Overview
- Improved radiocarbon dating has established that Neanderthals disappeared from Europe between approximately 41,000 and 39,000 years ago, after a period of coexistence with Homo sapiens lasting roughly 2,600 to 5,400 years.
- The leading hypotheses for Neanderthal extinction include competitive exclusion by modern humans who held demographic and possibly technological advantages, climate instability during Heinrich stadials, disease transmission from incoming Homo sapiens populations, and gradual genetic absorption through interbreeding.
- An emerging consensus holds that no single cause adequately explains the disappearance of Neanderthals; rather, the extinction resulted from the convergence of small population sizes, inbreeding depression, climatic stress, and competitive pressure from a numerically superior modern human population.
The disappearance of the Neanderthals (Homo neanderthalensis) approximately 40,000 years ago is one of the defining events in human evolutionary history. After occupying Europe and western Asia for more than 300,000 years, Neanderthals vanished within a few millennia of the arrival of anatomically modern humans (Homo sapiens) on the continent. The causes and circumstances of this extinction have been debated for more than a century, with hypotheses ranging from violent replacement by modern humans to gradual absorption through interbreeding. Advances in radiocarbon dating, ancient DNA analysis, paleoclimate reconstruction, and computational modelling have transformed understanding of this transition, revealing it to be a complex, multicausal process involving demographic, ecological, climatic, and genetic factors.1, 7, 21
The question of Neanderthal extinction is inseparable from the question of what made Homo sapiens successful. Neanderthals were not the cognitively impoverished brutes of popular imagination; they buried their dead, produced sophisticated stone tools, used pigments and ornaments, and exploited diverse environments from Mediterranean coasts to subarctic steppe.16, 19 That such a capable and long-lived species disappeared so soon after encountering modern humans demands explanation, and the answer that has emerged from decades of interdisciplinary research is not a single dramatic cause but rather the convergence of multiple vulnerabilities exploited by a subtle but decisive competitive imbalance.21
The revised chronology of disappearance
Establishing exactly when Neanderthals disappeared has proven to be one of the most challenging problems in Palaeolithic archaeology. For decades, radiocarbon dates from late Mousterian (Neanderthal-associated) sites in southern Iberia appeared to extend Neanderthal survival to as recently as 28,000 or even 24,000 years before present, suggesting that Neanderthal and modern human populations coexisted for more than 10,000 years across different regions of Europe.17 However, many of these late dates have been called into question by improvements in radiocarbon methodology.

The critical methodological advance was the application of ultrafiltration pretreatment protocols to bone collagen samples. Conventional radiocarbon dating of very old samples is highly susceptible to contamination by younger carbon, which can produce dates that are substantially too young. Even trace amounts of modern carbon can shift a date by thousands of years when the true age approaches the limits of the radiocarbon method (approximately 50,000 years).1 In 2014, Higham and colleagues applied improved accelerator mass spectrometry techniques and Bayesian age modelling to 196 samples from 40 key Mousterian and Neanderthal archaeological sites across Europe, from Russia to Spain. Their results demonstrated that Neanderthals disappeared from most of Europe between approximately 41,030 and 39,260 calibrated years before present (cal BP), with Mousterian technology disappearing within the same time window.1
This revised chronology had two major implications. First, it eliminated most of the anomalously late dates that had been used to argue for prolonged Neanderthal survival in southern refugia. Sites such as Gorham's Cave in Gibraltar, which had yielded dates as young as 24,000 years BP, were found to contain contaminated samples, and redating placed the final Neanderthal occupation significantly earlier.1, 18 Radiocarbon redating of purported late Neanderthal sites in southern Iberia confirmed that the Middle to Upper Palaeolithic transition occurred earlier than previously believed, casting doubt on the idea of a persistent Neanderthal refugium south of the Ebro River.18 Second, the revised dates established that the period of overlap between Neanderthals and modern humans in Europe was approximately 2,600 to 5,400 years — long enough for sustained interaction but far shorter than earlier estimates had suggested.1
Subsequent work has further refined the chronology. A 2021 study reexamined Neanderthal remains from Belgium, including the historically important specimens from Spy Cave and Engis, using single amino acid radiocarbon dating. The results confirmed that these Neanderthals disappeared from northwest Europe between approximately 44,200 and 40,600 cal BP, consistent with and slightly earlier than the broader European pattern established by Higham and colleagues.24
The overlap period with Homo sapiens
The arrival of anatomically modern humans in Europe is documented by the appearance of the Aurignacian technocomplex, the earliest unambiguously modern human cultural tradition on the continent, beginning around 43,000 to 42,000 cal BP in parts of central and western Europe.1, 17 The Bayesian modelling of Higham and colleagues demonstrated that the overlap between the latest Mousterian dates and the earliest Aurignacian dates at a continental scale was between 2,600 and 5,400 years, depending on the region.1 This period of coexistence was not uniform across the continent; the replacement appears to have been a mosaic process, with Neanderthals persisting in some areas while modern humans had already established themselves in others.
