Overview
- Rubidium-strontium dating exploits the beta decay of rubidium-87 to strontium-87, with a half-life of 49.61 billion years, making it one of the most important radiometric systems for dating ancient igneous and metamorphic rocks and for tracing the geochemical evolution of the Earth's crust and mantle.
- The isochron method, in which multiple co-genetic samples are plotted as 87Sr/86Sr versus 87Rb/86Sr, yields both the age from the slope of the regression line and the initial 87Sr/86Sr ratio from the y-intercept, providing information about the source rock's prior history without requiring assumptions about the initial daughter isotope abundance.
- Strontium isotope stratigraphy, based on the secular variation of seawater 87Sr/86Sr preserved in marine carbonates, has become a powerful tool for dating and correlating Phanerozoic sedimentary sequences with a temporal resolution that in some intervals rivals biostratigraphy.
Rubidium-strontium (Rb-Sr) dating is a radiometric method that exploits the radioactive decay of rubidium-87 (87Rb) to strontium-87 (87Sr) by beta emission. With a half-life of approximately 49.61 billion years — more than ten times the age of the Earth — the 87Rb-87Sr system is ideally suited to dating ancient igneous and metamorphic rocks, tracing the geochemical evolution of the crust and mantle, and establishing the initial isotopic composition of the Solar System.1, 3 Developed as a practical geochronological tool in the late 1950s and 1960s, the Rb-Sr method played a pivotal role in establishing the antiquity of Precambrian terranes and in demonstrating that the continental crust has grown progressively through geological time.14, 15 Although it has been partly supplanted by the uranium-lead and samarium-neodymium systems for high-precision geochronology, the Rb-Sr method remains widely used for dating metamorphic events, determining provenance, and as the basis for strontium isotope stratigraphy in marine sediments.
The method's unique strength lies in the isochron technique, which allows simultaneous determination of both the age of a rock and the initial 87Sr/86Sr ratio of its source — a parameter that encodes information about the prior geological history of the material from which the rock was derived.3, 5
The rubidium-87 decay scheme
The physical basis of Rb-Sr dating rests on the radioactive decay of 87Rb, one of two naturally occurring isotopes of rubidium (the other being stable 85Rb). Rubidium-87 constitutes approximately 27.83 percent of natural rubidium and decays by emission of a beta particle (electron) and an antineutrino to produce 87Sr, a stable isotope of strontium.1, 3 The decay is a single-step process with no intermediate daughter products, in contrast to the complex multi-step decay chains of the uranium-lead system. This simplicity is an analytical advantage: every atom of 87Rb that decays produces exactly one atom of 87Sr, and the age equation relates the present-day measured isotopic ratios directly to the elapsed time without the need to account for intermediate nuclides or secular equilibrium.
The half-life of 87Rb has been the subject of extensive experimental study over more than half a century. The value recommended by the International Union of Pure and Applied Chemistry (IUPAC) and the International Union of Geological Sciences (IUGS) is 49.61 ± 0.16 billion years, corresponding to a decay constant (λ87) of 1.3972 × 10−11 per year.1 This supersedes the earlier value of 48.8 billion years (λ = 1.42 × 10−11 a−1) recommended by Steiger and Jäger in 1977, which had been the international standard for three decades.2 The revision, based on a comprehensive evaluation of all published measurements including both direct activity counting and geological comparison experiments, resolved a long-standing discrepancy between the Rb-Sr and U-Pb age scales for the same rocks.1
The extremely long half-life of 87Rb means that even in rocks billions of years old, only a small fraction of the original rubidium has decayed. This has two important consequences. First, the method is insensitive to young materials: measurable quantities of radiogenic 87Sr accumulate only over tens of millions of years in minerals with high Rb/Sr ratios, so the practical lower limit of the method is typically 10 to 50 million years for mineral dating, though whole-rock analyses of rapidly spreading oceanic basalts have pushed this limit lower in favourable cases.3, 9 Second, for ancient rocks the changes in the 87Sr/86Sr ratio are large enough to be measured with high precision by modern thermal ionisation mass spectrometry (TIMS), which achieves reproducibilities of ±0.00002 or better on the 87Sr/86Sr ratio.3, 4
The isochron method
The isochron technique is the cornerstone of Rb-Sr geochronology and one of the most elegant analytical tools in isotope geology. It was introduced by Nicolaysen in 1961 and refined by Compston and Pidgeon in 1962, and its logic has since been adopted across virtually all long-lived radiometric systems.5, 6
The fundamental age equation for the Rb-Sr system is:
(87Sr/86Sr)measured = (87Sr/86Sr)initial + (87Rb/86Sr)measured × (eλt − 1)
where λ is the decay constant of 87Rb, t is the age, and 86Sr is the stable, non-radiogenic reference isotope used to normalise both ratios.3, 5 This equation has the form of a straight line (y = b + mx), where the y-axis is 87Sr/86Sr, the x-axis is 87Rb/86Sr, the y-intercept is the initial ratio (87Sr/86Sr)initial, and the slope m = (eλt − 1) is a direct function of the age.
