Overview
- The geographic distribution of species—which organisms live where, and why—is one of the most powerful lines of evidence for evolution, demonstrating that organisms descended from common ancestors that diversified as landmasses separated and ecological opportunities opened.
- Alfred Russel Wallace's systematic mapping of species boundaries in the Malay Archipelago established biogeography as a rigorous science, and his observation that neighboring islands can hold radically different faunas remains unexplained by any hypothesis other than common descent with modification.
- Molecular phylogeography has transformed biogeography from a descriptive discipline into a quantitative one, allowing scientists to date colonization events, trace dispersal routes, and reconstruct the histories of faunas separated by deep geological time.
Biogeography is the scientific study of the geographic distribution of species and ecosystems across Earth's surface and through geological time. It asks a deceptively simple question: why do particular organisms live where they do, and not elsewhere? The patterns that emerge from carefully mapping the living world—why flightless birds appear on every southern continent but nowhere in the Northern Hemisphere, why Australia's mammals are so unlike those of Asia, why each oceanic island hosts unique species found nowhere else on Earth—are among the most compelling lines of evidence for evolution by common descent.7 These distributions are not random. They reflect the history of life itself: the breakup of ancient supercontinents, the colonization of remote islands by rare dispersal events, the inexorable pressure of natural selection reshaping organisms to fit new environments.
Wallace, Darwin, and the birth of biogeography
The science of biogeography emerged in the nineteenth century from two intertwined intellectual traditions. Charles Darwin observed, during his voyage on HMS Beagle from 1831 to 1836, that the Galápagos Islands harbored species closely resembling mainland South American forms, yet distinctly modified for island conditions. He recognized that proximity, not climate or habitat, determined which species served as the colonizing stock for island faunas—a pattern only explicable if species descended from common ancestors and diversified over time.7
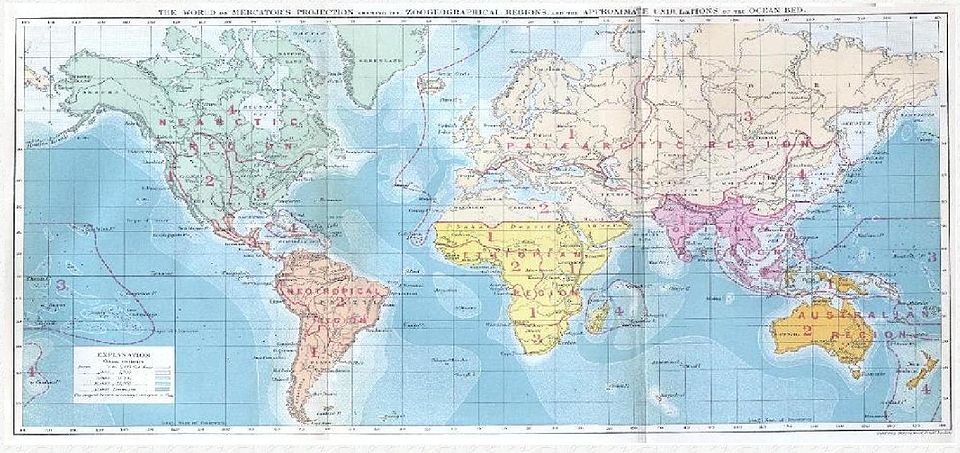
Alfred Russel Wallace, working independently in the Malay Archipelago from 1854 to 1862, arrived at the same conclusion through far more systematic means.1 Wallace collected over 125,000 specimens across the archipelago and painstakingly mapped the distributions of birds, mammals, and insects between islands. He discovered a striking discontinuity: the islands of Borneo, Java, and Bali hosted animals closely related to Asian species, while the neighboring islands of Lombok, Sulawesi, and New Guinea held an entirely different fauna of Australasian character. This boundary, now known as Wallace's Line, ran between Bali and Lombok, two islands separated by only 35 kilometers of water, yet divided by millions of years of evolutionary history.1, 2 Wallace co-published his mechanism of natural selection with Darwin in 1858, and his subsequent monumental work The Geographical Distribution of Animals (1876) established biogeography as a rigorous discipline anchored in evolutionary theory.2, 6
