Overview
- The multiregional hypothesis, championed by Milford Wolpoff and Alan Thorne, proposed that Homo erectus populations across Africa, Asia, and Europe evolved into modern humans in parallel, linked by sufficient gene flow to prevent speciation; the competing Recent African Origin model held that modern humans arose in Africa around 200–300 thousand years ago and replaced archaic populations elsewhere with little or no interbreeding.
- The genetic revolution of the late 1980s and 1990s — anchored by the 1987 mitochondrial DNA study by Cann, Stoneking, and Wilson that traced all living human mtDNA to a single African ancestral population — provided powerful evidence for African origin and decisively shifted the field toward the replacement model.
- Ancient DNA recovered from Neanderthal and Denisovan remains has added critical nuance: modern humans did interbreed with archaic populations, but at low levels (1–6% of non-African genomes), meaning the truth lies between the two poles — predominantly African replacement with limited archaic admixture, a position sometimes called the assimilation or leaky replacement model.
Few debates in the history of paleoanthropology have been as consequential, or as bitterly contested, as the dispute over where and how anatomically modern humans arose. Through the final two decades of the twentieth century, the field was divided between two mutually incompatible frameworks. The multiregional hypothesis held that Homo erectus populations scattered across Africa, Asia, and Europe evolved into modern humans simultaneously over the past million or more years, bound into a single evolving species by a web of gene flow. The Recent African Origin model — also called the Out of Africa replacement model or simply the replacement hypothesis — maintained that modern humans evolved in Africa during the past few hundred thousand years and subsequently spread across the globe, displacing or replacing the archaic populations they encountered. The resolution of this debate, achieved primarily through genetics and later through ancient DNA, ranks among the most significant advances in the science of human origins. The answer that emerged confounded both camps: modern humans are overwhelmingly of African descent, yet traces of archaic ancestry survive in the genomes of living people, a legacy of limited but real interbreeding at the margins of an essentially African replacement.
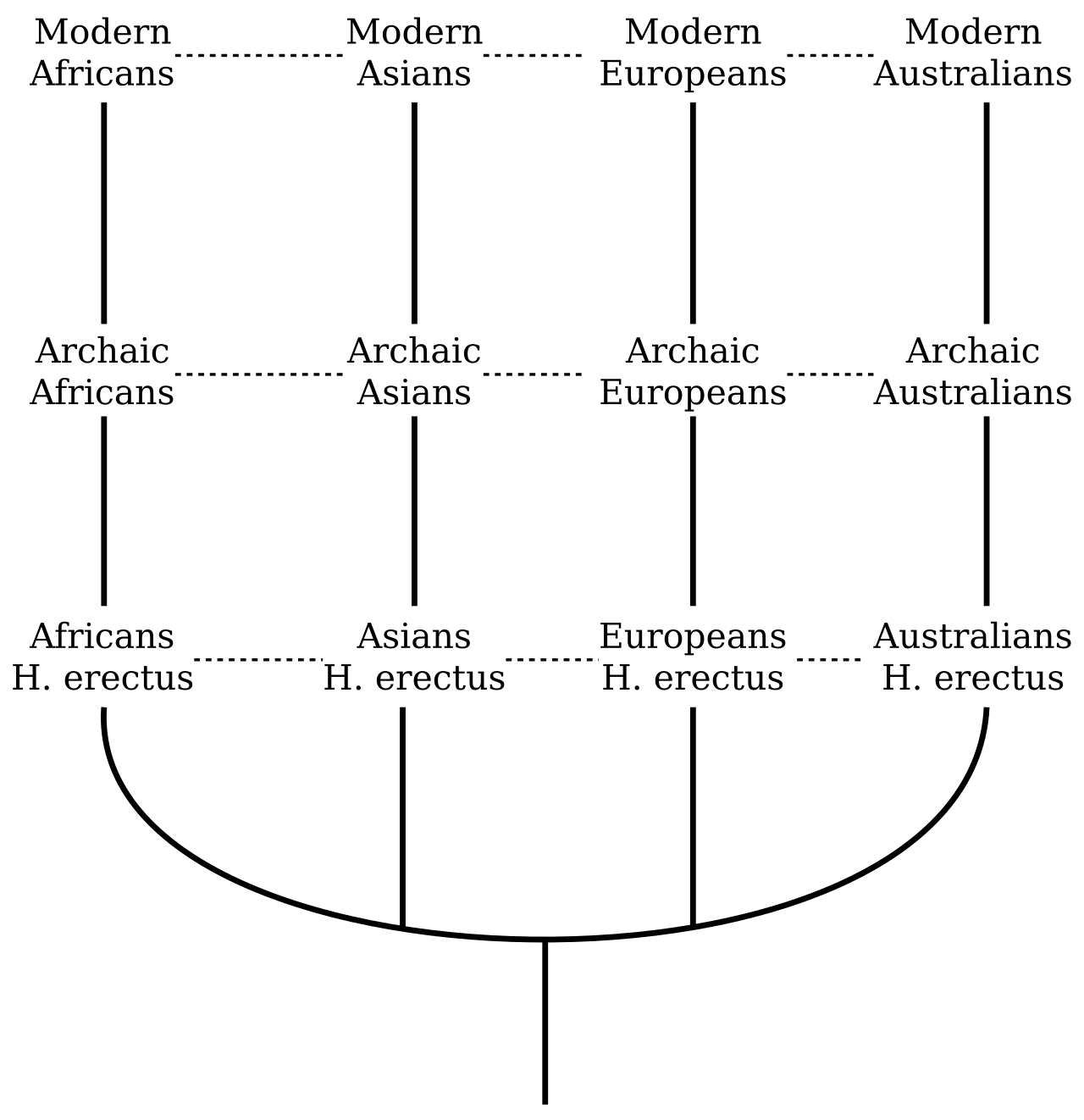
The multiregional hypothesis
The intellectual roots of multiregionalism trace to Franz Weidenreich, the German anatomist who worked on the Peking Man fossils (Homo erectus pekinensis) in the 1930s and 1940s and who argued that regional continuity of anatomical traits linked archaic Asian populations to modern Asians. The hypothesis was given its most rigorous modern formulation by Milford Wolpoff of the University of Michigan, Alan Thorne of the Australian National University, and Wu Xinzhi of the Chinese Academy of Sciences, who articulated it in a series of papers beginning in the late 1980s.3
