Overview
- Fitness landscapes, originally conceptualised by Sewall Wright in 1932, represent the relationship between genotype (or phenotype) and fitness as a multidimensional surface with peaks, valleys, and ridges, providing a powerful visual metaphor for understanding how populations evolve toward adaptive optima.
- The topology of a fitness landscape, whether smooth with a single global peak or rugged with many local peaks separated by fitness valleys, profoundly affects evolutionary dynamics: smooth landscapes permit gradual hill-climbing by natural selection, while rugged landscapes can trap populations on suboptimal local peaks.
- Modern research using empirical fitness landscapes from microbial experiments, NK models of epistasis, and neutral network theory has revealed that real biological landscapes are often highly rugged, that neutral mutations play a critical role in enabling populations to traverse fitness valleys, and that Wright's shifting balance theory remains controversial as an explanation for peak shifts in nature.
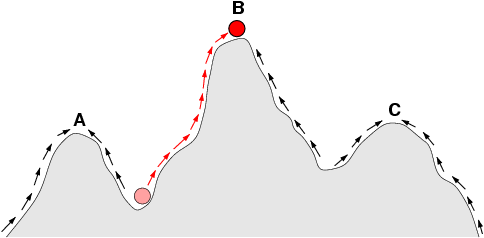
A fitness landscape is a conceptual and mathematical representation of the relationship between genotype (or phenotype) and biological fitness, depicted as a surface in multidimensional space where each point corresponds to a particular combination of genetic variants and the height of the surface at that point represents the fitness, typically measured as reproductive success, of an organism bearing that genotype. The concept was introduced by the American geneticist Sewall Wright in 1932 as a visual tool for thinking about how natural selection, genetic drift, mutation, and migration interact to drive the evolution of populations.1 On a fitness landscape, natural selection pushes populations "uphill" toward peaks of higher fitness, while genetic drift can cause random movement across the surface, and the topology of the landscape, whether it is smooth with a single global peak or rugged with many local peaks separated by valleys of lower fitness, determines which evolutionary trajectories are accessible and how predictable evolution will be.1, 6 The fitness landscape metaphor has become one of the most widely used conceptual tools in evolutionary biology, population genetics, and molecular evolution, although its interpretation and applicability have been debated vigorously for nearly a century.3, 15
Wright's original concept
Sewall Wright introduced the fitness landscape in a 1932 address to the Sixth International Congress of Genetics, in which he argued that the relationship between genotype and fitness could be visualised as a multidimensional surface with peaks and valleys. Wright imagined a coordinate system in which each axis represented the frequency of a different allele in the population, and the height of the surface at each point represented the mean fitness of a population with that combination of allele frequencies. Peaks on this surface corresponded to gene combinations that produced high fitness, and valleys corresponded to combinations that produced low fitness.1
Wright's landscape was explicitly a genotype-frequency landscape: the axes were allele frequencies in a population, and the surface described the population's mean fitness as a function of its genetic composition. This is distinct from a second common interpretation, the genotype landscape, in which each point represents a single individual genotype (not a population) and the height represents the fitness of that genotype. The two interpretations lead to qualitatively different pictures: a genotype landscape describes the fitness of individual organisms as a function of their genetic makeup, while Wright's population-level landscape describes how the average fitness of a population changes as allele frequencies shift. Much confusion in the literature has arisen from conflating these two meanings.3, 15
Wright argued that natural selection alone would drive a population to the nearest peak on the fitness landscape, but that this peak might not be the global optimum. In a rugged landscape with many local peaks of different heights, a population that ascends the nearest peak via natural selection becomes trapped there, unable to reach a higher peak without first descending through a valley of lower fitness, which selection opposes. This problem motivated Wright's shifting balance theory, his proposal for how populations might escape local peaks and find higher ones through a combination of genetic drift in small subpopulations, selection within subpopulations, and migration between them.1
Fisher's geometric model and the Fisher-Wright debate
Ronald Fisher, Wright's contemporary and intellectual rival, offered a fundamentally different perspective on the geometry of adaptation. In his 1930 book The Genetical Theory of Natural Selection, Fisher conceptualised adaptation not as a population navigating a rugged landscape but as an organism approaching a single optimal phenotype in a multidimensional phenotype space. Fisher's geometric model treats the organism as a point in n-dimensional phenotype space, with the optimum at the origin, and asks what size of mutational step is most likely to move the organism closer to the optimum.8
Fisher showed that the probability of a random mutation being beneficial decreases as the number of phenotypic dimensions increases and as the magnitude of the mutation increases. In one dimension, a mutation of any size has a 50 percent chance of moving toward the optimum (if the organism is not already at it). But in many dimensions, a large random mutation is almost certainly deleterious because it is overwhelmingly likely to overshoot or move in the wrong direction in at least some dimensions. The implication is that adaptation proceeds through many small mutational steps, not through large jumps, and that the fitness landscape near the optimum is approximately smooth and single-peaked, not rugged and multipeak as Wright envisioned.8, 9
The Fisher-Wright debate thus centred on whether the fitness landscape was fundamentally smooth (Fisher's view, implying that natural selection could efficiently drive populations to the global optimum through gradual change) or fundamentally rugged (Wright's view, implying that populations frequently become trapped on suboptimal local peaks and require additional mechanisms such as drift to escape).4, 15 This debate shaped the development of population genetics throughout the twentieth century and continues to influence how evolutionary biologists think about adaptation.
