Overview
- The evidence for evolution comes from multiple independent scientific disciplines—comparative anatomy, molecular biology, biogeography, embryology, paleontology, and direct observation—each providing a distinct line of support that converges on the same conclusion: all life on Earth shares common ancestry.
- At the molecular level, shared endogenous retroviruses at identical genomic locations, a fused chromosome matching two separate ape chromosomes, 98.7% DNA identity between humans and chimpanzees, and a broken vitamin C gene disabled by the same mutations in all simian primates all point unambiguously to common descent.
- Evolution is also directly observable: antibiotic resistance in bacteria is natural selection operating in real time, producing heritable changes in populations within days or weeks.
The case for evolution does not rest on a single line of argument. It is built from the convergence of evidence from every branch of the biological sciences—comparative anatomy, embryology, molecular biology, paleontology, biogeography, and direct observation—each discipline contributing an independent body of data that points to the same conclusion: all life on Earth shares common ancestry and has diversified through descent with modification over billions of years.1, 2 The strength of this evidence lies not merely in the volume of data but in its independence. The phylogenetic tree reconstructed from DNA sequences matches the tree reconstructed from the fossil record, which matches the tree reconstructed from comparative anatomy, which matches the tree implied by the geographic distribution of species. This concordance across entirely different types of data, collected by different scientists using different methods, is precisely what one would expect if the inferred evolutionary history is real.3, 5
Some of the most compelling evidence is molecular. The human genome carries within it a detailed record of evolutionary history: ancient viral infections shared with other primates at identical chromosomal locations, a fused chromosome that corresponds to two separate chromosomes in the other great apes, near-identity in DNA sequence with chimpanzees, and a broken gene for vitamin C synthesis disabled by the same mutations found in all simian primates. Each of these molecular signatures is independently explicable only by common descent.6, 7, 8, 9 Evolution is also directly observable in the present: the rapid emergence of antibiotic-resistant bacteria is natural selection acting on heritable variation in real time, documented in hospitals and laboratories worldwide.4, 10
Anatomy, embryology, and biogeography
Comparative anatomy reveals two patterns that make sense under common descent but are puzzling under independent creation.

The first is homology: structures in different species that share the same underlying architecture despite serving different functions. The forelimb of a human, the wing of a bat, the flipper of a whale, and the front leg of a horse all contain the same set of bones—humerus, radius, ulna, carpals, metacarpals, and phalanges—arranged in the same relative positions, modified in proportion and shape to suit different functions.1, 2 This shared structural plan is exactly what evolution predicts: these limbs were inherited from a common ancestor and modified by natural selection for different uses. The second pattern is vestigiality: structures that have lost all or most of their original function but persist as reduced remnants of ancestral anatomy. The human body retains the coccyx (a fused tailbone from tailed ancestors), ear muscles that cannot move the ears, a diminished appendix, and wisdom teeth that frequently lack room to erupt—each an inheritance from ancestors in which these structures served important functions.2, 3
Embryology provides a complementary line of evidence. Vertebrate embryos pass through developmental stages that recapitulate features of their evolutionary ancestors. Human embryos develop pharyngeal arches homologous to the gill arches of fish; these structures are remodeled during development into the jaw, middle ear bones, and throat cartilages rather than forming gills, but their transient appearance records the aquatic ancestry of all terrestrial vertebrates. Human embryos also develop and then resorb a tail during the first weeks of gestation—a structure that persists in adults only as the vestigial coccyx.2, 3
Biogeography—the study of where species live and why—adds a geographic dimension to the evidence. Oceanic islands are consistently populated by organisms related to those on the nearest continent, not by the species best suited to the island environment. Darwin's finches on the Galápagos are related to South American finches; the honeycreepers of Hawaii descended from a single Asian finch ancestor; the lemurs of Madagascar are all primates descended from a common ancestor that rafted from mainland Africa. These distributions reflect evolutionary history and dispersal, not optimal design.1, 3
Molecular evidence
The sequencing of whole genomes has provided evidence for common descent of a precision that Darwin could not have imagined. The 2005 sequencing of the chimpanzee genome revealed that human and chimpanzee DNA is 98.77% identical at aligned nucleotide positions—a degree of similarity that reflects the recent divergence of these two lineages from a common ancestor approximately six to eight million years ago.6 The differences that do exist between the two genomes are concentrated in precisely the categories predicted by population genetics: neutral substitutions that accumulated by drift, structural variants (insertions, deletions, and duplications), and a small number of positively selected changes in genes related to immunity, reproduction, and brain development.6

Human chromosome 2 provides a particularly striking piece of evidence. Humans have 23 pairs of chromosomes; chimpanzees, gorillas, and orangutans each have 24 pairs. In 1991, researchers at Yale University discovered that human chromosome 2 contains a fusion site where two ancestral chromosomes joined end to end, with telomeric repeat sequences arranged head to head at the junction and a vestigial centromere from the second ancestral chromosome still detectable nearby. The two ancestral chromosomes correspond precisely to chimpanzee chromosomes 2A and 2B.7 Endogenous retroviruses provide another class of evidence. When a retrovirus infects a germ-line cell and integrates into the host genome, the insertion is passed to all descendants. Because the probability of independent insertions at the same genomic location is vanishingly small, shared ERVs at identical positions in two species constitute strong evidence of common ancestry. Humans and chimpanzees share thousands of such insertions, and their distribution across primate genomes produces a phylogenetic tree that matches the tree derived from morphology and other molecular data.9
The broken vitamin C gene (GULO) adds yet another independent line of molecular evidence. Most mammals synthesize their own vitamin C, but humans and all other simian primates cannot because the GULO gene, which encodes the enzyme for the final step of vitamin C biosynthesis, is disabled by the same pattern of mutations in all these species. The shared pattern of inactivating mutations—the same exon deletions at the same positions—is explicable only by inheritance from a common ancestor in which the gene was first disabled.8
Direct observation
Evolution is not only inferred from historical evidence; it is directly observed in living populations.