The nature of interaction during this overlap period remains debated. Genetic evidence confirms that interbreeding occurred, as all non-African modern humans carry approximately 1.5 to 2.1 percent Neanderthal-derived DNA, with some East Asian populations carrying slightly more than European populations.2, 4 However, the bulk of Neanderthal gene flow into modern human populations appears to have occurred during an earlier phase of contact, likely in the Near East around 50,000 to 60,000 years ago, rather than during the final period of coexistence in Europe. Genomic analysis of five late Neanderthals who lived between 39,000 and 47,000 years ago showed no evidence of recent gene flow from modern humans into late Neanderthal populations, suggesting that interaction during the final overlap was limited or that any gene flow was predominantly unidirectional.15
Archaeological evidence for direct contact is similarly ambiguous. The Châtelperronian, a transitional industry found in France and northern Spain dating to roughly 43,000 to 39,000 cal BP, contains elements of both Middle and Upper Palaeolithic traditions, including blade technology and personal ornaments such as perforated animal teeth. The Châtelperronian has been attributed to Neanderthals based on skeletal associations at Saint-Césaire and the Grotte du Renne at Arcy-sur-Cure.20 Whether this industry reflects independent Neanderthal innovation or acculturation through contact with modern humans remains one of the most contentious questions in Palaeolithic archaeology. Proponents of acculturation argue that the Upper Palaeolithic elements appeared too suddenly to represent indigenous development, while proponents of independent innovation point to evidence that Neanderthals were producing symbolic objects long before modern humans arrived in Europe.19, 20
Competitive exclusion by modern humans
The competitive exclusion hypothesis holds that modern humans outcompeted Neanderthals for the same ecological resources, gradually displacing them from their habitats. This hypothesis draws on ecological niche modelling, archaeological site density data, and computational simulations to argue that modern humans held a decisive advantage in population size, dietary breadth, or cultural adaptability.5, 6
One of the strongest lines of evidence for competitive exclusion comes from the demographic disparity between the two populations. In 2011, Mellars and French compiled a comprehensive database of Neanderthal and modern human sites in a 75,000-square-kilometre region of southwestern France encompassing the Dordogne, one of the best-documented areas for both populations. Their analysis revealed a roughly tenfold increase in the density of occupied sites and in the quantity of stone tools and faunal remains during the transition from the Mousterian to the Aurignacian, implying that modern humans arrived in or achieved population densities approximately nine to ten times greater than those of the Neanderthals they replaced.8 This numerical superiority alone would have constituted a powerful competitive advantage, even in the absence of any cognitive or technological differences between the two species.