In the multiregional framework, Homo erectus — or more broadly, the archaic human populations that descended from early Homo dispersals out of Africa roughly 1.8 million years ago — constituted a single, polytypic species throughout the Pleistocene. Regional populations in East Asia, Southeast Asia, Europe, and Africa each evolved certain locally distinctive anatomical features over long periods, yet remained connected by sufficient gene flow to prevent reproductive isolation and speciation. Gene flow, in this model, acts as a cohesive force: it is not so intense as to homogenise populations, but it is regular enough to spread adaptive mutations and maintain the unity of the species. The prediction is therefore one of regional continuity: the same anatomical traits that characterise Homo erectus in a given region should persist as subtle signatures in the modern populations that ultimately emerged there.3, 18
Proponents marshalled substantial fossil evidence. In East Asia, Wolpoff and colleagues pointed to what they considered a continuous thread of features — among them flattened facial skeletons, pronounced cheekbones, and shovel-shaped incisors — linking the Zhoukoudian Homo erectus fossils through archaic Chinese crania such as Dali and Jinniushan to present-day East Asians. In Southeast Asia, Thorne argued that the robust, flat-faced morphology of Indonesian Homo erectus from Ngandong could be traced forward to the earliest modern humans at Australian sites such as Kow Swamp and Lake Mungo.18 In Europe, continuity advocates pointed to features such as pronounced brow ridges and certain mid-facial angles supposedly shared between late Neanderthals and early modern Europeans at sites like Mladec and Cioclovina. The multiregional model was emphatically not a claim of independent parallel evolution of the human species; that would require implausibly convergent evolution of complex shared traits. It was instead a claim that humanity evolved as a single, geographically structured and gene-flow-connected metapopulation over an extended period.3
The Recent African Origin model
The replacement hypothesis, developed most prominently by Chris Stringer of the Natural History Museum in London and Peter Andrews, holds that anatomically modern Homo sapiens evolved from archaic African ancestors — broadly grouped under the label Homo heidelbergensis — sometime between roughly 200,000 and 300,000 years ago on the African continent.6 These early modern Africans subsequently dispersed out of Africa, primarily through the Levantine corridor and possibly across the Bab-el-Mandeb strait, beginning around 60,000–70,000 years ago. As they spread, they replaced the incumbent archaic populations — Neanderthals in Europe and western Asia, late Homo erectus populations in East and Southeast Asia — either through ecological competition, demographic expansion, or both. In the strictest version of the model, this replacement occurred without significant interbreeding, leaving no genetic trace of the archaic populations in the descendants of modern humans.2, 6
The African fossil record provided the strongest empirical support. Fossils from Omo Kibish in Ethiopia, dated to approximately 195,000 years ago, display a combination of modern and archaic features but a cranial vault that is recognisably modern in its high, rounded dome and reduced brow ridges.8 The Herto fossils from the Middle Awash in Ethiopia, dated to around 154,000–160,000 years ago, were described as Homo sapiens idaltu and represent some of the most complete early modern human crania known, combining fully modern facial anatomy with a large, archaic cranial capacity.8 Even more striking, discoveries at Jebel Irhoud in Morocco, published in 2017, pushed the earliest appearance of morphologically modern-like anatomy back to at least 315,000 years ago, suggesting that the emergence of modern human features was a pan-African process rather than a single-origin event in one part of the continent.9