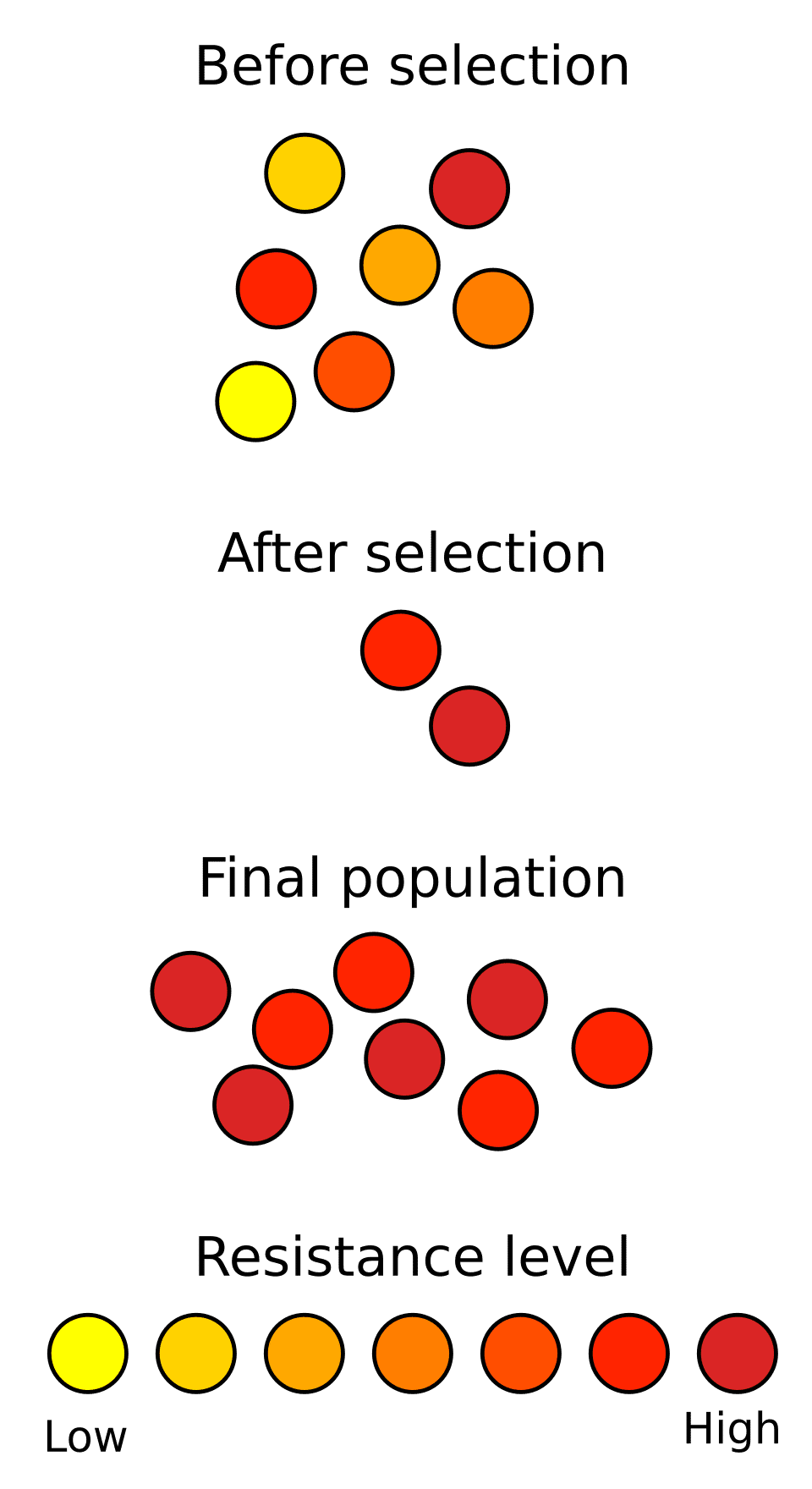
The most vivid contemporary example is antibiotic resistance in bacteria, in which populations exposed to antibiotics rapidly evolve resistance through heritable mutations and horizontal gene transfer. Methicillin-resistant Staphylococcus aureus (MRSA) evolved within two years of methicillin's introduction in 1959; vancomycin-resistant enterococci emerged within decades of vancomycin's clinical use. The World Health Organization now identifies antibiotic resistance as one of the greatest threats to global public health—a crisis driven entirely by the natural selection of heritable variation in bacterial populations.4, 10
These independent lines of evidence—from anatomy, embryology, biogeography, molecular biology, and direct observation—do not merely support evolution. They converge on the same detailed evolutionary history, the same branching tree of relationships, the same timeline of divergences. This convergence from independent sources, each subject to different types of error and bias, is the hallmark of a well-established scientific conclusion.3, 4
References
Lineage-specific expansions of retroviral insertions within the genomes of African great apes but not humans and orangutans