Overview
- Whales evolved from small, terrestrial artiodactyl ancestors in the early Eocene, approximately 53 million years ago, and the transition from land to sea is documented by a remarkably complete series of fossil intermediates including Pakicetus, Ambulocetus, Rodhocetus, and Basilosaurus.
- Molecular phylogenetics and morphological evidence converge in placing Cetacea as the sister group of Hippopotamidae within Artiodactyla, a relationship confirmed by shared retroposon insertions, protein-coding genes, and the uniquely derived double-pulley astragalus bone.
- The two living cetacean suborders diverged in the late Eocene: odontocetes evolved biosonar through progressive modification of the melon and nasal passages, while mysticetes replaced teeth with keratinous baleen plates, enabling filter-feeding strategies that exploit dense aggregations of small prey.
The evolution of whales from small, terrestrial mammals to the largest animals ever to inhabit the Earth is one of the most thoroughly documented macroevolutionary transitions in the fossil record. Over a span of approximately 15 million years during the Eocene epoch, the ancestors of modern cetaceans underwent a dramatic series of anatomical transformations: the hind limbs were progressively reduced and eventually lost as functional appendages, the nostrils migrated from the tip of the snout to the top of the skull to form a blowhole, the tail developed into a powerful horizontal fluke, and the body became streamlined for efficient aquatic locomotion.2, 13 This transition is supported by an extraordinary wealth of fossil intermediates discovered primarily in Eocene-age sediments of Pakistan, India, and Egypt, as well as by molecular phylogenetic evidence that places whales firmly within the even-toed ungulates (Artiodactyla) as the sister group of hippopotamuses.6, 7, 9
Phylogenetic position within Artiodactyla
For much of the twentieth century, the evolutionary origin of whales was deeply uncertain. The earliest known fossil cetaceans bore little obvious resemblance to any living mammalian order, and morphological analyses variously allied them with mesonychid condylarths, an extinct group of carnivorous ungulates that possessed similar triangular-cusped teeth. The molecular revolution of the 1990s overturned this picture. Analyses of mitochondrial DNA, nuclear protein-coding genes, and long interspersed nuclear elements (LINEs) consistently placed Cetacea within Artiodactyla, specifically as the sister group of Hippopotamidae.6, 23 This result was initially controversial because it rendered the traditional order Artiodactyla paraphyletic — hippos were more closely related to whales than to other even-toed ungulates such as pigs or camels.
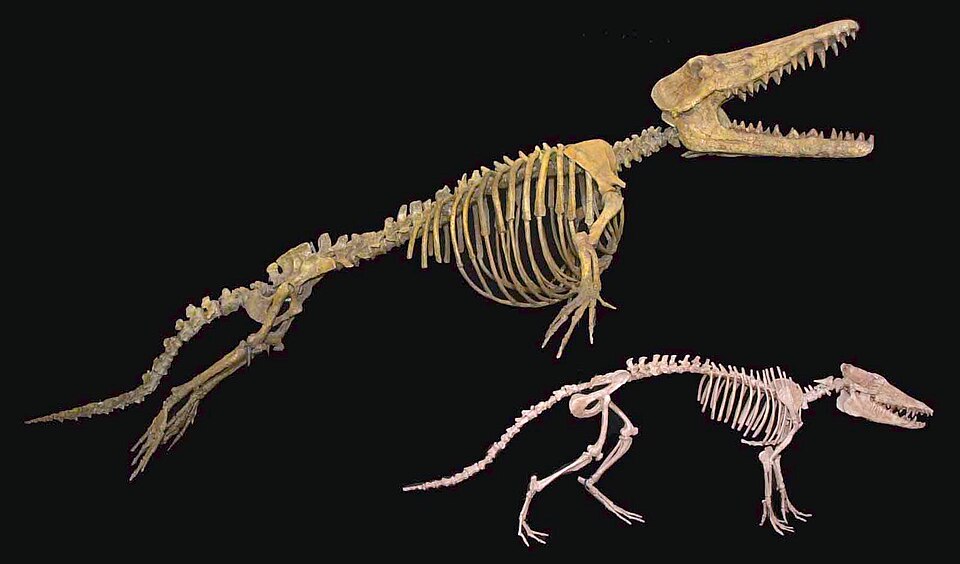
The molecular evidence was decisively corroborated by the discovery of early cetacean fossils preserving the ankle bones. In 2001, Thewissen and colleagues described Pakicetus and other pakicetid archaeocetes from Pakistan whose astragalus (ankle bone) exhibited the double-pulley morphology that is the defining skeletal synapomorphy of Artiodactyla.1 No mesonychid condylarth has ever been found with this feature. The same year, Gingerich and colleagues reported well-preserved feet of the protocetid Rodhocetus that also bore the artiodactyl astragalus.3 The convergence of molecular and morphological evidence on the same phylogenetic conclusion — that whales are artiodactyls, and that hippos are their closest living relatives — represents one of the most compelling cases of independent lines of evidence confirming a single evolutionary hypothesis.7
