Overview
- Teeth are the most commonly preserved hominin fossils because tooth enamel—the hardest tissue in the vertebrate body—survives millions of years of burial better than any other biological material, making dental anatomy the single most important data source in paleoanthropology.
- The six-million-year record of hominin teeth documents a sweeping dietary transition: canines shrank and lost their honing function, molars expanded dramatically in the robust australopiths then reduced again in Homo, and enamel thickened as hominins shifted from forest fruits toward harder, more varied foods.
- Modern analytical methods—dental microwear texture analysis, stable carbon isotope ratios, and ancient DNA from dental calculus—extract diet, ecology, and even microbiome data from individual fossil teeth, transforming these small mineralized structures into extraordinarily detailed archives of evolutionary history.
Teeth are the most informative fossils in paleoanthropology. Tooth enamel is the hardest tissue in the vertebrate body—harder than bone, dentin, or any other biological material—and it mineralizes almost entirely during formation, meaning that once laid down it does not remodel. A molar buried in sediment two million years ago can emerge from the matrix in near-perfect condition, its surface textures, its internal growth layers, and even the isotopic chemistry of the diet its owner ate all preserved in microscopic detail. Because hominin skeletal remains are rare, fragmentary, and unevenly distributed across time, the fossil record of human evolution is, to a remarkable degree, a record of teeth.10
The evolutionary story told by hominin teeth spans roughly six million years and crosses an enormous ecological distance: from a forest-dwelling ape with large, projecting canines and thin-enameled molars to a genus capable of surviving on everything from roots and tubers to megafaunal prey. Along the way, the dentition underwent some of the most dramatic and diagnostically useful transformations in the entire hominin fossil record—changes in canine size, molar proportions, enamel thickness, cusp geometry, and developmental timing that researchers have learned to read as reliably as any anatomical text.2, 3
The ancestral ape dental pattern
The last common ancestor of humans and chimpanzees almost certainly possessed a dental formula of 2.1.2.3 on each side of the upper and lower jaws: two incisors, one canine, two premolars, and three molars, the pattern shared by all Old World anthropoids. What distinguished this ancestor from modern humans was not the count but the proportions and functional relationships of those teeth. Living great apes retain a broadly ancestral hominoid pattern: large, sexually dimorphic canines that project well beyond the tooth row, thin enamel on the molars (well under one millimeter in chimpanzees), and a distinctive shearing complex between the upper canine and the first lower premolar.3
