Overview
- Isochron dating solves the fundamental problem of unknown initial daughter isotope abundance by plotting cogenetic samples with different parent-to-daughter ratios on a diagram whose slope yields the age and whose y-intercept reveals the initial isotopic composition, requiring no assumptions about the system's starting conditions.
- The method functions as a built-in test for closed-system behavior: samples that have remained chemically isolated since formation define a straight line (a true isochron), while open-system disturbance produces scatter that is detectable through statistical measures such as the mean square weighted deviation (MSWD).
- Isochron techniques underpin some of the most consequential age determinations in Earth science, including Patterson's 1956 Pb-Pb meteorite isochron that first established the 4.55-billion-year age of the Earth, and are applied across the Rb-Sr, Sm-Nd, Lu-Hf, Re-Os, and Pb-Pb isotopic systems.
Isochron dating is a graphical and mathematical technique used in radiometric geochronology to determine the age of rocks and minerals without requiring any assumption about the initial abundance of the radiogenic daughter isotope. In a conventional model age calculation, the analyst must either assume that no daughter isotope was present when the rock formed or independently estimate the initial daughter abundance — an assumption that is often difficult to justify and can introduce significant error.2, 3 The isochron method eliminates this problem by analysing a suite of cogenetic samples (samples that formed at the same time from the same source) with differing ratios of parent to daughter element. When plotted on a diagram of daughter-isotope ratio versus parent-isotope ratio, both normalised to a stable, non-radiogenic reference isotope of the daughter element, the samples define a straight line whose slope is a function of the elapsed time since formation and whose y-intercept directly reveals the initial isotopic composition of the system.1, 2
The isochron approach was formalised by the South African geochemist Louis Nicolaysen in 1961, who demonstrated that suites of cogenetic rock samples with variable rubidium-strontium ratios could be used to determine both the age and the initial strontium isotopic composition simultaneously from a single diagram.1 Since then, the isochron method has become the standard approach for age determination across multiple long-lived radioactive decay systems, including rubidium-strontium, samarium-neodymium, lutetium-hafnium, rhenium-osmium, and lead-lead, and has yielded some of the most important age measurements in Earth and planetary science, including the determination of the age of the Earth itself.2, 9
The problem of initial daughter abundance
All radiometric dating methods are based on the fundamental decay equation, which states that the present-day abundance of a radiogenic daughter isotope in a closed system equals the sum of the initial daughter abundance at the time of formation plus the amount produced by radioactive decay of the parent isotope over the intervening time. For any parent isotope P decaying to daughter isotope D with decay constant λ, the relationship is:
Dnow = Dinitial + Pnow × (eλt − 1)
where t is the age of the sample.2, 3 If Dinitial is known, a single measurement of Dnow and Pnow suffices to calculate t. In practice, however, most rocks incorporate some quantity of the daughter element at the time of their formation. A basalt crystallising from a magma will contain strontium, neodymium, osmium, or lead that was already present in the melt, and there is generally no way to measure the initial daughter abundance directly from a single sample after the rock has accumulated additional radiogenic daughter through radioactive decay.2
Early attempts at radiometric dating addressed this problem by analysing minerals with very high parent-to-daughter ratios, where the initial daughter contribution was assumed to be negligibly small relative to the radiogenic component. This approach works well in specific cases — the uranium-lead system in zircon, for example, benefits from zircon's strong exclusion of lead during crystallisation — but it is inapplicable to most rock-forming minerals, which incorporate both parent and daughter elements at the time of formation.2, 3 The isochron method provides a general solution to this problem that is applicable to any decay system and any suite of cogenetic samples, regardless of the initial daughter content.