The modern understanding of Wallace's Line incorporates the underlying geology. Borneo and the Sunda Shelf islands sit on the Asian continental plate, which was periodically exposed as a continuous land bridge during Pleistocene glaciations when sea levels fell. The Australasian islands, by contrast, share a separate geological and evolutionary heritage extending back to the ancient supercontinent Gondwana. Animals from either side rarely cross the deep-water straits between them, meaning each side has evolved in isolation for tens of millions of years.9 Ernst Mayr later refined the boundary by identifying Weber's Line further east, which marks the point of equal faunal influence from both Asia and Australia, delineating a transition zone called Wallacea in which the two faunas gradually intermix.26
The theory of island biogeography
The most influential quantitative framework in biogeography was developed in the 1960s by ecologist Robert MacArthur and naturalist E. O. Wilson. Their equilibrium theory of island biogeography proposed that the number of species on any island results from a dynamic balance between two opposing processes: immigration from a mainland source pool, which tends to increase species richness, and local extinction, which tends to reduce it.4 The equilibrium species number—the point at which these two rates are equal—is determined primarily by two factors: the island's area and its distance from the source of colonists. Larger islands sustain larger populations and thus experience lower extinction rates; nearer islands receive more frequent colonists and thus have higher immigration rates.3
The most direct empirical prediction of this model is the species-area relationship: as island area increases, species diversity increases in a consistent, mathematically predictable way. This pattern, first formalized as the equation S = cAz, where S is species number, A is area, c is a constant, and z is a scaling exponent typically between 0.20 and 0.35, has been documented across taxa and geographic regions ranging from Caribbean islands to mountaintop habitats.5 MacArthur and Wilson tested their model dramatically in 1969 by defaunating small mangrove islands in the Florida Keys with methyl bromide and tracking recolonization. The islands returned to their predicted equilibrium species numbers within about a year, with the nearest and largest islands recovering most rapidly.3
The MacArthur-Wilson framework transformed conservation biology by providing a theoretical foundation for understanding habitat fragmentation. A reserve surrounded by inhospitable land functions as an ecological island, and the same equations predict that fragmenting a continuous habitat into smaller patches will inevitably reduce the species each patch can support in the long term.23 The theory also provides an evolutionary framework: islands are incubators of diversification, because immigrants arriving on remote islands often encounter ecological vacancies filled nowhere else, and natural selection can rapidly remodel them to exploit those niches.24
Species-area relationship: approximate bird species richness on selected Caribbean islands5
Vicariance and the legacy of Gondwana
Biogeographers distinguish between two fundamental mechanisms by which populations become geographically isolated and diverge into distinct species. Dispersal biogeography attributes disjunct distributions to the active or passive movement of organisms across pre-existing geographic barriers, while vicariance biogeography attributes them to the formation of barriers that subsequently divide a previously continuous population. The recognition of plate tectonics in the 1960s gave vicariance a powerful geological mechanism: ancient landmasses that once formed a single supercontinent could carry their biotas with them as they drifted apart, isolating populations across ocean basins without any individual organism crossing water.9