Retroposon insertions provided particularly strong support for this relationship. Short interspersed nuclear elements (SINEs) are transposable elements that insert themselves at random locations in the genome. Because the probability of two independent insertions occurring at precisely the same genomic position is vanishingly small, shared SINE insertions between two lineages constitute virtually irreversible phylogenetic markers. Shimamura and colleagues identified multiple SINE insertions shared exclusively by cetaceans and hippopotamuses, confirming their sister-group relationship.23 Subsequent retroposon analyses further established the monophyly of toothed whales (Odontoceti) and the internal relationships among major cetacean lineages.8
Pakicetids: the earliest cetaceans
The oldest known cetaceans belong to the family Pakicetidae and date to the early Eocene, approximately 53 to 48 million years ago. Pakicetus inachus, first described by Gingerich and colleagues in 1983 from the Kuldana Formation of Pakistan, was initially known only from a partial skull, but subsequent discoveries have yielded much of the postcranial skeleton.12, 1 Pakicetids were wolf-sized animals with long snouts, heterodont dentition, and limbs fully capable of terrestrial locomotion. Their skeletal proportions indicate that they were cursorial or subcursorial runners, not yet committed to an aquatic lifestyle.1, 22
Despite their terrestrial body plan, pakicetids exhibit several features that link them to later, more aquatic cetaceans. The ear region of Pakicetus shows a thickened tympanic bulla (the bony capsule enclosing the middle ear) composed of dense bone, a feature shared with all later archaeocetes and modern cetaceans that enhances sound transmission in water.5 However, the ear of Pakicetus still retained an air-filled middle ear cavity and a tympanic membrane, indicating that it heard primarily through the airborne sound pathway, like terrestrial mammals, rather than through bone conduction of waterborne sound as in fully aquatic cetaceans.5, 15
Stable isotope analyses of oxygen in pakicetid tooth enamel have provided insights into their ecology. The oxygen isotope ratios of Pakicetus teeth are consistent with ingestion of freshwater rather than seawater, suggesting that these animals lived in or near freshwater habitats such as rivers and lakes in the subtropical environments of the Indo-Pakistani region.20, 16 This finding aligns with the sedimentological context of pakicetid fossils, which are typically recovered from fluvial and marginal marine deposits rather than open marine sediments.
Ambulocetids and remingtonocetids
The next stage in cetacean evolution is represented by the ambulocetids, known from middle Eocene sediments approximately 48 to 47 million years old. Ambulocetus natans, described by Thewissen, Hussain, and Arif in 1994, is one of the most informative transitional fossils in the cetacean record. Approximately 3 metres in length, Ambulocetus had a robust body with large hind feet and a long, powerful tail, suggesting that it swam using a combination of pelvic paddling and dorsoventral undulation of the spine, somewhat analogous to the locomotion of modern otters or crocodilians.10, 22 Its forelimbs were relatively short, and its hind limbs retained fully functional joints and digits, indicating that it was capable of locomotion on land, though it was almost certainly clumsier on shore than Pakicetus.
