Overview
- Modern whales retain vestigial hind limb bones—a reduced pelvis and, in some species, femur remnants—embedded in muscle with no connection to the spine and no locomotor function, directly tracing their ancestry to four-legged land mammals.
- Baleen whale embryos develop tooth buds that begin mineralization before being reabsorbed prior to birth, recapitulating a developmental program inherited from toothed ancestors documented in the fossil series from Pakicetus through Basilosaurus.
- Occasional atavistic hind limb protrusions in living whales, vestigial finger bones inside flippers preserving the ancestral pentadactyl pattern, and hundreds of non-functional olfactory receptor pseudogenes all confirm that cetacean genomes retain the developmental blueprints of their terrestrial forebears.
The evolutionary transition from land to sea in the cetacean lineage—from small, four-legged terrestrial mammals to the streamlined whales, dolphins, and porpoises of today—is one of the best-documented major transitions in the fossil record. But the evidence does not reside solely in fossils. The bodies of living cetaceans retain multiple vestigial structures inherited from their terrestrial ancestors: reduced pelvic and hind limb bones buried in muscle, finger bones concealed inside flippers, tooth buds in embryonic baleen whales that are reabsorbed before birth, and hundreds of non-functional olfactory receptor genes. Each of these vestiges records a chapter of cetacean evolutionary history, and together they trace a continuous line from the walking ancestors documented in the fossil record to the fully aquatic descendants alive today.1, 3
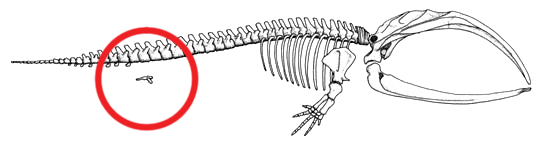
Vestigial hind limb bones
Modern whales possess vestigial pelvic bones—small, rod-shaped or irregular bony elements embedded in the ventral body wall musculature, with no articulation to the vertebral column and no locomotor function. In most species, only the pelvis (innominate bone) is present, but in some larger whales, remnants of the femur have also been identified. These bones are located in the posterior abdominal region, roughly where the hind limbs of a terrestrial mammal would attach.5, 6
Struthers's detailed nineteenth-century anatomical studies of the humpback whale (Megaptera novaeangliae) documented the vestigial pelvic bones and their associated musculature, noting their clear homology to the mammalian pelvis and their complete functional disconnection from the axial skeleton.5 Dines et al. demonstrated that cetacean pelvic bones, while vestigial for locomotion, have been maintained under sexual selection in some species because they serve as attachment points for muscles controlling the penis. The size and shape of the pelvis correlate with the degree of sexual dimorphism and mating behavior across cetacean species, suggesting that these vestigial bones have been co-opted for a secondary function even as their original locomotor role was entirely lost.6
The developmental basis of hind limb loss in cetaceans has been elucidated at the molecular level. Thewissen et al. showed that in dolphin embryos, hind limb buds form during early development and initially express the same suite of limb-patterning genes (including Shh, Fgf8, and Hox genes) found in all tetrapod embryos. However, expression of the critical signaling molecule Sonic hedgehog (Shh) is extinguished early in development, causing the limb bud to arrest its growth and regress. The hind limb developmental program is not absent from the cetacean genome; it is initiated and then aborted.7
Atavistic hind limb protrusions
Occasionally, individual whales or dolphins are found with externally visible hind limb protrusions—atavisms in which the normally suppressed hind limb developmental program is partially reactivated. Documented cases include humpback whales with small but recognizable hind limb buds protruding from the body wall, some containing cartilaginous or bony elements corresponding to femur and tibia remnants.8
These atavistic limbs are significant because they demonstrate that the genetic instructions for building hind limbs remain present in the cetacean genome, suppressed but not deleted. When the normal developmental repression fails, the ancient program can be partially reactivated, producing structures that have not been functional in the cetacean lineage for tens of millions of years. Atavisms are a predicted consequence of the evolutionary mechanism: if whales descended from four-legged ancestors, the genes for hind limb development should still be present (as they are needed for forelimb development as well, with the hind limb program suppressed by regulatory changes rather than gene deletion). Occasional reactivation is exactly what one would expect.7, 8
Finger bones inside flippers
Whale flippers, though externally paddle-shaped and functionally analogous to fish fins, contain an unmistakable internal skeleton that reveals their true nature: they are modified tetrapod forelimbs. Inside every whale flipper are bones homologous to the humerus, radius, ulna, carpals, metacarpals, and phalanges of a terrestrial mammal's arm and hand. The pentadactyl (five-fingered) pattern is clearly recognizable, though individual bones are shortened and flattened into a more paddle-like configuration.1, 3
In some cetacean species, the number of phalanges (finger bones) per digit has increased beyond the ancestral mammalian condition—a phenomenon called hyperphalangy—elongating the flipper for more efficient swimming. But the underlying five-digit architecture is preserved. This is the pattern expected if flippers evolved from ancestral limbs through modification rather than being designed from scratch for aquatic locomotion: an engineer designing a paddle would not embed five separate chains of small bones inside it, but evolution works by modifying existing structures rather than starting over.1
Vestigial tooth buds in baleen whales
Baleen whales (mysticetes)—including blue whales, humpback whales, and right whales—are filter feeders that strain small organisms from seawater using plates of baleen, keratinous structures that hang from the upper jaw. Adult baleen whales have no teeth. Yet during embryonic development, baleen whale fetuses develop tooth buds that begin the process of mineralization before being reabsorbed prior to birth.9, 9