Martin and colleagues tested Fisher's geometric model empirically by measuring the fitness effects of random mutations in Arabidopsis thaliana plants transplanted into wild field conditions. They found that the distribution of fitness effects was well predicted by the model's key parameter, the number of effective phenotypic dimensions under selection, which they estimated at approximately six to eight for their study organism. Importantly, the data confirmed Fisher's prediction that mutations of larger effect were more likely to be deleterious, and that beneficial mutations tended to be of small effect, lending empirical support to the gradualist view of adaptation that Fisher championed.9
Rugged landscapes and epistasis
The topography of a fitness landscape is determined largely by epistasis, the phenomenon in which the fitness effect of a mutation depends on which other mutations are present in the genome. When epistasis is absent (that is, when the effects of all mutations on fitness are strictly additive), the fitness landscape is smooth with a single global peak, and natural selection can reach the optimum by accumulating beneficial mutations one at a time in any order. When epistasis is present, the landscape can become rugged, with multiple peaks separated by valleys, ridges connecting peaks, and saddle points where the direction of selection reverses depending on the genetic background.2, 12
Stuart Kauffman's NK model, introduced in his 1993 book The Origins of Order, provides a tuneable mathematical framework for studying rugged fitness landscapes. In the NK model, a genome consists of N loci, each of which contributes to fitness in a way that depends on K other loci (its epistatic partners). When K = 0, there is no epistasis and the landscape has a single smooth peak. As K increases, the landscape becomes progressively more rugged, with more local peaks, deeper valleys, and a more jagged topography. When K = N − 1 (maximum epistasis, every locus interacts with every other), the landscape is essentially random, with every genotype having a fitness that is uncorrelated with its neighbours.2
Effect of epistasis (K) on landscape ruggedness in the NK model2
The NK model demonstrates a fundamental tradeoff in landscape architecture. Smooth landscapes (low K) are easy to navigate: natural selection reliably finds the single global peak. However, the global peak on a smooth landscape is typically of modest height because the contributions of individual loci are independent and simply add up. Rugged landscapes (high K) have higher peaks (because epistatic combinations can produce fitness values that exceed the sum of individual effects), but these peaks are harder to find because populations become trapped on suboptimal local peaks. Kauffman called the intermediate regime, where landscapes are rugged enough to have high peaks but smooth enough to be navigable, the "edge of chaos" and argued that biological organisms evolve in this regime.2
Neutral networks and holey landscapes
A major development in fitness landscape theory came from the recognition that many mutations are selectively neutral, having no measurable effect on fitness. Motoo Kimura's neutral theory of molecular evolution, proposed in 1968, demonstrated that the majority of evolutionary changes at the molecular level are driven not by natural selection but by the random fixation of neutral mutations through genetic drift. In the context of fitness landscapes, neutral mutations correspond to horizontal moves across the landscape: steps that neither ascend nor descend but traverse a flat region of equal fitness.3
The concept of neutral networks formalises this idea. A neutral network is a connected set of genotypes that all have the same fitness, forming a plateau or ridge on the fitness landscape through which a population can drift without experiencing selection for or against the change. Van Nimwegen, Crutchfield, and Huynen showed that populations evolving on neutral networks tend to drift toward the centre of the network, where genotypes have the most neutral neighbours, a phenomenon they called "neutral evolution of mutational robustness." This drift toward the interior of neutral networks means that populations under neutral evolution become more robust to future mutations, a form of preadaptation that emerges without direct selection for robustness.7
Sergey Gavrilets extended neutral network theory into the concept of holey landscapes, in which the fitness landscape consists of a vast network of high-fitness genotypes connected through neutral or nearly neutral paths, perforated by "holes" of low fitness (lethal or highly deleterious genotypes). On a holey landscape, populations can move between very different genotypes, and even between different adaptive peaks, without ever descending through a fitness valley, simply by diffusing along the neutral network. Gavrilets argued that holey landscapes provide a resolution to the problem of peak shifts without requiring Wright's shifting balance mechanism: if neutral networks connect distant regions of genotype space, populations can reach new adaptive zones through neutral drift followed by selection on the new genetic background.3
The holey landscape model has particular relevance for speciation. Gavrilets showed that on holey landscapes, geographically separated populations drifting independently along neutral networks will inevitably diverge in genotype, and when they diverge sufficiently, hybrids between them may fall into the "holes" in the landscape (have low fitness), producing reproductive isolation as a by-product of neutral divergence. This provides a theoretical basis for the Dobzhansky-Muller model of genetic incompatibilities without requiring any adaptive divergence between the populations.3
Empirical fitness landscapes