Ecological niche modelling has provided further support. Banks and colleagues used high-resolution paleoclimatic simulations combined with archaeological site distributions to reconstruct the eco-cultural niches occupied by Neanderthals and modern humans during Marine Isotope Stage 3 (roughly 60,000 to 27,000 years ago). Their results showed that the two populations exploited broadly similar niches, and that the southerly contraction of Neanderthal range during Greenland Interstadial 8 (approximately 38,000 years ago) coincided not with a climatic shift but with the geographic expansion of modern humans into the same environments.5 This pattern is consistent with the ecological principle of competitive exclusion, whereby two species competing for identical resources cannot coexist indefinitely; the more efficient competitor will eventually displace the other.
Timmermann's 2020 supercomputer simulations, which combined realistic climate reconstructions with population dynamics models, arrived at a complementary conclusion. The simulations found that rapid Dansgaard-Oeschger climate oscillations alone could not account for Neanderthal extinction across their entire range, and that interbreeding alone would have been insufficient to absorb the Neanderthal population. Only models incorporating a significant competitive advantage for Homo sapiens in resource exploitation produced extinction timelines matching the archaeological record.7
The nature of this competitive advantage remains debated. Possible factors include the broader dietary range of modern humans, who exploited aquatic resources, small game, and plant foods more intensively than Neanderthals; superior long-range weaponry, such as spear-throwers and eventually bows; larger and more interconnected social networks facilitating information exchange and buffering against local resource shortfalls; and higher reproductive rates possibly linked to shorter interbirth intervals.8, 10 Stable isotope analysis has shown that Neanderthals were heavily dependent on large terrestrial herbivores for protein, whereas early modern humans in Europe exhibited a broader isotopic range indicating more diverse diets including freshwater fish and small game.10
Climate instability and environmental stress
The period of Neanderthal extinction coincided with some of the most dramatic and rapid climatic oscillations of the Late Pleistocene. The North Atlantic climate during Marine Isotope Stage 3 was punctuated by Dansgaard-Oeschger events — rapid warmings of 8 to 15 degrees Celsius occurring within decades, followed by gradual cooling — and by Heinrich events, episodes of massive iceberg discharge into the North Atlantic that triggered severe cold stadials lasting centuries to millennia.23
Heinrich Stadial 4 (HS4), which occurred around 40,200 to 38,300 cal BP, has attracted particular attention because its timing closely coincides with the final disappearance of Neanderthals from most of Europe. HS4 was associated with a near-shutdown of the Atlantic Meridional Overturning Circulation, producing extreme cold and aridity across western and central Europe. Pollen records indicate that forests retreated and were replaced by open steppe and semi-desert vegetation across much of the Iberian Peninsula and Mediterranean region.23 Staubwasser and colleagues demonstrated that two cold stadials associated with Heinrich events (HS4 and an earlier stadial around 44,000 cal BP) coincided with marked declines in Neanderthal site frequency, suggesting that these climatic shocks fragmented Neanderthal populations and contracted their range into isolated refugia.23
However, climate change alone is insufficient to explain Neanderthal extinction. Neanderthals had survived numerous glacial-interglacial cycles and Heinrich events over the preceding 300,000 years without going extinct. The critical difference during the final episodes was the simultaneous presence of modern human populations that could rapidly recolonize territories vacated by Neanderthals during climatic downturns.9 Stewart and Stringer argued that the dynamics of refugia were central to this process: during cold periods, both populations would have contracted into southern refugia, intensifying competition; during subsequent warmings, modern humans, with their larger population sizes and broader adaptive toolkit, would have expanded more rapidly, occupying territories before Neanderthal populations could recover.9 In this view, climate change did not directly cause extinction but rather amplified the competitive disadvantage that Neanderthals already faced.