This C/P3 honing complex, as it is known, means that in apes the lower third premolar (P3) has a single dominant cusp oriented obliquely so that its front face slides against the back of the upper canine. Each time the jaw closes, the canine is actively resharpened by this occlusal contact. The P3 therefore tends to be sectorial—blade-like and unicuspid—rather than bicuspid as in modern humans. The entire system is an adaptation for producing and maintaining a sharp canine that functions in displays and combat between males, and possibly in food processing as well. When this honing complex was lost in hominin evolution, the lower P3 transformed from a single-cusped shearing tooth into a fully molarized, two-cusped tooth, and this transformation is one of the clearest anatomical markers paleoanthropologists use to identify membership in the hominin clade.3
Canine reduction and the loss of the honing complex
The reduction of the canine is among the earliest and most phylogenetically significant trends in hominin evolution. In Ardipithecus ramidus, dated to approximately 4.4 million years ago, the canines are already substantially reduced compared to chimpanzees, projecting only minimally beyond the tooth row, and the lower P3 has shifted from the purely sectorial ape form toward a more bicuspid shape—though it retains some asymmetry. This combination suggests that the honing complex was already breaking down at the base of the hominin lineage, or even before the split from the chimpanzee lineage in some interpretations.3 In Australopithecus, canine size is still somewhat variable but is clearly reduced below the ape condition in both sexes, and the sexual dimorphism in canine size that characterizes chimpanzees and gorillas is substantially attenuated. By the time of early Homo, and certainly in modern humans, the canines are spatulate and non-projecting, fully integrated into the incisal battery rather than functioning as separate weapons or honing instruments.2
The reasons for canine reduction remain debated. The most influential hypothesis links it to dietary change—as hominins shifted from forest fruits toward ground-level foods requiring more powerful crushing or grinding, the premium on sharp canines may have diminished. Social hypotheses propose that reduced canine dimorphism reflects a shift toward more egalitarian or monogamous social systems in which male-male competition was moderated. A third class of explanation emphasizes the functional integration of the dentition: as molars enlarged and the jaw deepened to accommodate powerful chewing muscles, geometric constraints may have reduced the space available to large canines. These hypotheses are not mutually exclusive, and the canine reduction may have been driven by a confluence of social, dietary, and biomechanical pressures acting over millions of years.3
Molar size changes: megadontia and reduction
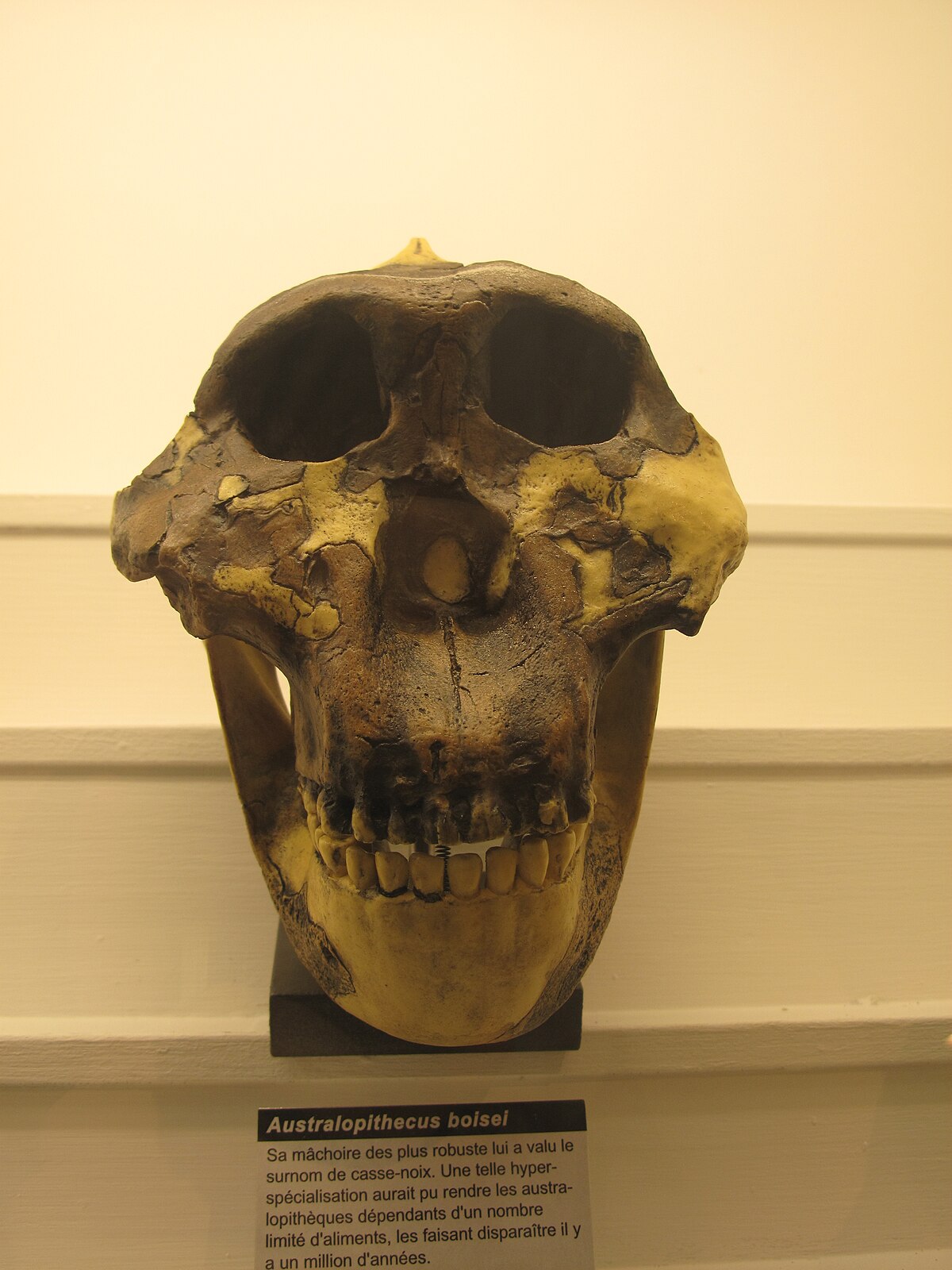
The history of hominin molar size describes an arc rather than a monotonic trend. In early australopiths such as Australopithecus afarensis, molars are already larger relative to body size than in any living ape—a condition referred to as relative megadontia. This trend reached its extreme expression in the robust australopiths of the genus Paranthropus, which lived in Africa between approximately 2.7 and 1.2 million years ago. In Paranthropus boisei, the upper and lower molars are among the largest ever recorded in any hominin, with molar surface areas several times those of modern humans, set in enormous jaws braced by sagittal and nuchal crests for the attachment of massive temporalis muscles. The entire craniofacial architecture of Paranthropus appears organized around the demands of high-force, repetitive chewing.4, 16