The isochron equation
The isochron equation is derived directly from the basic decay equation by dividing both sides by a stable, non-radiogenic isotope of the daughter element, which serves as a normalising reference. In the rubidium-strontium system, for example, the decay of 87Rb to 87Sr (half-life 48.8 billion years) is normalised to the stable isotope 86Sr, yielding:2, 17
(87Sr/86Sr)now = (87Sr/86Sr)initial + (87Rb/86Sr)now × (eλt − 1)
This equation has the form of a straight line, y = b + mx, where y is the measured 87Sr/86Sr ratio, x is the measured 87Rb/86Sr ratio, b is the initial 87Sr/86Sr ratio (the y-intercept), and m is the slope (eλt − 1), from which the age t can be calculated directly.1, 2 The critical insight is that a suite of cogenetic samples will share the same age t and the same initial isotopic ratio (87Sr/86Sr)initial, but will differ in their 87Rb/86Sr ratios owing to mineralogical or compositional variation. Minerals and rocks with higher rubidium-to-strontium ratios will have accumulated more radiogenic 87Sr over the same time interval, and therefore will plot at higher positions along a line of constant slope and intercept.2, 3
At the time of formation (t = 0), the slope (eλt − 1) equals zero, and all cogenetic samples plot as a horizontal line at the initial isotopic ratio, regardless of their parent-to-daughter ratios. As time passes and radioactive decay proceeds, samples with higher parent isotope concentrations accumulate radiogenic daughter faster, and the array of data points rotates upward around the y-intercept, with the slope increasing as a monotonic function of age.2, 18 At any given time, all samples lie on a straight line — the isochron, from the Greek isos (equal) and chronos (time) — whose slope encodes the age and whose intercept preserves the initial isotopic ratio.
Rubidium-strontium: the classic isochron system
The rubidium-strontium (Rb-Sr) system is historically the most important and widely applied isochron dating method, and it was in this system that the isochron concept was first developed and formalised.1, 2 Rubidium-87 undergoes beta decay to strontium-87 with a half-life of 48.8 billion years, a value adopted by international convention.17 Because rubidium and strontium are both lithophile elements that are widely distributed in crustal rocks and minerals, and because their concentrations vary substantially among common rock-forming minerals (micas and potassium feldspars are rubidium-rich, plagioclase and apatite are strontium-rich), the Rb-Sr system is ideally suited to the isochron approach.
A typical Rb-Sr isochron analysis involves measuring the 87Sr/86Sr and 87Rb/86Sr ratios of several mineral separates or whole-rock samples from a single rock unit. Biotite, muscovite, and potassium feldspar have high Rb/Sr ratios and plot at the high-x end of the isochron diagram, while plagioclase and apatite have low Rb/Sr ratios and plot near the y-intercept, close to the initial 87Sr/86Sr ratio.2, 18 The greater the spread in Rb/Sr ratios among the analysed samples, the more precisely the slope (and therefore the age) can be determined.
Papanastassiou and Wasserburg demonstrated the power of the Rb-Sr isochron method in their 1969 study of basaltic achondrite meteorites, in which samples with a total spread of only 0.2 percent in 87Sr/86Sr defined a well-constrained isochron with an age of 4.39 ± 0.26 billion years and an initial 87Sr/86Sr ratio of 0.698976 ± 0.000055, one of the most precisely determined initial ratios for any meteorite class and a fundamental parameter for understanding the early differentiation of the Solar System.8 This initial ratio, known as BABI (Basaltic Achondrite Best Initial), serves as a reference point for strontium isotope geochemistry, representing the strontium isotopic composition of the Solar System at the time of planetary differentiation.2, 8
Isochrons in other decay systems
The isochron approach is not limited to the Rb-Sr system. It has been applied to every long-lived radioactive decay system used in geochronology, with each system offering distinct advantages depending on the geological context and the materials being dated.