Disease transmission
The disease hypothesis proposes that modern humans arriving from Africa carried pathogens to which Neanderthal populations had no immunity, analogous to the devastating epidemics that followed European contact with the Americas in the fifteenth and sixteenth centuries. Neanderthals and modern humans had been genetically separated for approximately 500,000 to 700,000 years, during which time each lineage would have evolved distinct immune repertoires adapted to their respective pathogen environments.11
In 2019, Greenbaum and colleagues developed a mathematical model integrating disease transmission dynamics with gene flow between Neanderthals and modern humans. Their model showed that the exchange of novel pathogens at the contact zone could have created an invisible epidemiological barrier that initially prevented either population from expanding into the other's territory, explaining the long-lasting geographic boundary between the two populations in the Levant over tens of thousands of years. Critically, the model predicted that interbreeding would eventually transfer immune-related genes between the populations, gradually eroding this disease barrier and allowing modern humans — with their larger reservoir population in Africa continually supplying migrants — to expand into Neanderthal territory.11
Genomic evidence provides indirect support for this hypothesis. Analysis of Neanderthal-derived DNA segments in modern humans has revealed an enrichment of Neanderthal alleles at immune-related loci, including genes of the human leukocyte antigen (HLA) system, which plays a central role in pathogen recognition. This pattern suggests that Neanderthal immune gene variants were positively selected in the hybrid population because they provided protection against pathogens encountered in Eurasian environments.4 The reciprocal implication is that Neanderthals may have been vulnerable to tropical and subtropical pathogens carried by modern humans migrating from Africa, particularly given their small population sizes and limited genetic diversity, which would have constrained the range of their immune responses.
Assimilation through interbreeding