Paradoxically, dental microwear texture analysis of P. boisei teeth does not show the heavy pitting expected if this species regularly consumed hard objects such as seeds or nuts. Instead, the microwear signature is relatively smooth and scratch-dominated, more consistent with a diet of tough, fibrous vegetation—possibly including grasses, sedges, and their underground storage organs—than with the hard seed-cracking diet long assumed from the morphology alone.16 Stable carbon isotope data from P. boisei tooth enamel confirm that this species consumed predominantly C4 plants, the photosynthetic pathway characteristic of tropical grasses, while its relatives P. robustus and early Homo show more mixed C3/C4 signatures at the same time period.4, 8 The large molars may therefore reflect fallback feeding on hard or tough foods during lean seasons rather than a year-round hard-object dietary specialization.
In the genus Homo, the trajectory reversed. Homo erectus, appearing around 1.9 million years ago, had substantially smaller molars than contemporary Paranthropus, and molar size continued to decline through the Pleistocene. Modern humans have the smallest molars of any hominin. The reduction correlates in time with the advent of fire and cooking, stone tool use, and other forms of food processing that pre-digest food outside the mouth, reducing the mechanical demands on the dentition. Richard Wrangham’s cooking hypothesis specifically predicts that the dietary shift to cooked, softened food would have relaxed selection for large molars and robust jaws, and the fossil record is broadly consistent with this prediction, though the precise timing of habitual cooking remains debated.14
Enamel thickness and dietary inference
The thickness of the enamel cap on hominin molars varies systematically across the lineage and provides one of the clearest contrasts between hominins and their closest living relatives. Chimpanzee molars have thin enamel, generally less than one millimeter at the cusp, while australopiths and early Homo have thick enamel, often exceeding two millimeters at the apex and grading to still greater thickness on the lateral walls. This pattern was recognized as a hominin synapomorphy—a shared derived character—by the 1980s, though more recent work has nuanced the picture considerably.5
Thick enamel increases the resistance of teeth to fracture under compressive loads, which is consistent with diets that include hard, brittle objects such as seeds, nuts, dried fruits, or underground storage organs with hard outer coats. It also increases the lifespan of the tooth surface by providing more material to be worn away before the softer dentin is exposed. The thin enamel of chimpanzees, by contrast, suits a diet of ripe, soft fruit in which abrasive seeds and hard shells are typically discarded. The transition to thick enamel early in hominin evolution thus marks a broad dietary shift away from the ripe fruit specialization of the forest ape toward harder, more varied, more terrestrial food sources.5 Among the earliest hominins, Australopithecus anamensis already shows thick enamel, and the condition persists through the australopith grade and into early Homo. Homo heidelbergensis and Neanderthals show enamel thickness comparable to or slightly less than that of modern humans, reflecting the progressive softening of the diet through cooking and tool-assisted processing.