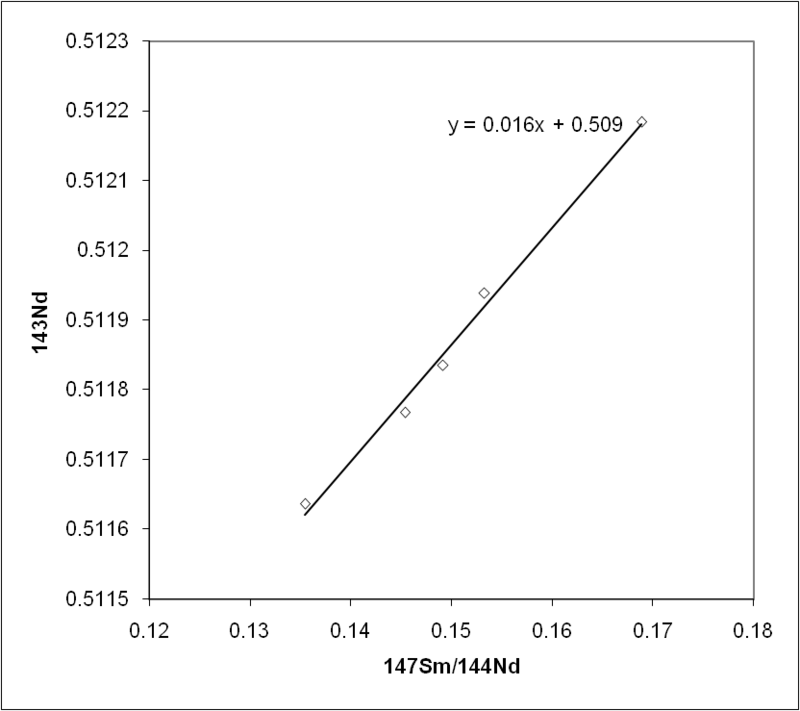
The samarium-neodymium (Sm-Nd) system is based on the alpha decay of 147Sm to 143Nd (half-life 106 billion years), normalised to the stable reference isotope 144Nd. DePaolo and Wasserburg pioneered the application of Sm-Nd isotope systematics to terrestrial rocks in 1976, demonstrating that variations in 143Nd/144Nd among mantle-derived rocks recorded the long-term chemical evolution of the Earth's mantle.11 Because samarium and neodymium are both rare-earth elements with very similar geochemical behaviour, the Sm/Nd ratio is much less susceptible to disturbance by metamorphism and weathering than the Rb/Sr ratio, making the Sm-Nd isochron method particularly valuable for dating rocks that have experienced significant post-crystallisation alteration.2, 11 The tradeoff is that the limited fractionation of Sm from Nd in most geological processes means that Sm-Nd isochrons typically have small spreads in the x-axis variable, reducing age precision compared to Rb-Sr isochrons from the same rocks.
The lutetium-hafnium (Lu-Hf) system exploits the beta decay of 176Lu to 176Hf (half-life approximately 37.1 billion years), normalised to 177Hf. Patchett and Tatsumoto obtained the first Lu-Hf isochron in 1980 by analysing a suite of eucrite meteorites, demonstrating that the system could provide independent chronological information complementary to the Rb-Sr and Sm-Nd methods.12 Like Sm-Nd, the Lu-Hf system is relatively resistant to metamorphic resetting because lutetium and hafnium are both high-field-strength elements with limited mobility in crustal fluids, and Lu-Hf isochrons have become an important tool for dating garnet-bearing metamorphic rocks and constraining the timing of mantle depletion events.2, 12
The rhenium-osmium (Re-Os) system is based on the beta decay of 187Re to 187Os (half-life 41.6 billion years), normalised to 188Os. This system is uniquely valuable because rhenium and osmium are strongly partitioned into sulphide minerals and metallic phases, making Re-Os isochrons the method of choice for dating iron meteorites, komatiites, mantle peridotites, and ore deposits — materials that are largely inaccessible to the lithophile decay systems.13 The very large fractionation of Re from Os that occurs during mantle melting and sulphide segregation produces wide spreads in 187Re/188Os among cogenetic samples, enabling precise isochron ages even from relatively small sample suites.13
Major isochron decay systems in geochronology2, 17
| System | Parent → Daughter | Half-life (Ga) | Reference isotope | Primary applications |
|---|---|---|---|---|
| Rb-Sr | 87Rb → 87Sr | 48.8 | 86Sr | Igneous & metamorphic rocks, meteorites |
| Sm-Nd | 147Sm → 143Nd | 106 | 144Nd | Mafic rocks, mantle evolution |
| Lu-Hf | 176Lu → 176Hf | 37.1 | 177Hf | Garnet-bearing rocks, crustal evolution |
| Re-Os | 187Re → 187Os | 41.6 | 188Os | Iron meteorites, komatiites, ore deposits |
| Pb-Pb | 235U → 207Pb | 0.704 / 4.47 | 204Pb | Meteorites, ancient crustal rocks |
Pb-Pb isochrons and the age of the Earth
Lead-lead (Pb-Pb) isochrons occupy a special place in geochronology because they do not require measurement of a parent element concentration. In the uranium-lead system, both 238U (half-life 4.468 billion years) and 235U (half-life 703.8 million years) decay to different lead isotopes (206Pb and 207Pb, respectively). If the two standard decay equations are divided by one another after normalising to the non-radiogenic reference isotope 204Pb, the uranium concentrations cancel out, and the resulting equation relates the present-day 207Pb/204Pb ratio to the 206Pb/204Pb ratio as a function of time only.2, 10 A suite of cogenetic samples with different U/Pb ratios will define a line in 207Pb/204Pb versus 206Pb/204Pb space whose slope is determined solely by the age, independent of the uranium content of the individual samples.