For most of its history, the fitness landscape concept was primarily a theoretical and metaphorical tool. Measuring a fitness landscape requires determining the fitness of every genotype in a defined set, which is feasible only for small numbers of loci with small numbers of alleles. However, advances in molecular biology, high-throughput mutagenesis, and microbial experimental evolution have made it possible to measure empirical fitness landscapes for small sets of mutations in specific genes or organisms.6
One of the most influential empirical studies was conducted by Weinreich and colleagues, who measured the fitness landscape for five mutations in the β-lactamase gene of Escherichia coli that together confer a 100,000-fold increase in resistance to the antibiotic cefotaxime. With five binary loci, there are 25 = 32 possible genotypes, and the researchers measured the fitness (cefotaxime resistance) of all 32. They found that of the 120 possible mutational pathways from the ancestral genotype to the fully resistant genotype, only 18 were selectively accessible (meaning that every step along the path increased fitness). The remaining pathways were blocked by sign epistasis, in which a mutation that is beneficial on some genetic backgrounds is deleterious on others, creating fitness valleys that natural selection cannot cross.5
This finding had profound implications for the predictability of evolution. If the number of accessible pathways is small relative to the total number of possible pathways, then evolution on rugged landscapes may be more predictable than previously thought, because only a few trajectories are available for selection to follow. Conversely, the existence of multiple accessible pathways means that replicate populations starting from the same genotype may follow different evolutionary trajectories to the same peak, a phenomenon observed in experimental evolution studies with bacteria and viruses.5, 6
De Visser and Krug reviewed the growing body of empirical fitness landscape studies and identified several general patterns. First, sign epistasis is common: approximately 25 to 50 percent of mutations change sign (from beneficial to deleterious or vice versa) depending on genetic background. Second, reciprocal sign epistasis, the type of epistasis that creates multiple fitness peaks, is less common but not rare. Third, empirical landscapes tend to be less rugged than the random landscapes predicted by NK models with high K, suggesting that real biological landscapes have a structure that facilitates, rather than impedes, adaptation.6
The shifting balance theory
Wright's shifting balance theory was his proposed solution to the problem of local peak trapping on rugged fitness landscapes. The theory proceeds in three phases. In the first phase (random drift), small subpopulations undergo genetic drift that moves them away from their current local peak, occasionally pushing them into the domain of attraction of a different, possibly higher, peak. In the second phase (mass selection), natural selection within the shifted subpopulation drives it uphill to the new peak. In the third phase (interdeme selection), the subpopulation on the higher peak produces more emigrants than subpopulations on lower peaks, and the spread of migrants carrying the higher-fitness genotype causes other subpopulations to shift to the new peak as well.1
The shifting balance theory was one of the most debated ideas in evolutionary genetics throughout the twentieth century. Its proponents argued that it provided a mechanism for populations to explore the fitness landscape more broadly than natural selection alone would permit, and that the theory explained the prevalence of population subdivision and the importance of drift-selection interactions in shaping genetic architecture.14
Critics raised several objections. Coyne, Barton, and Turelli argued in an influential 1997 review that the conditions required for the shifting balance to operate are highly restrictive. Phase one requires that subpopulations are small enough for drift to move them off a local peak, which requires effective population sizes on the order of tens to hundreds, much smaller than many natural populations. Phase three requires that the fitness advantage of the new peak is large enough to overcome the homogenising effects of gene flow from surrounding subpopulations still on the lower peak. And the entire process requires that the fitness landscape has the right topography: multiple peaks of substantially different heights, separated by shallow enough valleys that drift can cross them. Coyne and colleagues argued that these conditions are rarely met simultaneously in nature and that most evidence cited in support of the shifting balance can be explained more parsimoniously by direct selection within large populations.4
Wade and Goodnight countered that experimental studies of interdeme selection in laboratory populations of Tribolium beetles provided evidence for all three phases of the shifting balance, and that the theory's requirements were not as restrictive as critics claimed when applied to species with naturally subdivided population structures.14 The debate remains unresolved, with most evolutionary geneticists regarding the shifting balance as theoretically possible but of uncertain importance relative to other mechanisms of adaptation in nature.4, 15
Genotype-phenotype maps and evolvability
The fitness landscape depends critically on the genotype-phenotype map, the complex set of developmental, physiological, and molecular processes that translate genetic variation into phenotypic variation. Because natural selection acts on phenotypes rather than directly on genotypes, the structure of the genotype-phenotype map determines the shape of the fitness landscape as seen by selection. Two genotypes that differ by a single mutation may produce very different phenotypes (if the mutation falls in a critical regulatory region) or nearly identical phenotypes (if the mutation is in a redundant or buffered part of the genome), and these differences profoundly affect the local topography of the fitness landscape.11, 15