The breakup of Gondwana, the southern supercontinent that began fragmenting approximately 180 million years ago, provides some of the most instructive examples of vicariant biogeography. The modern continents of South America, Africa, Antarctica, Australia, and the Indian subcontinent, along with New Zealand and Madagascar, all derive from Gondwana, and the organisms living on them often reflect those ancient connections. The southern beeches (Nothofagus), which dominate cool temperate forests from southern Chile and Argentina to New Zealand, southeastern Australia, and New Guinea, were long regarded as a textbook case of Gondwanan vicariance. Molecular phylogenetic analyses have broadly confirmed that the major Nothofagus lineages diverged in a pattern consistent with the sequence of Gondwanan landmass separations, though some trans-oceanic dispersal events have also been implicated.10
The lungfishes offer another striking example. Only six species of lungfish survive today, distributed between three southern-hemisphere continents: the Australian lungfish (Neoceratodus forsteri), four African species of the genus Protopterus, and the South American lungfish (Lepidosiren paradoxa). These three independently isolated lineages trace their divergence to the breakup of Gondwana, and molecular clock analyses date their separation to time periods consistent with the known geological sequence of continental fragmentation.13
The ratites—the group of large, flightless birds that includes the ostrich of Africa, the rhea of South America, the emu and cassowary of Australia and New Guinea, and the extinct moa of New Zealand—were traditionally interpreted as the quintessential Gondwanan vicariance story. However, molecular phylogenetics has substantially complicated this picture. Several studies, including a comprehensive 2008 analysis of 40 independent nuclear loci, have demonstrated that ratites are not monophyletic; that is, they do not form a single natural group descended from one flightless ancestor.11 Rather, flightlessness evolved independently multiple times within a group whose ancestors were capable of flight. A 2014 study using ancient DNA from elephant bird bones confirmed that the kiwi of New Zealand is most closely related to the elephant bird of Madagascar, implying that ancestors of these birds flew across ocean barriers after the relevant continents had already separated.12 This finding illustrates that vicariance and dispersal are not mutually exclusive; distinguishing between them requires careful integration of molecular phylogenetics and geological timescales.
Adaptive radiation on islands
Oceanic islands—those formed by volcanic activity rising from the ocean floor and never connected to any continent—provide the clearest examples of adaptive radiation, the process by which a single colonizing lineage diversifies rapidly into multiple species occupying different ecological roles. Because such islands are colonized rarely, the first organisms to establish themselves often encounter ecological space with few competitors, and natural selection acts powerfully to diversify morphology and behavior as populations spread into different niches.14
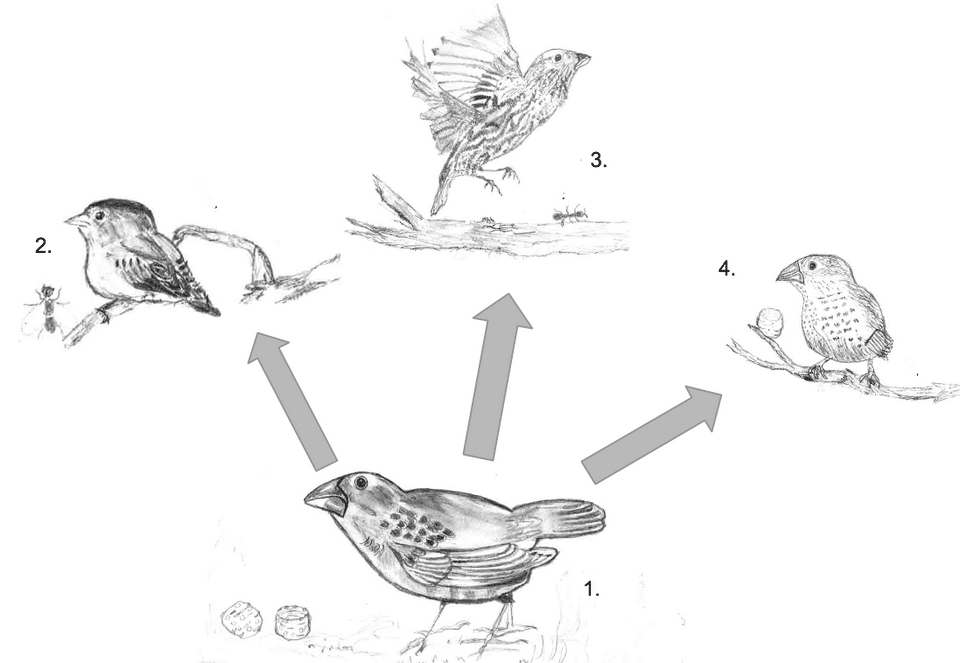
Darwin's finches of the Galápagos Islands represent the most famous case. From a single ancestor that colonized the archipelago from mainland South America, approximately 18 species have evolved with bills ranging from the massive, nut-cracking beak of the large ground finch to the needle-like bill of the warbler finch to the woodpecker-like bill of the tool-using woodpecker finch.15 Long-term field studies by Peter and Rosemary Grant on Daphne Major island have documented natural selection acting on beak morphology in real time, with beak size and shape shifting measurably across years in response to changes in seed availability driven by El Niño events and droughts.15 Whole-genome sequencing of all Galápagos finch species confirmed that they form a monophyletic group, all descended from one founding population, and identified specific genomic regions associated with beak shape variation.16