This property makes Pb-Pb isochrons exceptionally powerful for dating very old materials and materials in which uranium has been partially or completely lost, because the lead isotopic ratios preserve the age information even after the parent element has been removed. It also means that a Pb-Pb age can be determined from a single phase that contains lead but no measurable uranium, provided it can be combined with other cogenetic phases spanning a range of original U/Pb ratios.2, 9
The most consequential Pb-Pb isochron ever constructed was published by Clair Patterson in 1956. Patterson measured lead isotope ratios in five meteorites — three stone meteorites and two iron meteorites — and plotted them in 207Pb/204Pb versus 206Pb/204Pb space. The iron meteorite troilite (iron sulphide) from the Canyon Diablo meteorite, which contains negligible uranium, provided the initial lead isotopic composition of the Solar System (the y-intercept of the isochron), while the stone meteorites, which had accumulated radiogenic lead from uranium decay over the history of the Solar System, defined the high end of the array.9 The five points fell on a well-defined isochron with a slope corresponding to an age of 4.55 ± 0.07 billion years.9, 10
Patterson further demonstrated that a sample of modern deep-ocean sediment, taken as representative of the average lead isotopic composition of the Earth's surface, plotted on the same isochron as the meteorites. This observation established that the Earth and the meteorite parent bodies formed at the same time and from the same reservoir of primordial material, and the resulting age — refined to 4.55 billion years by subsequent work — remains the accepted age of the Earth.9, 10 The isochron passing through the primordial lead composition (as sampled by Canyon Diablo troilite) and the modern terrestrial lead composition is known as the Geochron, and it represents the locus of all lead isotopic compositions that could have evolved in a single-stage closed system over 4.55 billion years.10, 16
The two-stage terrestrial lead evolution model of Stacey and Kramers (1975) extended Patterson's work by demonstrating that the lead isotopic composition of modern terrestrial rocks is better explained by a two-stage history in which the bulk Earth's U/Pb ratio changed at approximately 3.7 billion years ago, possibly reflecting the onset of major crust-mantle differentiation.16 This model provides the standard framework for correcting common lead contamination in U-Pb geochronology and remains one of the most widely cited papers in isotope geology.
Internal versus whole-rock isochrons
Isochrons can be constructed from two fundamentally different types of sample suites, each providing different geological information. An internal isochron (also called a mineral isochron) is constructed from different mineral phases separated from a single rock specimen. Because the minerals crystallised from the same melt or recrystallised during the same metamorphic event, they share the same age and the same initial isotopic ratio, satisfying the cogenetic requirement of the isochron method. The spread in parent-to-daughter ratios is provided by the contrasting partition coefficients of the parent and daughter elements among the different mineral phases.2, 3
Internal isochrons typically date the last event at which the minerals achieved isotopic equilibrium, which may be the time of igneous crystallisation or, in metamorphosed rocks, the time at which the rock was heated above the closure temperatures of the constituent minerals. If a rock has experienced a later thermal event that partially re-equilibrated the isotopic compositions of its minerals, the internal isochron will record the age of the metamorphic resetting rather than the original crystallisation age.2, 18
A whole-rock isochron is constructed from multiple whole-rock samples taken from the same geological unit, such as different lava flows from a single volcanic sequence or different samples from a plutonic body. The spread in parent-to-daughter ratios arises from bulk compositional variation within the unit rather than from inter-mineral partitioning. Because the diffusion distances required for isotopic homogenisation at the whole-rock scale are vastly larger than at the mineral scale, whole-rock isochrons are far more resistant to metamorphic resetting than internal isochrons. A regional metamorphic event that completely re-equilibrates strontium isotopes among minerals within individual rock samples (resetting the internal isochron) may leave the whole-rock compositions essentially unchanged, preserving the original crystallisation age on the whole-rock isochron.2, 3
