest widely accepted evidence comes from Wonderwerk Cave in South Africa, dated to approximately 1.0 million years ago, where Francesco Berna and colleagues documented microscopic traces of burnt bone and plant material within intact Acheulean deposits.19 Wrangham has argued that cooking must have been practiced substantially earlier, at or near the origin of Homo erectus approximately 1.9 million years ago, to account for the anatomical changes visible in the fossil record at that transition, including reduced tooth and jaw size, reduced gut capacity (inferred from rib cage and pelvic morphology), and increased brain size.8 Whether the absence of earlier fire evidence reflects genuine absence or the poor preservation potential of ephemeral campfires in open-air sites remains unresolved.
The social brain hypothesis, articulated most fully by Robin Dunbar, proposes that the primary selective pressure for neocortical expansion in primates was the cognitive demand of managing complex social relationships in large groups.13 This hypothesis is not inherently in tension with the expensive tissue hypothesis; rather, the two frameworks address different aspects of the same problem. The social brain hypothesis explains why selection favoured larger brains (the adaptive benefit of enhanced social cognition), while the expensive tissue hypothesis explains how the metabolic costs of those larger brains were met (through dietary upgrading and gut reduction).1, 13 Leonard, Robertson, and Snodgrass explicitly linked the two frameworks in 2003, arguing that the dietary quality improvements predicted by the expensive tissue hypothesis were themselves facilitated by the cooperative foraging, food sharing, and division of labor that characterize human social organization — behaviors whose evolution the social brain hypothesis helps to explain.3
Modern synthesis
Three decades after its publication, the expensive tissue hypothesis occupies a paradoxical position in the literature on human evolution. Its original formulation — a strict, organ-for-organ metabolic trade-off between brain and gut within a fixed metabolic budget — has been undermined by the findings of Navarrete and colleagues on adipose tissue and, more decisively, by the findings of Pontzer and colleagues on elevated total energy expenditure.5, 6 The human metabolic budget is not fixed relative to other primates; it is elevated, and the brain's costs are met partly through higher total energy throughput and not solely through compensatory gut reduction.
Yet the broader intellectual contribution of the hypothesis has only grown in influence. By identifying the metabolic cost of the brain as a central problem for understanding hominin evolution and by linking brain expansion to dietary quality in a testable, quantitative framework, Aiello and Wheeler set the agenda for an entire generation of research on the energetics of brain evolution.3, 4 The dietary predictions of the hypothesis have been largely supported by archaeological, isotopic, dental, and experimental evidence. The concept that organ systems compete for limited metabolic resources has been confirmed experimentally in at least one vertebrate system.16 And the integration of the expensive tissue hypothesis with the cooking hypothesis and the social brain hypothesis has produced a richer, more complete picture of human brain evolution than any single framework could provide alone.
The current consensus, as articulated by Isler and van Schaik in their expensive brain framework, is that hominin encephalization was enabled by a suite of interacting changes rather than by any single mechanism.4 These changes included a shift toward higher-quality diets rich in animal foods and, later, cooked foods; an absolute increase in total energy intake and expenditure; a reduction in gut size that was both a consequence and a facilitator of dietary change; changes in life history including slower growth, later maturation, and extended juvenile dependence; and the evolution of cooperative breeding and food sharing that allowed mothers to provision nutritionally dependent offspring through an energetically expensive period of brain growth.3, 4, 6 The expensive tissue hypothesis, even in its original strict form, captured a real and important aspect of this complex process. Its enduring legacy is the recognition that the evolution of the human brain was, at its foundation, an energetic problem — one that could not be solved without fundamentally changing what hominins ate and how they obtained their food.
References
The expensive-tissue hypothesis: the brain and the digestive system in human and primate evolution
Evolutionary perspectives on human nutrition: the influence of brain and body size on diet and metabolism
Evidence for stone-tool-assisted consumption of animal tissues before 3.39 million years ago at Dikika, Ethiopia
Early hominin diet included diverse terrestrial and aquatic animals 1.95 Ma in East Turkana, Kenya
Impact of meat and Lower Palaeolithic food processing techniques on chewing in humans
Artificial selection on relative brain size in the guppy reveals costs and benefits of evolving a larger brain
Encephalization, expensive tissues, and energetics: an examination of the relative costs of brain size in strepsirrhines
Microstratigraphic evidence of in situ fire in the Acheulean strata of Wonderwerk Cave, Northern Cape province, South Africa