The assimilation hypothesis offers an alternative framing of Neanderthal disappearance: not extinction in the classical sense but rather absorption into the expanding modern human gene pool through interbreeding. Under this model, Neanderthals did not vanish biologically so much as they were demographically swamped by a much larger incoming population, with their genes persisting at low frequency in the hybrid descendants.2, 14
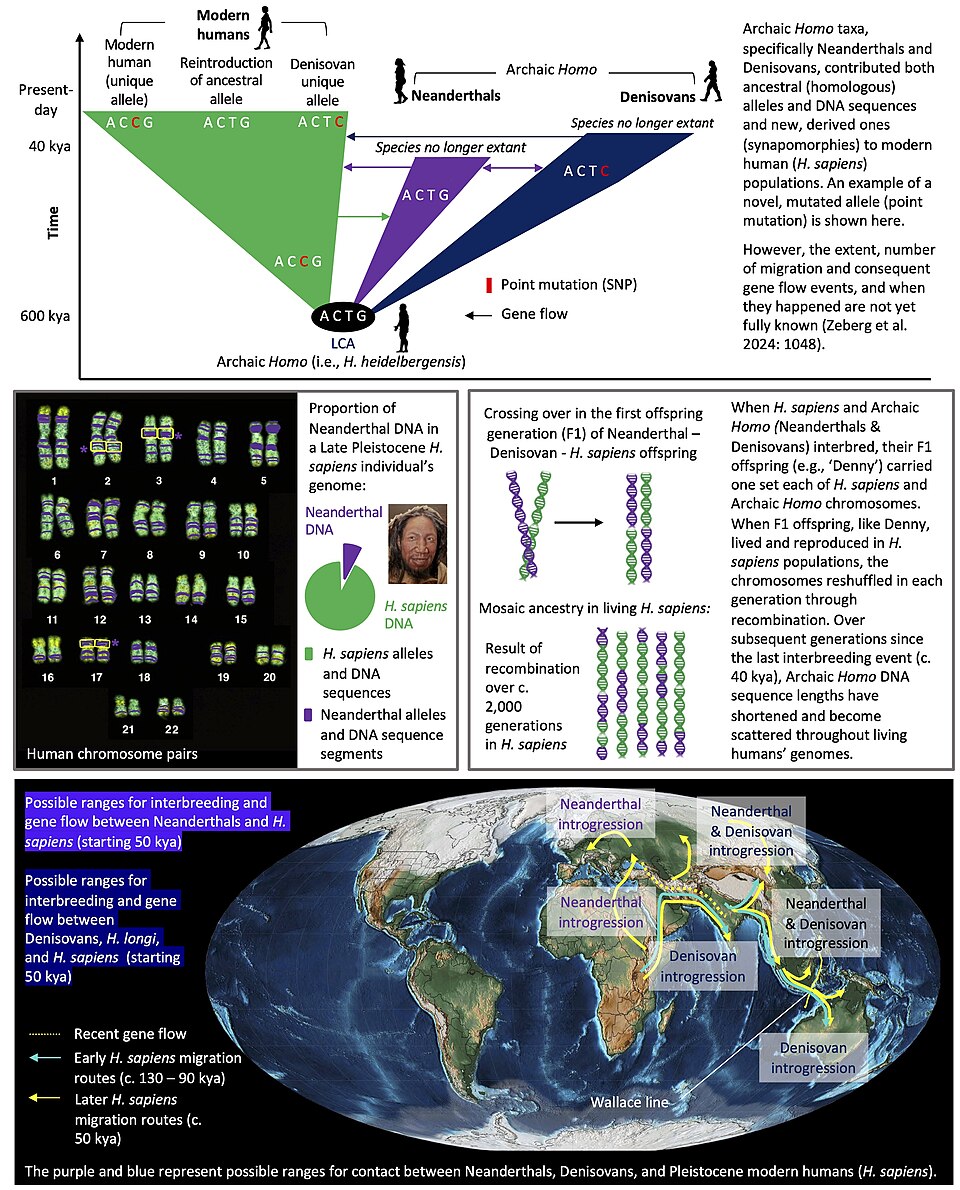
The 2010 draft Neanderthal genome provided the first direct evidence that interbreeding had occurred, revealing that approximately 1 to 4 percent of the genomes of all non-African modern humans derives from Neanderthal ancestors.2 Subsequent studies with larger sample sizes and improved reference genomes refined this estimate to approximately 1.5 to 2.1 percent, with East Asian populations carrying slightly more Neanderthal ancestry than European populations, likely due to additional pulses of admixture or differential selection after the initial contact.4
Kolodny and Feldman developed a neutral model of species drift demonstrating that even without invoking any selective advantage for modern humans, the simple dynamics of migration from a large African source population into a smaller resident Neanderthal population would inevitably lead to the replacement of the latter. In their model, a continuous trickle of modern human migrants from Africa, each contributing genes to the local population, would gradually dilute Neanderthal ancestry below detectable levels over thousands of years, producing an outcome indistinguishable from competitive replacement at the archaeological level.14
However, the genomic evidence also reveals clear signatures of selection against Neanderthal-derived DNA. Sankararaman and colleagues found that Neanderthal ancestry is strongly depleted on the X chromosome and near genes expressed in the testes, a pattern consistent with reduced fertility in male hybrids — a hallmark of partial reproductive incompatibility between divergent populations.4 This finding suggests that while interbreeding occurred, it came with a fitness cost, and natural selection progressively purged Neanderthal alleles from many regions of the hybrid genome. The assimilation model therefore accounts for gene flow but does not fully explain the disappearance of Neanderthals as a morphologically and genetically coherent population.