The Hawaiian honeycreepers provide an equally striking example. From a single ancestral cardueline finch that colonized the Hawaiian Islands roughly 5.7 to 7.2 million years ago, approximately 56 species are known (though many are now extinct), exhibiting extraordinary variation in bill morphology correlated with diet: thin, curved bills for nectar feeding from tubular flowers, thick bills for seed cracking, and straight, insect-probing bills for bark gleaning.17 Molecular phylogenetic analysis confirmed the monophyletic origin of the entire Hawaiian honeycreeper radiation within the finch family, demonstrating that the spectacular diversity arose entirely through in situ evolution rather than multiple colonizations.17
Madagascar presents perhaps the most dramatic example of island radiation at a continental scale. The island separated from the African mainland approximately 165 million years ago and from the Indian subcontinent roughly 88 million years ago, allowing its biota to evolve in near-complete isolation.18 The lemurs, which today encompass over 100 species ranging from the 30-gram mouse lemur to the 9-kilogram indri, descended from a single ancestral primate that likely rafted from Africa on floating vegetation mats approximately 60 million years ago, after Madagascar had already separated.19 In the absence of competing monkeys and apes, lemurs radiated into an extraordinary diversity of ecological niches and are found nowhere else on Earth. More than 90 percent of Madagascar's plant and animal species are endemic, making it one of the world's foremost biodiversity hotspots and a living demonstration of the evolutionary consequences of prolonged isolation.18
Oceanic islands and the absence of native mammals
One of the most striking biogeographic patterns, first noted by Darwin, is that oceanic islands are almost entirely devoid of native non-flying land mammals.7 Continental islands—those formed by the fragmentation of continental landmasses or separated from continents by shallow seas that were periodically exposed during glaciations—typically retain mammals because their ancestors walked across land bridges before isolation. By contrast, oceanic islands, formed volcanically from the ocean floor, can only be colonized by organisms capable of crossing open water: birds, bats (which can fly), reptiles and insects (which can survive long ocean voyages on debris), and seeds transported by wind, water, or animals. Mammals, with their high metabolic demands and limited ability to survive prolonged ocean crossings, almost never establish themselves on distant oceanic islands under natural conditions.
This pattern is globally consistent. The Hawaiian Islands, which lie over 3,000 kilometers from the nearest continent, had no native land mammals at all before the arrival of Polynesian settlers, who introduced the rat and the dog. The native mammalian fauna consisted entirely of two bat species and the Hawaiian monk seal, which is marine.17 The Galápagos, the Azores, and the Canary Islands all show the same pattern. Where mammals do appear on oceanic islands—such as the small rodents that colonized the Galápagos—they arrived by rafting, usually from nearby continental shorelines, and the absence of other mammalian competitors allowed them to diversify into ecological roles normally occupied by other mammals on continents.7 This predictable absence of native mammals on oceanic islands, and its neat explanation by colonization capability and isolation distance, is a straightforward prediction of evolutionary biogeography.