Outside Africa, replacement advocates pointed to the apparent abruptness of the transition from archaic to modern morphology at many sites as evidence against gradual in-situ evolution. In Europe, fully modern humans appear around 40,000 to 45,000 years ago in the archaeological record, after which Neanderthal sites disappear within a few thousand years — a pattern more consistent with replacement by incoming populations than with autochthonous modernisation. The Neanderthal-associated Chatelperronian industry, once cited as evidence of cultural diffusion from moderns, has been reinterpreted by many researchers as evidence of independent development or acculturation under contact pressure rather than smooth indigenous transition.2
Fossil evidence in contention
The fossil record was genuinely ambiguous enough to sustain the debate for decades, and both camps found material to support their interpretations. The crux of the disagreement often came down to how much weight to assign to shared morphological features: were they evidence of regional continuity through genetic inheritance, or were they primitive retentions shared across the genus Homo, or the result of parallel adaptation to similar environments?
The Australian fossil record was one of the most contentious battlegrounds. Thorne and Wolpoff argued that the robust Kow Swamp skeletons, which date to around 9,000–13,000 years ago but display an archaic-looking, sloping forehead and pronounced brow ridges, were best explained by descent from Indonesian Homo erectus rather than from the gracile, modern-looking skull at Lake Mungo.18 Replacement advocates countered that the Kow Swamp variation falls within the range of modern human variation when population-level diversity is properly accounted for, and that the robust features reflect dietary adaptation and cultural head-binding practices rather than archaic ancestry. The issue was further complicated when ancient mtDNA extracted from the Lake Mungo 3 skeleton initially appeared to show a deeply divergent mitochondrial lineage — a result that would have supported multiregionalism — but this finding was subsequently attributed to contamination or DNA degradation and has not been replicated.2
In Europe, early modern humans such as the Peştera cu Oase individual from Romania, dated to approximately 37,000–42,000 years ago, display what appear to be mosaic features combining a modern cranial vault with a notably large posterior mandibular dentition. Trinkaus and colleagues interpreted this as evidence of Neanderthal admixture, consistent with a partial assimilation rather than complete replacement model.10 Ancient DNA subsequently confirmed that the Oase 1 individual carried around 6–9% Neanderthal ancestry — the highest proportion yet detected in any individual — indicating a Neanderthal ancestor within four to six generations.13 This finding vindicated those who had long argued that the European fossil record showed evidence of Neanderthal-modern hybridisation, while also demonstrating that this admixture was limited enough to be consistent with predominantly African replacement.