Small population sizes and inbreeding depression
Perhaps the most fundamental vulnerability of Neanderthal populations was their small size. Archaeological site distributions, genetic diversity estimates, and ecological carrying capacity models all indicate that Neanderthals lived in small, geographically scattered groups with a total effective population size (the genetically relevant number of breeding individuals) estimated at only 1,000 to 5,000 across their entire range in Europe and western Asia.3, 13 By comparison, early modern human populations in Europe, drawing on a much larger African source population, achieved effective population sizes an order of magnitude larger within a few thousand years of arrival.8
The high-coverage genome of a Neanderthal woman from Denisova Cave in the Altai Mountains provided striking molecular evidence of the consequences of small population size. Her genome exhibited long runs of homozygosity indicating that her parents were related at approximately the level of half-siblings. Even after accounting for this recent inbreeding, the Altai Neanderthal genome contained substantially more runs of homozygosity than genomes of modern human populations known to have small effective sizes, and her overall heterozygosity was only 16 to 22 percent of that observed in present-day non-African humans.3
Analysis of a community of eleven Neanderthals from Chagyrskaya Cave in Siberia, reported in 2022, confirmed that this pattern was not anomalous. The group exhibited very low genetic diversity, with estimated community sizes of only 10 to 20 individuals, and showed signs of predominantly patrilocal social organization with female exogamy. The genetic data indicated that this small community was part of a broader population with an effective size of only a few hundred individuals.22
Estimated effective population sizes of Neanderthals and early modern humans3, 8, 13, 22
| Population | Estimated Ne | Evidence basis |
|---|---|---|
| Neanderthals (total metapopulation) | ~1,000–5,000 | Genomic heterozygosity, archaeological site density |
| Altai Neanderthals (local population) | ~3,000–4,000 | High-coverage genome, runs of homozygosity |
| Chagyrskaya community | ~10–20 (community); few hundred (wider population) | Nuclear DNA from 11 individuals |
| Early modern humans in Europe | ~10,000–50,000+ | Archaeological site density, genetic diversity |
| Modern human African source | ~10,000–15,000 | Autosomal genetic diversity |
In 2019, Vaesen and colleagues applied conservation biology principles to ask whether Neanderthal populations could have gone extinct without any external cause at all. Using population viability analyses with the VORTEX simulation software, they modelled the effects of inbreeding depression, Allee effects (the reduction in individual fitness at low population densities, for example through difficulty finding mates), and demographic and environmental stochasticity on populations of the sizes estimated for Neanderthals. Their simulations showed that populations of fewer than approximately 5,000 individuals faced substantial extinction risk from these factors alone over timescales of a few thousand years, even without competition from modern humans or adverse climate change.12 This finding does not mean that Neanderthals were inevitably doomed — they had persisted at low population sizes for hundreds of thousands of years — but it does mean that any additional stressor, whether competitive, climatic, or epidemiological, could have tipped already vulnerable populations below the threshold of viability.
Archaeological evidence for behavioral overlap
A critical question underlying the extinction debate is whether modern humans possessed cognitive or behavioral capacities that Neanderthals lacked. For much of the twentieth century, the prevailing view held that Neanderthals were behaviourally inferior: incapable of symbolic thought, limited to simple stone tool technologies, and lacking the innovative flexibility that characterised modern human culture. This characterisation has been progressively dismantled by archaeological discoveries of the past three decades.16
Roebroeks and Soressi's 2016 review marshalled evidence that the biological and cultural gap between Neanderthals and modern humans was substantially smaller than traditionally assumed. Neanderthals produced Levallois flakes and points requiring sophisticated knapping sequences, manufactured birch bark pitch adhesives implying multi-step chemical processes, constructed wooden hunting spears, and exploited a range of environments from coastal to alpine.16 At Cueva de los Aviones and Cueva Antón in southeastern Spain, perforated and pigment-stained marine shells dating to approximately 50,000 years ago — well before any modern human presence in the region — provide evidence of Neanderthal symbolic behaviour independent of any modern human influence.19
The Châtelperronian assemblages of France and northern Spain provide the most debated case study. At the Grotte du Renne, Neanderthal remains were found in association with personal ornaments including perforated animal teeth and bone awls, in layers stratigraphically interstratified with Aurignacian (modern human) deposits.20 Critics have questioned the stratigraphic integrity of these associations, suggesting that mixing of deposits could have produced a spurious link between Neanderthal fossils and Upper Palaeolithic artefacts. Proponents counter that the stratigraphic evidence, when combined with radiocarbon dating and spatial analysis, supports a genuine association. The debate remains unresolved, but its outcome bears directly on whether Neanderthals were capable of the kind of cultural innovation previously attributed exclusively to modern humans.16, 20