The skull of Ambulocetus exhibits eyes positioned high on the head and retracted nostrils that had begun to shift posteriorly from the tip of the snout, both adaptations consistent with an ambush-predator lifestyle at the water surface.10, 13 Isotopic data from Ambulocetus teeth indicate tolerance of both freshwater and brackish or marine conditions, suggesting a transition into coastal and estuarine environments.16
The remingtonocetids, a contemporaneous family known from the middle Eocene of India and Pakistan, represent a parallel experiment in aquatic adaptation. These animals had remarkably elongated skulls with long, narrow snouts, small eyes, and large auditory bullae, suggesting an emphasis on underwater hearing and possibly prey detection in turbid waters. Their postcranial skeleton indicates semiaquatic habits, though they retained four functional limbs.9, 19 Remingtonocetids are generally considered a side branch of early cetacean evolution rather than direct ancestors of later whales, but they illuminate the diversity of ecological strategies explored by early cetaceans as they transitioned into aquatic environments.
Protocetids: entering the open sea
The Protocetidae, dating from approximately 48 to 38 million years ago, represent the first cetaceans to disperse beyond the Indo-Pakistani region and into open marine environments. Protocetid fossils have been found across a wide geographic range, including Pakistan, India, Egypt, Nigeria, and the southeastern United States, indicating that these animals had achieved sufficient aquatic competence to cross oceanic barriers.3, 4
Rodhocetus kasrani, described from the middle Eocene of Pakistan, is among the best-known protocetids. Its skeleton reveals a body plan intermediate between the semiaquatic ambulocetids and the fully aquatic later archaeocetes. The vertebral column of Rodhocetus shows adaptations for powerful dorsoventral flexion of the lumbar and caudal regions, consistent with tail-powered swimming, yet the sacral vertebrae were still partially fused to the pelvis, and the hind limbs retained functional knee and ankle joints.3, 22 The feet were large and probably webbed, serving as effective paddles. Critically, the ankle of Rodhocetus preserved the artiodactyl double-pulley astragalus, providing one of the key pieces of morphological evidence linking cetaceans to even-toed ungulates.3
The protocetid Maiacetus inuus, described in 2009, yielded a remarkable specimen: a pregnant female preserving a near-term fetus positioned for head-first delivery, the orientation typical of terrestrial mammals. This finding indicates that protocetids still gave birth on land, despite spending much of their lives in the water, and suggests that the transition to fully aquatic parturition (tail-first delivery, as in modern cetaceans) had not yet occurred at this stage.4
Basilosaurids and the loss of hind limbs
The Basilosauridae, which flourished from approximately 41 to 34 million years ago during the late Eocene, were the first fully aquatic cetaceans. Basilosaurus isis, known from spectacular specimens recovered in the Fayum Depression of Egypt, reached lengths of 15 to 18 metres and had an elongated, serpentine body powered by a large tail fluke. Its forelimbs had been transformed into paddle-like flippers, and the sacrum was completely unfused from the pelvis, severing the structural connection between the hind limbs and the vertebral column that is necessary for weight-bearing locomotion on land.11, 13

The hind limbs of Basilosaurus were dramatically reduced but not entirely absent. Gingerich, Smith, and Simons described small but well-formed hind limbs in B. isis that included a femur, patella, tibia, fibula, and three-toed foot with all the major joints intact. These limbs were far too small to support the animal's weight or contribute to locomotion, measuring only approximately 60 centimetres in length on an animal exceeding 15 metres. Their function, if any, remains debated, though their retention of articular surfaces has led to speculation that they may have served as clasping organs during mating.11 Regardless of function, the vestigial hind limbs of Basilosaurus constitute powerful evidence for descent from terrestrial ancestors: they are a morphological remnant of the four-limbed body plan that the lineage inherited from its artiodactyl forebears.