Reading diet from fossil teeth
Beyond gross morphology, three analytical methods have transformed fossil teeth into detailed dietary archives: dental microwear texture analysis, stable isotope geochemistry, and ancient biomolecule recovery from dental calculus.
Dental microwear analysis examines the microscopic scratches and pits produced on enamel surfaces by food particles and grit during chewing. Hard, brittle foods such as seeds and nuts produce many deep pits as they fracture against the enamel; tough, fibrous foods such as leaves and stems produce long, parallel scratches as they are sheared. The surface is generated primarily from the last days and weeks of an individual’s diet before death, providing a snapshot of recent feeding behavior rather than a lifetime average. White-light confocal microscopy and scale-sensitive fractal analysis now allow quantitative characterization of microwear surfaces with high reproducibility. Applied to fossil hominins, these methods have revealed that Australopithecus africanus had a microwear texture consistent with hard-object feeding, while A. afarensis shows a more mixed signal, and Paranthropus boisei—as noted above—surprisingly lacks the heavy pitting predicted by its megadont morphology.7, 16
Stable carbon isotope analysis of tooth enamel provides a longer-term dietary signal, integrating years to decades of food intake during the years when the enamel was mineralizing. The carbon isotope ratio (δ13C) of enamel reflects the relative consumption of C3 plants (trees, shrubs, temperate grasses, whose photosynthesis discriminates more strongly against the heavier isotope) versus C4 plants (tropical grasses and sedges, which produce enamel enriched in 13C). A δ13C value near –12‰ in enamel carbonate indicates a diet composed predominantly of C4 foods, while values near –22‰ indicate C3 dominance. Early australopiths from 3–4 million years ago show predominantly C3 signatures, consistent with forest or woodland diets, but by 2.5–2 million years ago several hominin taxa shift toward more positive values, suggesting increasing consumption of grasses, sedges, or animals that had themselves eaten C4 plants.4, 8
Dental calculus—the mineralized plaque that accumulates on tooth surfaces during life—preserves a remarkable biological archive. Ancient DNA and protein analysis of calculus has recovered plant DNA identifying consumed species, starch granules that can be taxonomically identified under microscopy, and microbial DNA revealing the composition of the oral microbiome in deep time. Work on Neanderthal dental calculus has identified plant foods including water lily and yarrow in the diet, and has recovered intact bacterial genomes that are reconstructing the evolutionary history of oral pathogens including the cavity-causing bacterium Streptococcus mutans.11, 12 The shift in oral microbiome composition at the Neolithic—when diets changed dramatically with the adoption of agriculture and subsequently with industrialization—is detectable in calculus from archaeological skeletons, making human oral bacteria an unexpected recorder of dietary and technological transitions across thousands of years.
Dental development and life history
Teeth record not just diet but developmental tempo. Enamel is secreted in a highly regular daily rhythm, producing microscopic incremental lines called cross-striations at intervals of approximately 24 hours. Groups of daily increments are separated by slightly more prominent lines called Retzius lines or striae of Retzius, which represent weekly or near-weekly fluctuations in the secretion rate. Where these Retzius lines reach the outer enamel surface they produce fine ridges called perikymata, which can be counted on the outer surface of unerupted or unworn teeth like rings on a tree. By counting perikymata and using known cross-striation periodicity values, researchers can calculate the total time taken to form the enamel of any given tooth with considerable precision—and therefore reconstruct the schedule of dental eruption and, by extension, the pace of overall growth and life history.9, 10
The landmark study by M. Christopher Dean and colleagues, published in Nature in 2001, used these methods to demonstrate that early Homo taxa including Homo habilis and Homo ergaster had dental development rates intermediate between those of australopiths and modern humans.10 Australopiths appear to have completed dental development on a schedule closer to that of great apes than to modern humans, with first molar eruption around three years of age (compared to roughly six years in modern humans) and a correspondingly accelerated overall growth trajectory. The prolonged period of dental development in modern humans, and the extended childhood it implies, is a derived condition of relatively recent origin within the genus Homo. This extended childhood is thought to be crucial for the acquisition of complex cognitive and social skills, making the evolutionary slowing of dental development one of the key life-history changes underlying modern human cognitive and behavioral capacities.9, 10
Neanderthals, examined by Smith and colleagues using synchrotron imaging to reveal internal enamel microstructure without destructive sectioning, show dental development rates broadly similar to those of modern humans but with some subtle differences, including faster anterior tooth formation and slightly earlier molar eruption in some specimens.1 These differences are consistent with a modestly accelerated life history relative to modern humans, though the magnitude of the difference is considerably smaller than that between modern humans and australopiths.