The genetic revolution
While paleoanthropologists debated the fossils, geneticists were approaching the question from a fundamentally different angle. The decisive early contribution came in 1987, when Rebecca Cann, Mark Stoneking, and Allan Wilson at the University of California, Berkeley published a study of mitochondrial DNA (mtDNA) sequences sampled from 147 people representing five geographically diverse populations.1 Because mtDNA is maternally inherited without recombination, its phylogenetic tree accurately reflects the history of maternal lineages. Cann and colleagues found that all 147 sequences coalesced into a single ancestral sequence — the so-called Mitochondrial Eve — in a woman who lived in Africa approximately 200,000 years ago. The two deepest-branching mtDNA lineages were both African, and African populations harboured significantly more mtDNA diversity than populations on any other continent, a pattern consistent with Africa being the oldest and most genetically diverse human population.1
The Mitochondrial Eve study was immediately seized upon as vindication of the replacement model and attracted enormous public attention. It was also immediately attacked. Critics noted that the study’s phylogenetic analysis had methodological weaknesses — notably that the tree-building algorithm could produce multiple equally parsimonious trees, some of which rooted outside Africa. These objections were eventually answered through improved computational methods and larger sample sizes, all of which continued to support an African root and a coalescence time in the range of 150,000–200,000 years ago. Crucially, the mtDNA data were incompatible with the multiregional model in one specific and testable sense: if modern humans had been continuously evolving as a single species across the Old World for a million or more years, as multiregionalists claimed, then the mtDNA tree should have much older, non-African roots and far greater depth than what was observed.2
Parallel evidence came from the non-recombining portion of the Y chromosome, which tracks paternal lineages in the same way that mtDNA tracks maternal ones. Studies of Y-chromosome diversity consistently found that all living male lineages coalesce to a common ancestor — Y-chromosome Adam — who also lived in Africa, with estimates ranging from approximately 200,000 to 340,000 years ago.7 The concordance of independently inherited uniparental markers in pointing to a recent African common ancestor was powerful evidence. By the late 1990s and early 2000s, genome-wide studies of autosomal diversity told the same story: genetic diversity decreases with distance from sub-Saharan Africa in a pattern consistent with a serial founder effect as populations expanded outward from an African source population, not with the deep, geographically distributed polymorphism expected under the multiregional model.7
Ancient DNA and the resolution
For all the power of the genetic data from living populations, one question remained open: did any genetic contribution from archaic populations outside Africa survive in modern genomes? The coalescence-based studies of mtDNA and the Y chromosome could only detect lineages that had persisted to the present; even substantial admixture could theoretically go undetected if the archaic lineages happened to go extinct by genetic drift in the descendants. The answer required direct sequencing of archaic genomes, which became possible only with the development of ancient DNA extraction and sequencing techniques capable of working with highly degraded, contamination-prone fossil material.
The landmark publication came in 2010, when Svante Pääbo and colleagues at the Max Planck Institute for Evolutionary Anthropology published a draft sequence of the Neanderthal genome assembled from three bones recovered from Vindija Cave in Croatia.4 Comparison of the Neanderthal genome with those of five present-day humans from different continents revealed that non-African individuals — from Europe, East Asia, and Papua New Guinea — shared significantly more derived alleles with Neanderthals than did West African individuals. This asymmetry, quantified with the D-statistic, was most parsimoniously explained by gene flow from Neanderthals into the ancestors of non-Africans after the Out of Africa dispersal but before the divergence of European and East Asian populations. The estimated Neanderthal contribution to non-African genomes was approximately 1 to 4 percent.4
Later in the same year, a second major discovery emerged from a finger bone and molar found in Denisova Cave in the Altai Mountains of Siberia. DNA extracted from these fragments revealed a previously unknown archaic human lineage — the Denisovans — that was a sister group to Neanderthals and had contributed genetic material to certain modern populations.5 Present-day Melanesians and Aboriginal Australians carry approximately 3 to 6 percent Denisovan-derived DNA, and populations across Island Southeast Asia show varying proportions of Denisovan ancestry.16, 17 A high-coverage Vindija Neanderthal genome published in 2017 refined the estimate of Neanderthal admixture in Eurasians to approximately 1.8 to 2.6 percent and confirmed that the admixture occurred in a relatively narrow window around 47,000–65,000 years ago, consistent with initial modern human dispersal into the Near East and subsequent expansion into Europe and Asia.13, 19
Crucially, gene flow was not exclusively from archaic populations into moderns. Analysis of the high-coverage Altai Neanderthal genome revealed that this individual carried fragments of modern human DNA, pointing to an earlier episode of gene flow from an early modern African population — possibly associated with the brief modern human expansion into the Levant around 100,000 years ago that is attested by fossils at Skhul and Qafzeh — into the Neanderthal gene pool of Western Asia.15 The picture that emerged was one of sporadic, geographically and temporally limited contact events rather than continuous, range-wide gene flow of the kind posited by multiregionalists.