This difference carries an important caveat. The resilience of whole-rock isochrons to metamorphism depends on the assumption that strontium (or neodymium, or whatever daughter element is under consideration) was not exchanged between different whole-rock samples on a scale larger than the individual sample. If metasomatic fluids or high-temperature diffusion achieved isotopic re-equilibration between adjacent rocks at the outcrop or regional scale, the whole-rock isochron will also be reset, and may yield a mixed or meaningless age. Several well-documented cases of geologically meaningless whole-rock Rb-Sr isochrons have been reported in the literature, typically from regions that experienced pervasive fluid-rock interaction or granulite-facies metamorphism.2, 3
Why isochrons are self-checking
One of the most important properties of the isochron method is that it is intrinsically self-checking: a successful isochron simultaneously tests for both of the key assumptions underlying radiometric dating — closed-system behaviour and initial isotopic homogeneity — without requiring any external validation.2, 3
The closed-system assumption requires that no parent or daughter isotope has entered or left the system since the time of formation, except through radioactive decay. If one or more samples in a suite have experienced open-system behaviour — for example, rubidium loss through weathering, or strontium gain through fluid infiltration — those disturbed samples will no longer plot on the same straight line as the undisturbed samples. The resulting scatter is detectable by statistical analysis of the fit of the data to a linear model, and a suite of samples that shows excess scatter beyond analytical uncertainty does not yield a valid isochron age.6, 7
The assumption of initial isotopic homogeneity requires that all samples in the suite had the same daughter-isotope ratio at the time of formation. If the magma from which a set of rocks crystallised was isotopically heterogeneous — for example, because it incorporated material from isotopically distinct sources without fully mixing — the samples will not share a common initial ratio and will scatter off the isochron line. Like open-system disturbance, this heterogeneity is detectable through the same statistical tests applied to the regression.2, 6
The ability of the isochron method to flag its own failures is what distinguishes it from model-age approaches, where violations of the underlying assumptions simply produce incorrect ages without any internal warning. A model age calculated from a single sample will return a number regardless of whether the system has remained closed, whereas an isochron that displays excess scatter immediately alerts the geochronologist that something has gone wrong and that the resulting age should not be trusted. This self-checking property is a principal reason why isochron dating remains the standard approach in geochronology for decay systems that lack the dual-chronometer redundancy of the U-Pb concordia system.2, 3, 7
MSWD and statistical assessment of isochron quality
The primary statistical tool for evaluating whether a set of data points defines a true isochron is the mean square weighted deviation (MSWD), also known as the reduced chi-squared statistic. The MSWD compares the observed scatter of data points about the best-fit regression line to the scatter expected from the assigned analytical uncertainties alone. If the data are well described by a straight line and the analytical uncertainties are correctly estimated, the MSWD should equal approximately 1.0.6, 7
Wendt and Carl (1991) derived the probability distribution of the MSWD and showed that its expectation value is 1 for all values of the degrees of freedom f (equal to the number of data points minus two, for a two-parameter fit), with a standard deviation of ±(2/f)1/2.6 For a dataset with many points, the acceptable range of MSWD narrows around 1.0, while for small datasets the tolerable range is broader. As a practical guideline, an MSWD value significantly greater than 1 indicates that the scatter exceeds what can be explained by analytical errors alone, suggesting either open-system behaviour, initial isotopic heterogeneity, or underestimated analytical uncertainties. An MSWD significantly less than 1 suggests that the analytical uncertainties have been overestimated.6
Acceptable MSWD ranges as a function of sample size6
A dataset whose MSWD falls within the acceptable range at a chosen confidence level (typically 95 percent) is classified as a true isochron, indicating that the linear relationship is statistically robust and the age and initial ratio can be interpreted with confidence. A dataset whose MSWD exceeds the upper bound is classified as an errorchron — a term coined to describe a linear array that shows excess scatter, indicating either geological disturbance or unrecognised analytical problems.6, 7 An errorchron may still approximate the true age if the disturbance was minor, but the age and its uncertainty must be interpreted with caution. In classical practice, when the MSWD exceeds the acceptable range, the analytical uncertainties are sometimes expanded by multiplying them by the square root of the MSWD, a procedure that increases the reported age uncertainty to account for the observed overdispersion but does not explain its geological cause.6, 15