Third molar reduction and the modern jaw
The third molar, colloquially called the wisdom tooth, is the most variable tooth in the human dentition and the one most frequently absent. Congenital agenesis of one or more third molars—failure to develop the tooth germ at all—affects approximately 20–25% of people in many populations, with rates varying considerably across geographic groups.13 In some East Asian populations the rate exceeds 30%. This variability, combined with the high frequency of impaction (failure to erupt properly) in populations with jaws insufficient to accommodate the tooth, reflects ongoing evolutionary change: third molars are functionally less important in modern humans than they were in our ancestors, and natural selection against the costs of impaction—pain, infection, and abscess—appears to be actively favoring their loss.
The underlying cause is a mismatch between jaw size and tooth size that emerged as hominin jaws shrank over the Pleistocene. Cooking and other forms of food processing reduced the mechanical demands on the posterior dentition, relaxing selection for large jaws while selection for the fetal cranium to pass through the birth canal simultaneously constrained overall head size. The result was a jaw that in many modern humans is simply too small to accommodate three full molars on each side. Archaeological evidence suggests this mismatch is ancient—tooth crowding and impaction are documented in Neolithic populations and become more pronounced with the adoption of agriculture and the shift toward softer, more processed diets—but it has intensified in industrialized populations.14 The trend toward third molar agenesis, now documented in multiple large meta-analyses, is one of the clearest examples of ongoing evolution in the human lineage.
Dental morphology and taxonomic identification
Because teeth preserve so well and vary in diagnosable ways between populations and species, dental morphology has become a powerful tool for taxonomic identification. Several features of tooth shape, cusp arrangement, and root form are sufficiently distinctive to allow researchers to assign isolated teeth to species, track population movements across continents, and even identify individual family relationships in archaeological samples.
Shovel-shaped incisors—upper central incisors with pronounced marginal ridges on the lingual surface that create a scoop or shovel shape—are among the most striking examples of dental population markers. This trait occurs at very high frequencies in East Asian and Indigenous American populations (often exceeding 90% in some groups), at moderate frequencies in some African populations, and at low frequencies in European and South Asian groups.15 The trait maps onto the gene EDAR (ectodysplasin A receptor), a single derived allele of which produces not only shovel-shaped incisors but also increased sweat gland density, thicker hair shafts, and differences in breast morphology, suggesting the selection that swept the allele to high frequency in East Asia acted on one of these pleiotropic effects rather than on tooth shape itself.
At the genus and species level, cusp patterns on the lower molars are particularly diagnostic. The Y-5 or Dryopithecus pattern, in which five cusps are separated by a groove system that forms a Y-shape, is ancestral for hominoids and retained in modern humans and chimpanzees. The +4 pattern, in which four cusps are present with an X-shaped groove, is derived and more common in later Homo populations. Neanderthals are characterized by a suite of distinctive dental features including taurodontism (enlarged pulp chambers with fusion of the roots toward the apex), a distal trigonid crest on the lower molars, and distinctive root morphologies, all of which help researchers identify Neanderthal-derived teeth in mixed archaeological assemblages and detect admixture signals in populations with partial Neanderthal ancestry.6
The integration of morphological, developmental, isotopic, and biomolecular data from hominin teeth has made dental paleoanthropology one of the most productive and technically sophisticated subfields in evolutionary biology. A single worn molar, properly analyzed, can yield the species attribution of its owner, the broad character of the diet consumed during the years it was forming, the pace of development and implied life-history schedule, and the microbiome that colonized the individual’s mouth. As new methods continue to be developed—particularly in ancient protein sequencing, which promises taxonomic identification of fragmentary enamel going back millions of years beyond the range of ancient DNA—the informational yield of the hominin dental record is likely to grow still further.