The assimilation model
The ancient DNA findings placed both extreme positions in an untenable position. The strict replacement model, which held that modern humans replaced archaics with zero interbreeding, was falsified: Neanderthal and Denisovan DNA demonstrably persists in living humans, a legacy of real reproductive contact. Yet the low proportions of archaic ancestry — 1 to 4 percent from Neanderthals, up to 6 percent from Denisovans, totalling far less than 10 percent of any living human genome — were equally incompatible with the multiregional model, which would predict far deeper and more evenly distributed archaic ancestry if Eurasian populations had been continuously contributing to the modern human lineage for a million years. All living humans derive the overwhelming majority of their ancestry from the African source population that expanded out of Africa in the Late Pleistocene.14
The framework that best accounts for all available evidence is the assimilation model, sometimes called the leaky replacement or partial replacement model, which had been advocated by Fred Smith and colleagues as early as 1989 as a middle ground between the two poles.11 In this framework, an essentially African origin of modern humans is maintained — consistent with the genetic data — while allowing for episodic, geographically circumscribed interbreeding with archaic populations at contact zones. The archaic populations were genuine Neanderthal and Denisovan groups, not simply regional variants of a continuously evolving H. sapiens metapopulation; they had diverged from the modern human lineage on the order of 500,000 to 700,000 years ago and had accumulated substantial genetic and anatomical differences.4, 5 Their genetic contribution to living humans is real but modest, and some of the introgressed alleles have been adaptively retained — most famously, the Denisovan-derived EPAS1 allele that facilitates high-altitude adaptation in Tibetans — while others have been purged by purifying selection in regions now recognised as introgression deserts.14
Where the field stands
By the early 2020s, the broad outlines of the debate had been resolved to a degree that would have seemed impossible during the height of the controversy. Modern humans are primarily of African origin, with the bulk of their ancestry tracing to a population that diversified within Africa over several hundred thousand years before dispersing globally. Recent work has refined this picture further, revealing that the within-Africa diversification was itself complex: rather than a single founding population in one region of Africa, genetic evidence points to a structured African metapopulation with multiple lineages contributing to the ancestry of all living humans, with some deep divergences within Africa dating back 200,000 to 300,000 years.9
The specific claims of multiregionalism — that there was meaningful genetic continuity between, for instance, Chinese Homo erectus and modern East Asians, or between Indonesian Homo erectus and Aboriginal Australians — have not been substantiated by the genetic evidence. Genome-wide studies, ancient DNA from early modern humans in Europe and Asia, and analyses of population structure all point to recent common African ancestry for all non-African populations, with no detectable deep regional roots in Eurasian archaics beyond the low-level Neanderthal and Denisovan admixture documented by ancient DNA.12 The putative regional continuity features cited by multiregionalists are now generally interpreted as shared primitive retentions, as convergent adaptations, or as falling within the normal range of modern human variation.
At the same time, the strict replacement model has been softened irreversibly. Modern humans were not a tightly bounded species incapable of producing fertile offspring with their closest archaic relatives. The Neanderthal and Denisovan admixture events were real, their genetic legacies persist in every non-African alive today, and some of those archaic alleles have been positively selected because they conferred adaptive benefits in new environments. The field now understands modern human origins not as a simple bifurcation between replacement and continuity, but as a predominantly African origin punctuated by episodes of archaic introgression — a story that is both more African and more interconnected than either of the original competing models had imagined.
Open questions remain. The timing, location, and number of admixture events between modern humans and Neanderthals are still being refined, and the geographic range of the Denisovans — and which modern populations carry their DNA — continues to expand as ancient and modern genomes from South and Southeast Asia are analysed.17 The identity of the African archaic populations that contributed to deep African population structure, sometimes called “ghost” lineages, has yet to be resolved by fossil evidence. And the question of what drove the ecological and demographic success of modern humans relative to archaics — what gave the expanding African population its competitive advantage — remains one of the central unsolved problems of paleoanthropology. The debate between Wolpoff and Stringer may be settled in its broad outlines, but the finer grain of how modern humans came to be who they are continues to be written.
References
Assimilation or replacement? Evidence from the late Neanderthal and early modern human record of the Levant