Regression methods and computational approaches
The statistical regression of isochron data — fitting a straight line to data points with uncertainties in both the x and y variables — is a more challenging problem than ordinary least-squares regression, which assumes that one variable is measured without error. The foundational treatment for isochron regression was provided by McIntyre, Brooks, Compston, and Turek in 1966, who developed a weighted regression method specifically for Rb-Sr isochron data that accounted for the proportional relationship between the analytical variance in 87Rb/86Sr and its magnitude.7
In 1969, Derek York published a general least-squares algorithm for fitting a straight line when both variables have uncertainties and those uncertainties may be correlated.4 York's method became the standard regression approach in isotope geochronology for the next three decades, implemented in widely used software packages such as Isoplot. The original algorithm was refined in several subsequent papers, culminating in York and colleagues' 2004 publication of "unified equations" that provided simplified, equivalent expressions for the slope, intercept, and their standard errors.5
An alternative statistical framework, based on maximum likelihood estimation, was introduced by Titterington and Halliday in 1979.19 The maximum likelihood approach offers several advantages over the York regression, particularly in its ability to handle overdispersed data (errorchrons) in a statistically rigorous manner. Rather than simply inflating the reported uncertainties to account for excess scatter, a maximum likelihood model can incorporate an additional dispersion parameter that quantifies the magnitude of the geological or unrecognised analytical scatter, providing a more physically meaningful treatment of the overdispersion.15, 19
Modern isochron calculations are performed using dedicated software tools. Ludwig's Isoplot, an Excel-based add-in first released in the 1990s, became the de facto standard for geochronological data reduction and visualisation for over two decades.14 More recently, Vermeesch's IsoplotR, a free and open-source toolbox written in R and JavaScript, has emerged as its successor, implementing York regression, maximum likelihood estimation, and Bayesian approaches for isochron fitting across all major decay systems including Rb-Sr, Sm-Nd, Lu-Hf, Re-Os, Pb-Pb, and K-Ca.14 Vermeesch (2024) further extended the IsoplotR framework to handle errorchrons through a maximum likelihood algorithm that accommodates overdispersion, and to fit anchored isochrons in which the slope or intercept is constrained by external information, addressing a long-standing limitation of the York method.15
A separate line of methodological development has explored the use of mixture models for interpreting complex isochron data that may represent the superposition of multiple geological events. Davies (2018) demonstrated that Bayesian mixture models can deconvolve isochron arrays that result from the mixing of two or more age populations, extracting individual component ages from datasets that would yield meaningless errorchrons under conventional single-isochron fitting.20
Historical development
The intellectual foundations of isochron dating were laid in the 1950s and 1960s, during a period of rapid growth in isotope geochemistry driven by improvements in thermal ionisation mass spectrometry and the availability of isotopically enriched spike solutions for isotope dilution analysis.2, 10
Clair Patterson's 1956 meteorite Pb-Pb isochron, which yielded the first reliable determination of the age of the Earth, was conceptually an isochron analysis avant la lettre: Patterson recognised that cogenetic meteorite samples with different U/Pb ratios could be used to define a linear array in lead isotope space whose slope gave the age of the Solar System without assuming an initial lead composition.9 However, the formal articulation of the isochron method as a general approach to radiometric dating came five years later, when Nicolaysen (1961) published his paper on the graphic interpretation of discordant age measurements, demonstrating that suites of cogenetic rock samples with variable Rb/Sr ratios defined straight lines in 87Sr/86Sr versus 87Rb/86Sr space whose slopes gave the age of the rock and whose intercepts gave the initial strontium isotopic composition.1
The 1960s saw the rapid application of the Rb-Sr isochron method to a wide range of geological problems, accompanied by the development of the statistical framework needed to assess the quality of isochron fits. McIntyre and colleagues (1966) published the first rigorous treatment of error propagation and weighted regression for Rb-Sr isochrons, establishing the statistical tools that would be refined by subsequent workers.7 By the late 1960s, Papanastassiou and Wasserburg had demonstrated the extraordinary precision achievable with the Rb-Sr isochron method through their studies of meteorites and lunar samples, obtaining initial 87Sr/86Sr ratios with uncertainties of a few parts per hundred thousand.8