Dorudon atrox, a smaller basilosaurid approximately 5 metres in length and contemporaneous with Basilosaurus, is generally considered a better model for the body plan ancestral to modern cetaceans. Dorudon had a more compact body, a well-developed tail fluke, and an even more reduced pelvis and hind limb than Basilosaurus. Its skull morphology, particularly the structure of the ear region, shows features transitional between the earlier archaeocetes and the two modern suborders, Odontoceti and Mysticeti.13, 14
Progressive hind limb reduction during cetacean evolution11, 22
Nostril migration and blowhole development
One of the most visually striking anatomical transformations in cetacean evolution is the progressive migration of the nostrils from the front of the snout to the top of the skull, forming the blowhole that allows modern whales to breathe while keeping most of the body submerged. This migration is documented in exquisite detail by the fossil sequence. In Pakicetus, the nostrils are positioned at the tip of the snout, as in typical terrestrial mammals.1, 12 In Ambulocetus, the nasal opening has shifted slightly posteriorly, to a position roughly midway along the upper surface of the snout.10 In protocetids such as Rodhocetus, the nostrils have migrated further toward the middle of the skull.3 In basilosaurids, the nasal opening is located well behind the snout, approaching the position seen in modern cetaceans. And in extant whales, the blowhole sits at or near the apex of the skull, enabling efficient breathing at the water surface with minimal expenditure of energy.13, 14
The developmental mechanism underlying nostril migration involves the differential growth of the bones of the face and skull roof. During embryonic development, the premaxillary and maxillary bones of the snout elongate posteriorly relative to the nasal bones, effectively telescoping the skull so that the nasal opening is displaced rearward. This process of cranial telescoping also reshuffles the positions of other skull elements and is a defining feature of cetacean cranial anatomy. Modern odontocetes carry telescoping to an extreme, with the maxillary and premaxillary bones extending far behind the nasal opening to overhang the braincase, a configuration associated with the evolution of the melon and the sound-producing nasal passages used in echolocation.18
Evolution of hearing and echolocation
The transition from aerial to underwater hearing represents one of the most functionally significant sensory transformations in cetacean evolution. Terrestrial mammals hear by conducting airborne sound waves through the external ear canal to the tympanic membrane (eardrum), which transmits vibrations through the ossicular chain (malleus, incus, stapes) to the cochlea of the inner ear. This mechanism is poorly suited to the aquatic environment because the impedance difference between water and air causes most waterborne sound energy to be reflected rather than transmitted at air-filled interfaces.5, 15
The fossil record documents the stepwise modification of the cetacean ear from a terrestrial to an aquatic configuration. In Pakicetus, the tympanic bulla is thickened and composed of dense bone (pachyosteosclerotic), a derived cetacean feature, but the middle ear still relied on the tympanic membrane for sound reception, as in terrestrial mammals.5 In the remingtonocetid Remingtonocetus, the tympanic plate had begun to thin and the mandibular foramen had enlarged, suggesting the initial development of a novel sound pathway through the lower jaw — the fat-filled mandibular canal that transmits waterborne vibrations directly to the middle ear in modern odontocetes.15, 19 By the basilosaurid stage, the ear was essentially isolated from the skull by air-filled sinuses, enhancing directional hearing underwater by preventing sound from reaching the cochlea through the skull bones and allowing precise localization of sound sources.5
Echolocation, or biosonar, is the active production and reception of high-frequency sound used by odontocetes to navigate, find prey, and perceive their environment. Toothed whales generate clicks in specialized nasal passages (the phonic lips, or monkey lips/dorsal bursae) located just below the blowhole, and these clicks are focused into a directional beam by the melon, a fatty structure in the forehead. Echoes returning from objects in the environment are received through the fat-filled mandibular canal and transmitted to the cochlea.21 Fossil evidence suggests that the anatomical prerequisites for echolocation — the asymmetric skull, enlarged nasal passages, and fat-filled lower jaw — were present in early odontocetes by the late Eocene or early Oligocene, approximately 34 to 30 million years ago, coinciding with the divergence of odontocetes from the stem cetacean lineage.8, 18
Evolution of baleen in mysticetes
The Mysticeti, or baleen whales, are the other major living cetacean suborder and include the largest animals ever to have existed, notably the blue whale (Balaenoptera musculus). Modern mysticetes are edentulous, possessing instead plates of keratinous baleen that hang from the upper jaw and function as a sieve for filter-feeding on krill, copepods, and small fish. The evolutionary origin of baleen from a toothed ancestor has been illuminated by a series of fossil discoveries and developmental genetic studies.17