References
- Smith, T. M. et al. “Dental evidence for ontogenetic differences between modern humans and Neanderthals.” PNAS 107(49): 20923–20928, 2010. doi:10.1073/pnas.1010906107
- Ungar, P. S. & Grine, F. E. “Incisor size and diet revisited: The view from African apes and early hominids.” American Journal of Physical Anthropology 83: 137–147, 1991. doi:10.1002/ajpa.1330830202
- Aiello, L. C. & Dean, M. C. An Introduction to Human Evolutionary Anatomy. Academic Press, 1990. Publisher page
- Cerling, T. E. et al. “Diet of Paranthropus boisei in the early Pleistocene of East Africa.” PNAS 108(23): 9337–9341, 2011. doi:10.1073/pnas.1104627108
- Grine, F. E. & Martin, L. B. “Enamel thickness and dental function in Homo and Australopithecus.” In Evolutionary History of the “Robust” Australopithecines, Aldine de Gruyter, 1988. Publisher page
- Bailey, S. E. & Lynch, J. M. “Mandibular molar root morphology in Neanderthals and Late Pleistocene and recent Homo sapiens.” American Journal of Physical Anthropology 119: 326–338, 2002. doi:10.1002/ajpa.10130
- Ungar, P. S. et al. “Microwear textures of Australopithecus afarensis and Australopithecus africanus incisors.” Journal of Human Evolution 54: 167–180, 2008. doi:10.1016/j.jhevol.2007.09.010
- Lee-Thorp, J. et al. “Isotopic evidence for an early shift to C4 resources by Pliocene hominins in Chad.” PNAS 109(50): 20369–20372, 2012. doi:10.1073/pnas.1204209109
- Dean, M. C. & Smith, B. H. “Dental development and life history in living African and Asian apes.” In Primate Life History and Socioecology, Cambridge University Press, 2009. doi:10.1017/CBO9780511605567
- Dean, M. C. et al. “Growth processes in teeth distinguish modern humans from Homo erectus and earlier hominins.” Nature 414: 628–631, 2001. doi:10.1038/414628a
- Warinner, C. et al. “Calcified microorganisms from the dental calculus of a Neanderthal.” Scientific Reports 4: 6127, 2014. doi:10.1038/srep06127
- Warinner, C. et al. “Metagenomic analysis of dental calculus in ancient and modern humans.” Nature Genetics 46: 1000–1008, 2014. doi:10.1038/ng.3064
- Khalaf, K. et al. “Third molar agenesis: A systematic review and meta-analysis of its prevalence.” Journal of Dental Research 93(7): 696–702, 2014. doi:10.1177/0022034514536386
- Perry, G. H. et al. “Diet and the evolution of human amylase gene copy number variation.” Nature Genetics 39: 1256–1260, 2007. doi:10.1038/ng2123
- Turner, C. G. “Shovel-shaped incisors and the population prehistory of the Pacific Rim.” American Journal of Physical Anthropology 34: 413–422, 1971. doi:10.1002/ajpa.1330340308
- Scott, R. S. et al. “Dental microwear texture analysis of Paranthropus and early Homo.” Nature 436: 693–695, 2005. doi:10.1038/nature03822
References
Mandibular molar root morphology in Neanderthals and Late Pleistocene and recent Homo sapiens
Microwear textures of Australopithecus afarensis and Australopithecus africanus incisors: Evidence for diet and ingestion behaviors
Growth processes in teeth distinguish modern humans from Homo erectus and earlier hominins