The 1970s brought the extension of the isochron concept to new decay systems. DePaolo and Wasserburg (1976) established the Sm-Nd system as an independent geochronological tool, demonstrating that terrestrial rocks defined Sm-Nd isochrons consistent with their Rb-Sr ages while also preserving information about the long-term chemical evolution of the mantle.11 The Lu-Hf system followed in 1980, when Patchett and Tatsumoto published the first Lu-Hf whole-rock isochron for eucrite meteorites.12 The Re-Os system, developed primarily in the 1980s and 1990s, opened the siderophile and chalcophile geochemistry of the mantle and core to isochron dating, complementing the lithophile decay systems that dominated earlier work.13
Throughout these developments, advances in mass spectrometric precision, clean-laboratory chemistry, and computational tools have continuously improved the resolution and reliability of isochron ages. The transition from manually plotted isochron diagrams to computer-assisted regression, beginning with the McIntyre algorithm and progressing through York's method to modern maximum likelihood and Bayesian approaches, has been an integral part of this story, ensuring that the statistical assessment of isochron quality keeps pace with the analytical precision of the measurements.4, 5, 14, 15
Limitations and potential pitfalls
Despite its self-checking properties, the isochron method is not immune to systematic errors and geological complications. Several well-characterised pitfalls must be considered when interpreting isochron results.
Partial resetting occurs when a thermal or metasomatic event partially re-equilibrates the isotopic compositions of the samples without achieving complete homogenisation. In such cases, the data may still define a statistically acceptable linear array, but the slope of the resulting isochron will correspond to neither the original crystallisation age nor the age of the disturbing event, yielding a geologically meaningless hybrid age. Partial resetting is particularly problematic for whole-rock Rb-Sr isochrons from regions that have experienced moderate-grade metamorphism, where the strontium isotopes may be sufficiently mobile to reduce the scatter below the detection threshold of the MSWD test while still systematically biasing the slope.2, 3
Mixing lines can mimic isochrons in certain geological settings. If two isotopically distinct end-member compositions mix in variable proportions, the resulting samples will define a straight line in isotope-ratio space that is indistinguishable on purely geometric grounds from a true isochron. Distinguishing a mixing line from an isochron requires independent geological or geochemical evidence, such as major-element compositional trends, trace-element patterns, or isotopic data from a second independent decay system that should yield the same age if the linear array is a true isochron but a different age if it is a mixing line.2, 3 Cross-validation with multiple isotopic systems, as discussed in the context of radiometric cross-validation, is the most powerful method for resolving this ambiguity.
Decay constant uncertainties propagate directly into the absolute ages calculated from isochron slopes. The decay constants for the major long-lived radioactive isotopes were established by the IUGS Subcommission on Geochronology in 1977 and have not been officially revised since, although ongoing experimental work has identified discrepancies at the percent level for several systems, particularly 176Lu and 87Rb.17 These systematic uncertainties set a floor on the absolute accuracy of isochron ages that cannot be reduced by increasing analytical precision alone, and they are a significant concern when ages from different decay systems are compared at high precision.2, 17
Violation of the cogenetic assumption is perhaps the most fundamental pitfall. If the samples included in an isochron analysis did not actually form at the same time from the same isotopically homogeneous source, the isochron equation does not apply, and any apparent linear array is coincidental or reflects mixing rather than age. Careful geological context, field relationships, and petrographic characterisation are essential prerequisites for meaningful isochron dating, and no amount of statistical sophistication can substitute for sound geological understanding of the samples being analysed.2, 3, 20
References
Unified equations for the slope, intercept, and standard errors of the best straight line
Initial strontium isotopic abundances and the resolution of small time differences in the formation of planetary objects
Subcommission on geochronology: convention on the use of decay constants in geo- and cosmochronology