The distribution of marsupials
The geographic distribution of marsupials—the pouched mammals that give birth to highly altricial young and nurture them in a pouch—presents one of biogeography's most instructive puzzles. Today, marsupials are overwhelmingly concentrated in Australia and New Guinea, with one family, the opossums, extending through South America and one species reaching North America. This peculiar distribution once seemed anomalous, but it makes complete sense in the context of plate tectonics and evolutionary history.

Marsupials originated in the Northern Hemisphere during the Cretaceous period and dispersed southward, reaching South America and, via Antarctica, Australia, when these landmasses were still connected as part of the remnants of Gondwana.20 The subsequent isolation of Australia—which fully separated from Antarctica approximately 34 million years ago—allowed its marsupial fauna to evolve independently, radiating into the diverse array of kangaroos, wallabies, wombats, possums, bandicoots, quolls, and the thylacine (now extinct) that characterized the continent before European arrival.20 In the absence of placental mammals (which separately diversified in the Northern Hemisphere and colonized the southern continents later), marsupials filled ecological niches occupied by placentals elsewhere, producing the classic example of convergent evolution: the thylacine converging on the morphology of a wolf, the marsupial mole on the body plan of a placental mole, and the sugar glider paralleling the flying squirrel.8
A comprehensive molecular phylogenetic study published in 2011, drawing on data from 26 genetic loci across all living marsupial orders, reconstructed the branching order of marsupial diversification and confirmed that the timing and sequence of major lineage splits are broadly consistent with the geological timing of Gondwanan landmass separations and subsequent dispersal routes.21 The distribution of marsupials thus provides a coherent story only when plate tectonics and evolutionary theory are combined: without one, the other leaves the distribution inexplicable.
Molecular phylogeography
The development of molecular genetics transformed biogeography from a discipline relying primarily on morphology, fossils, and the geometry of continental configurations into one capable of reconstructing the specific histories of populations across landscapes. Molecular phylogeography, a term formalized by John Avise in the 1980s and elaborated in his 2000 synthesis, uses DNA sequence variation—especially in mitochondrial DNA and nuclear markers—to infer the geographic histories of species: where populations originated, how they dispersed, where they contracted during past climate changes, and how they subsequently re-expanded.22
The power of molecular phylogeography lies in its ability to assign approximate dates to demographic and geographic events using molecular clocks, which calibrate the rate at which DNA sequences diverge over time. By comparing the genetic divergence between populations on either side of a geographic barrier—a mountain range, a river, a sea strait—with the independently known timing of that barrier's formation, researchers can test whether observed genetic breaks correspond to the expected vicariant events or imply earlier or later dispersal.22 This approach has been applied at scales ranging from the phylogeography of individual species across river drainages to the reconstruction of entire regional faunas.
Molecular phylogeography has also shed light on the role of Pleistocene climate cycles in shaping present-day distributions. During glacial maxima, large parts of Europe, North America, and northern Asia were covered by ice sheets, forcing cold-adapted species southward and compressing the ranges of temperate organisms into refugia—areas that remained habitable. When climates warmed and ice retreated, species expanded out of these refugia, and the genetic signatures of these expansions are detectable in the reduced genetic diversity at range peripheries compared to refugial cores.22 The concept of refugia and post-glacial recolonization has since been applied globally, from the forests of tropical Africa to the insect faunas of the Himalayas.
The discipline has also contributed to understanding biological invasions: the introduction of species to regions outside their native ranges through human activity. Molecular markers can identify the specific source populations of invasives, distinguish single from multiple introduction events, and track the spread of invasions over time.25 These insights are directly relevant to conservation and management, enabling practitioners to assess whether different populations of an invasive represent independent introductions or a single founding event, and to trace the pathways through which invasions proceed.