The earliest known mysticetes, such as Aetiocetus from the late Oligocene of Oregon, possessed both teeth and nutrient foramina on the palate that are consistent with the vascularization needed to support baleen growth. This combination of teeth and putative baleen in a single individual suggests that the transition from teeth to baleen involved an intermediate stage in which both structures coexisted, with the teeth eventually being lost as baleen became the primary feeding apparatus.17 Genetic studies have identified the molecular remnants of this transition: modern mysticetes retain pseudogenes (non-functional, mutated copies) of the enamel-related genes enamelin and ameloblastin, confirming that their ancestors possessed functional teeth and that the genes for tooth formation were inactivated after baleen evolved as a replacement structure.17, 14
The evolution of baleen-based filter feeding opened an entirely new ecological niche for cetaceans, enabling them to exploit vast populations of small organisms at low trophic levels. This feeding strategy, combined with the evolution of lunge feeding in rorqual whales and continuous ram filtering in right and bowhead whales, permitted mysticetes to achieve body sizes far exceeding those of any toothed predator, culminating in the blue whale, which can reach 30 metres in length and over 170 tonnes in mass.14
Converging lines of evidence
The strength of the case for whale evolution rests not on any single line of evidence but on the convergence of multiple independent data sources. The fossil record provides a temporally ordered series of morphological intermediates spanning the transition from a terrestrial artiodactyl body plan to a fully aquatic one. Molecular phylogenetics, using mitochondrial DNA, nuclear genes, and retroposon insertions, independently confirms the placement of cetaceans within Artiodactyla and their sister-group relationship with hippopotamuses. Stable isotope geochemistry traces the ecological transition from freshwater-dependent Pakicetus through estuarine Ambulocetus to fully marine protocetids and basilosaurids. And developmental biology reveals the genetic toolkit for limb reduction, nostril migration, and tooth loss that was modified during the land-to-sea transition.6, 7, 9, 16

The isotopic evidence has been particularly informative for reconstructing the ecological habitats of successive cetacean genera. Oxygen isotope ratios (δ18O) in tooth enamel reflect the isotopic composition of ingested water: freshwater has lower δ18O values than seawater. Analyses by Roe and colleagues and by Clementz and colleagues have shown that pakicetids consumed freshwater, ambulocetids drank both freshwater and marine water, and protocetids were fully marine in their water intake, providing a geochemical record of the ecological transition that parallels the morphological one.20, 16
The combined weight of these independent lines of evidence has made whale evolution one of the most celebrated examples of macroevolutionary change in the scientific literature. The transition is frequently cited in evolutionary biology textbooks and reviews as a case in which the fossil record, molecular systematics, isotope geochemistry, and developmental genetics converge on a single, coherent narrative of adaptive transformation.2, 13, 14
Key transitional genera in cetacean evolution2, 13, 22
| Genus | Age (Ma) | Locomotion | Habitat | Key features |
|---|---|---|---|---|
| Pakicetus | ~53–48 | Terrestrial | Freshwater | Artiodactyl ankle, thickened tympanic bulla |
| Ambulocetus | ~48–47 | Semiaquatic | Freshwater/estuarine | Large hind feet, retracted nostrils |
| Remingtonocetus | ~48–43 | Semiaquatic | Coastal marine | Elongated skull, enlarged auditory bullae |
| Rodhocetus | ~47–41 | Aquatic (tail-powered) | Marine | Reduced sacral fusion, artiodactyl ankle |
| Basilosaurus | ~41–34 | Fully aquatic | Marine | Vestigial hind limbs, tail fluke |
| Dorudon | ~41–34 | Fully aquatic | Marine | Compact body, transitional ear |
References
Origin of whales from early artiodactyls: hands and feet of Eocene Protocetidae from Pakistan
New protocetid whale from the middle Eocene of Pakistan: birth on land, precocial development, and sexual dimorphism
Retroposon analysis of major cetacean lineages: the monophyly of toothed whales and the paraphyly of river dolphins
Pakicetus inachus, a new archaeocete (Mammalia, Cetacea) from the early-middle Eocene Kuldana Formation of Kohat (Pakistan)
Isotopic approaches to understanding the terrestrial-to-marine transition of the earliest cetaceans
A new, diminutive whale from the middle Eocene of Pakistan and its implications for the evolution of cetacean hearing
Stable isotope ratios of carbon and oxygen in early whale teeth from Pakistan record diet and climate
Land-to-sea transition in early whales: evolution of Eocene Archaeoceti (Cetacea) in relation to skeletal proportions and locomotion of living semiaquatic mammals