Karlsen first documented this phenomenon in the minke whale, showing that tooth germs form in both the upper and lower jaws of fetal mysticetes, pass through the bud, cap, and bell stages of normal tooth development, and then undergo resorption as baleen plates begin to form in the upper jaw.9 Thewissen et al. provided a more detailed developmental analysis, demonstrating that the tooth buds of fetal baleen whales express the same genetic markers (including Shh, Bmp4, and Fgf8) found in the developing teeth of toothed mammals. The genes for tooth formation are present and active in baleen whale embryos; they are simply shut down before the teeth emerge.9
This observation is powerfully consistent with the fossil record, which documents a transition from toothed whale ancestors (archaeocetes such as Basilosaurus and Dorudon) to early mysticetes that possessed both teeth and rudimentary baleen, to modern mysticetes that have lost teeth entirely and rely exclusively on baleen filtration. The vestigial tooth buds of living baleen whales are a developmental echo of this evolutionary transition, preserved because the genetic program for tooth development is deeply integrated into the mammalian developmental toolkit and has not been entirely deleted.9
Non-functional olfactory receptor genes
Terrestrial mammals depend heavily on olfaction, and the olfactory receptor (OR) gene family—the largest gene family in the mammalian genome—reflects this dependence. Mice possess approximately 1,200 OR genes, the majority of which are functional. Cetaceans, having returned to an aquatic environment where airborne odorants are irrelevant, have undergone massive pseudogenization of their OR gene repertoire. Toothed whales (odontocetes) have lost nearly all functional OR genes, retaining only a handful; baleen whales retain somewhat more, consistent with their retention of limited olfactory capability for detecting airborne chemical cues at the water's surface.10
The broken OR pseudogenes in cetacean genomes are not simply absent—they are present, recognizable by sequence comparison, and disabled by the accumulation of stop codons, frameshift mutations, and deletions. They occupy the same genomic positions as their functional counterparts in terrestrial mammals, bearing witness to an ancestral condition in which these genes worked.10, 11
The pseudogenization of olfactory genes in cetaceans extends beyond OR genes to other components of the olfactory signaling pathway. Kishida and colleagues demonstrated that the olfactory marker protein (OMP) gene, which encodes a protein essential for normal olfactory signal transduction in terrestrial mammals, has been independently inactivated in multiple cetacean lineages. In toothed whales, which have completely lost the olfactory apparatus, OMP carries multiple frameshift mutations and premature stop codons. In baleen whales, which retain rudimentary olfactory structures, OMP shows a lesser degree of degradation, consistent with the retention of limited olfactory function in mysticetes.14
Pseudogenized dental genes in baleen whales
Genomic analyses have extended the evidence from embryonic tooth buds to the genes themselves. Demere and colleagues examined the genes encoding enamel-specific proteins (amelogenin, ameloblastin, and enamelin) and dentin-specific proteins in baleen whales and found that these genes are progressively degraded in mysticete genomes. The enamelin gene (ENAM), for instance, carries inactivating mutations in all baleen whales examined, consistent with the complete loss of functional enamel production. The shared inactivating mutations in ENAM across baleen whale species follow a phylogenetic pattern that matches the known relationships among mysticete families, with some mutations shared by all baleen whales (indicating inactivation in a common ancestor) and others specific to particular lineages (indicating continued degradation after divergence).15
Broader genomic surveys have revealed that the transition to aquatic life involved the coordinated loss of function across entire suites of genes associated with terrestrial traits. Chikina and colleagues identified convergent patterns of accelerated sequence evolution in genes associated with skin, hair, and lung function across three independently aquatic mammalian lineages: cetaceans, sirenians (manatees), and pinnipeds (seals). Genes that are highly conserved in terrestrial mammals have experienced relaxed selection or positive selection in aquatic lineages, reflecting the shared demands of the aquatic environment.16 These convergent genomic signatures complement the anatomical evidence from vestigial structures, showing that the land-to-sea transition is recorded not only in the visible structures of living whales but also in the molecular details of their genomes.
Connection to the fossil record
The vestigial structures of living cetaceans gain their full significance when placed in the context of the fossil series documenting the land-to-sea transition. Pakicetus (~50 million years ago) was a small, wolf-sized mammal with four fully functional limbs, a long tail, and a skull bearing features diagnostic of early whales.12 Ambulocetus (~49 Ma) was a semiaquatic predator with large, webbed feet adapted for both walking and swimming. Rodhocetus (~47 Ma) had reduced hind limbs and a more streamlined body. Basilosaurus (~40 Ma) was fully aquatic with a serpentine body and tiny, non-functional hind limbs that retained a complete set of leg bones including femur, tibia, fibula, and toes.4, 13
The vestigial structures of modern whales are the end point of this series. The vestigial pelvis and femur remnants of living whales are the further-reduced descendants of the tiny hind limbs of Basilosaurus. The finger bones inside whale flippers are the modified descendants of the walking forelimbs of Pakicetus. The tooth buds of embryonic baleen whales recapitulate a developmental stage that corresponds to the toothed condition of archaeocete ancestors. The broken olfactory receptor genes record the progressive loss of a sensory system no longer needed in aquatic life.1, 3, 13 Taken together, the vestigial structures of living cetaceans and the transitional fossils of their ancestors tell a coherent, mutually reinforcing story of evolutionary transformation from land to sea.
References
A morphological analysis of the pelvis and hind limb vestige of the humpback whale (Megaptera novaeangliae)
Developmental basis for hind-limb loss in dolphins and origin of the cetacean body plan
Loss of olfactory receptor genes coincides with the acquisition of full trichromatic vision in primates
Convergent patterns of adaptive evolution associated with the aquatic lifestyle in mammals