The biogeographic regions of the world
Wallace's systematic mapping of animal distributions led him to propose a division of the Earth's land surface into major biogeographic realms, each characterized by a distinctive assemblage of animal families found there and nowhere else. His six realms—the Palearctic (Europe and most of Asia), Nearctic (North America), Neotropical (Central and South America), Ethiopian (sub-Saharan Africa), Oriental (tropical Asia), and Australasian—remain in wide use today, though refined by subsequent work.2 These regions reflect the deep history of continental configurations: the Holarctic realms (Palearctic and Nearctic) share many families because North America and Eurasia were periodically connected by the Bering land bridge, while the Australasian realm is the most distinctive because Australia's prolonged isolation has resulted in the highest proportion of endemic families of any major landmass.
The marine realm presents different biogeographic patterns, because the ocean is a continuous medium with no absolute barriers comparable to mountain ranges or ocean trenches. Marine biogeographers identify provinces defined by temperature regimes, current systems, and the distribution of suitable habitat such as coral reefs. The Indo-Pacific province, stretching from the eastern coast of Africa to the central Pacific, is the most species-rich marine region on Earth and is thought to have served as a center of origin for many reef-associated taxa, with species dispersing outward from this center to colonize peripheral regions.27
The coherence of biogeographic regions and the predictability of their faunal compositions from geological and evolutionary history constitute a global-scale confirmation of the predictions of evolutionary theory. If species had been created independently and placed arbitrarily across the Earth's surface, no such regions would exist. The existence of clearly defined realms, each harboring families that evolved from common ancestors within that region and that differ from families in neighboring realms in proportion to the age and completeness of the intervening barriers, is exactly what evolutionary theory predicts and what the evidence confirms.2, 7
Biogeography as evidence for evolution
The consilience between multiple independent lines of biogeographic evidence and the predictions of evolutionary theory is among the strongest arguments for common descent. The same distributions that Wallace and Darwin found compelling in the nineteenth century are now confirmed and deepened by plate tectonics, molecular clocks, and whole-genome sequencing. Organisms are not distributed according to climate or habitat alone: tropical Africa and tropical South America share broadly similar climates but host entirely different families of animals, because their faunas evolved independently after the supercontinent of Gondwana fragmented.9 Oceanic islands consistently lack native land mammals but harbor spectacular radiations of birds and reptiles, exactly as predicted by their colonization history rather than by any property of the climate or habitat.7 The genetic divergence between island populations and their mainland relatives is proportional to the time since colonization, as measured independently by geology—a prediction that falls naturally out of evolutionary theory but is otherwise unmotivated.22
The Australian fauna demonstrates the ultimate biogeographic argument for evolution. When Australia was isolated from Antarctica and subsequently from all other landmasses, it carried with it a marsupial-dominated mammalian fauna. Cut off from the placental mammals evolving elsewhere, that fauna diversified into ecological analogs of wolves, moles, flying squirrels, and anteaters—convergent in body plan but entirely distinct in ancestry, as confirmed by every level of comparison from anatomy to chromosome structure to DNA sequence.8, 21 The pattern of convergent evolution on isolated landmasses, in which unrelated lineages independently evolve similar forms in response to similar selective pressures, would be deeply puzzling under any hypothesis except evolution. Under evolutionary theory, it is the expected consequence of the same selective forces acting on different raw material in geographically separated environments.
In this sense, biogeography does not merely provide ancillary support for evolution. It provides an independent, continent-scale natural experiment, replicated across every ocean and repeated across geological time, whose results consistently match the predictions of evolutionary theory and consistently fail to match any alternative explanation. The geography of life is, in the end, the autobiography of evolution written in the distribution of species.
References
Gondwanan vicariance revealed by the phylogeny of the Southern beech genus Nothofagus
Ancient DNA reveals elephant birds and kiwi are sister taxa and clarifies ratite bird evolution
Islands as model systems in ecology and evolution: prospects fifty years after MacArthur-Wilson