The concept of evolvability, the capacity of a population to generate heritable phenotypic variation that is adaptive, is closely linked to the structure of the genotype-phenotype map. Kirschner and Gerhart argued that certain features of biological organisation, including modularity (the semi-independent development of distinct body parts), weak regulatory linkage (the ability of regulatory circuits to be rewired without disrupting other functions), and exploratory systems (developmental processes that can produce adaptive outcomes without specific genetic instructions), enhance evolvability by ensuring that the phenotypic effects of mutations are biased toward the types of variation that are most likely to be selectively useful.11
In terms of fitness landscapes, evolvability corresponds to the property that mutational steps from a given genotype preferentially access regions of genotype space with higher or at least equal fitness, rather than falling randomly into valleys. Organisms with high evolvability inhabit regions of the landscape where the local topography is relatively smooth, with many neutral or beneficial mutations available, while organisms with low evolvability are surrounded by steep fitness drops on all sides.7, 11
The concept of recombination adds a further dimension to genotype-phenotype mapping and its effect on landscape navigation. Sexual recombination can bring together beneficial mutations that arose in different genetic backgrounds, potentially allowing populations to cross fitness valleys that would be impassable for asexual populations restricted to single-mutation steps. De Visser and colleagues explored the effect of sex on empirical fitness landscapes and found that recombination can either facilitate or impede adaptation depending on the pattern of epistasis: when beneficial mutations interact positively (synergistic epistasis), recombination helps by assembling them faster; when they interact negatively (diminishing returns epistasis), recombination can break apart favourable combinations and slow adaptation.17 This finding connects the study of fitness landscapes to the long-standing question of why sexual reproduction is so widespread despite its twofold cost, suggesting that the topology of the fitness landscape itself may be a key determinant of the advantage of sex.
Population structure also affects how populations navigate fitness landscapes. Lieberman, Hauert, and Nowak showed that the spatial arrangement of individuals in a population, modelled as a graph of interactions, can amplify or suppress the effects of natural selection relative to drift. Certain population structures act as "evolutionary amplifiers" that increase the fixation probability of beneficial mutations, effectively smoothing the landscape by making it easier for selection to drive populations uphill. Other structures act as "suppressors" that increase the relative importance of drift, potentially facilitating valley-crossing at the cost of slowing adaptation on smooth landscapes.16
Criticisms and continuing significance
The fitness landscape concept has been criticised on several grounds. The most fundamental criticism is that real fitness landscapes are not static: they change as the environment changes, as the population evolves, and as other species coevolve. An adaptive peak under one set of environmental conditions may become a valley under different conditions, and the landscape on which a population is evolving is constantly being reshaped by the population's own evolution (a phenomenon sometimes called "dancing landscapes" or "seascape" models). This dynamism complicates the static picture of populations climbing fixed peaks and raises questions about how useful the metaphor of a fixed landscape really is for understanding long-term evolutionary dynamics.3, 13
A second criticism concerns dimensionality. Wright's original two-dimensional diagrams are visually intuitive but potentially misleading, because real genotype spaces are astronomically high-dimensional. In high-dimensional spaces, the topological properties of landscapes differ qualitatively from those in two or three dimensions. For example, in high dimensions, nearly every point on the landscape is connected to nearly every other point by a path that does not descend, a property of high-dimensional spaces that makes fitness valleys less of a barrier than the two-dimensional metaphor suggests.3
Despite these criticisms, the fitness landscape remains one of the most productive concepts in evolutionary biology. It provides a unifying visual and mathematical framework for thinking about adaptation, constraint, epistasis, neutral evolution, and speciation. Empirical measurements of small-scale fitness landscapes have moved the concept from metaphor to measurement, and theoretical advances in neutral network theory and holey landscapes have enriched the framework far beyond Wright's original formulation.5, 6 As genomic tools continue to improve, enabling the measurement of fitness landscapes for larger numbers of loci across a broader range of organisms, the concept promises to become an increasingly precise and powerful tool for understanding how evolutionary trajectories are shaped by the relationship between genotype, phenotype, and fitness.6
References
Fisher's geometric model predicts the effects of random mutations when tested in the wild
The molecular clock of neutral evolution can be accelerated or slowed by asymmetric spatial structure