The narrowing of the perceived behavioural gap has important implications for extinction models. If Neanderthals and modern humans were behaviourally similar, then explanations invoking cognitive superiority become less persuasive, and demographic and ecological factors assume greater explanatory weight. Conversely, subtle differences in social organisation, information transmission, or subsistence flexibility — differences that might not be preserved archaeologically — could still have produced the competitive asymmetry necessary to drive one population to extinction.6, 16
The emerging multicausal consensus
The trajectory of research over the past two decades has converged on a multicausal framework for Neanderthal extinction. No single factor — not competition, not climate, not disease, not assimilation — can account for the pattern of disappearance observed in the archaeological and genetic records. Instead, the emerging consensus holds that Neanderthal extinction resulted from the interaction of multiple factors operating simultaneously and synergistically.21
Degioanni and colleagues argued in 2021 that demography was the primary underlying vulnerability. Neanderthal populations were already small and fragmented, rendering them susceptible to the stochastic fluctuations, inbreeding depression, and Allee effects that conservation biologists recognise as threats to any small population. The arrival of modern humans did not need to involve violence, cognitive superiority, or even direct competition in the conventional sense; the simple addition of a more numerous population competing for broadly the same resources would have been sufficient to push Neanderthal populations below viable thresholds, particularly when combined with the episodic climatic stresses of Heinrich stadials that fragmented populations and disrupted social networks.12, 21
The Gilpin, Feldman, and Aoki ecocultural model formalised a version of this scenario, showing that even a small competitive advantage in cultural capacity — defined as the ability to exploit environmental resources — would lead to Neanderthal replacement within a few thousand years when combined with realistic population dynamics.6 Timmermann's climate-competition simulations confirmed that competition was the necessary ingredient: climate fluctuations alone could cause regional population declines but not global extinction, and interbreeding alone could dilute but not eliminate the Neanderthal population. Only competition, whether through demographic advantage, resource exploitation efficiency, or some combination of both, produced extinction timelines consistent with the observed record.7
The genetic legacy of Neanderthals in living humans underscores the complexity of the process. The 1.5 to 2.1 percent of Neanderthal-derived DNA in non-African populations represents the residue of interbreeding that occurred during the period of coexistence — evidence that the two populations were not entirely reproductively isolated and that the boundary between them was permeable.2, 4 At the same time, the strong depletion of Neanderthal ancestry near functionally important genes, particularly those involved in male fertility, indicates that the hybrid offspring carried a fitness cost, and that natural selection has been progressively purging Neanderthal DNA from the modern human genome over the past 40,000 years.4 Neanderthals thus neither went entirely extinct nor were entirely assimilated; they disappeared as a coherent population while leaving a small but measurable genetic footprint in the species that replaced them.
The disappearance of the Neanderthals serves as a case study in the vulnerability of small, fragmented populations to the arrival of a more numerous competitor, particularly during a period of environmental instability. It also demonstrates that extinction need not be sudden or violent; it can be the cumulative result of subtle demographic pressures operating over millennia. In this sense, the extinction of the Neanderthals was less an event than a process — one that unfolded across thousands of years and thousands of kilometres, and whose full complexity is only now, through the integration of archaeology, genetics, and climate science, beginning to be understood.17, 21
References
An ecocultural model predicts Neanderthal extinction through competition with modern humans
Quantifying the potential causes of Neanderthal extinction: abrupt climate change versus competition and interbreeding
Tenfold population increase in Western Europe at the Neandertal-to-modern human transition
Disease transmission and introgression can explain the long-lasting contact zone of modern humans and Neanderthals
Inbreeding, Allee effects and stochasticity might be sufficient to account for Neanderthal extinction
A parsimonious neutral model suggests Neanderthal replacement was determined by migration and random species drift
Radiocarbon dating casts doubt on the late chronology of the Middle to Upper Palaeolithic transition in southern Iberia
Confirmation of Neanderthal/modern human interstratification at the Chatelperronian type-site
An emerging consensus in palaeoanthropology: demography was the main factor responsible for the disappearance of Neanderthals
Impact of climate change on the transition of Neanderthals to modern humans in Europe