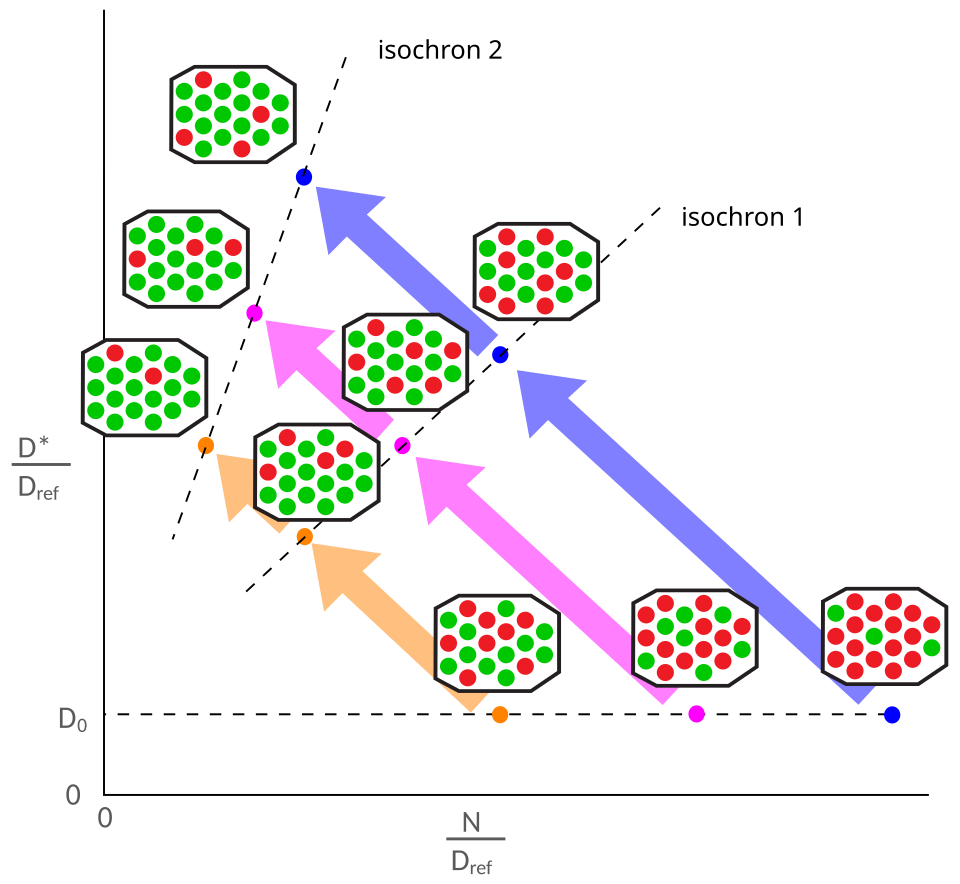
In practice, multiple co-genetic samples — either different minerals separated from a single rock (a mineral isochron) or multiple whole-rock samples from a genetically related igneous suite (a whole-rock isochron) — are analysed for their 87Rb/86Sr and 87Sr/86Sr ratios and plotted on an isochron diagram. If all samples crystallised at the same time from a reservoir with a homogeneous 87Sr/86Sr ratio, they will have different 87Rb/86Sr ratios (because different minerals incorporate different proportions of rubidium and strontium) but identical initial 87Sr/86Sr ratios. At the time of crystallisation, all samples plot along a horizontal line at the initial ratio. With the passage of time, each sample's 87Sr/86Sr ratio increases in proportion to its 87Rb/86Sr ratio, causing the data points to rotate upward around the y-intercept, defining a line of progressively steeper slope.3, 5
The power of the isochron method is that it requires no prior knowledge of the initial 87Sr/86Sr ratio: both the age and the initial ratio are determined simultaneously from the regression of the data. Furthermore, the degree to which the data points conform to a straight line provides a built-in test of the closed-system assumption. If any sample has gained or lost rubidium or strontium after crystallisation, it will plot off the isochron, and the scatter in the data provides a diagnostic warning that the system has been disturbed.3, 9
The initial ratio and source history
The y-intercept of a Rb-Sr isochron — the initial 87Sr/86Sr ratio — is far more than a mathematical parameter required for the age calculation. It is a geochemical fingerprint that records the integrated Rb/Sr history of the source material from which the dated rock was derived, and it has become one of the most important tracers in igneous and mantle geochemistry.3, 7
The concept is straightforward. A reservoir with a high Rb/Sr ratio accumulates radiogenic 87Sr rapidly, so any magma extracted from it will inherit a high initial 87Sr/86Sr ratio. Conversely, a reservoir with a low Rb/Sr ratio, such as the depleted upper mantle, evolves much more slowly and produces magmas with low initial ratios.3, 4 The initial ratio of a rock therefore reveals whether its source was the depleted mantle, enriched subcontinental lithospheric mantle, recycled continental crust, or some mixture of these reservoirs. This principle has been exploited extensively in studies of mantle heterogeneity, crustal recycling at subduction zones, and the petrogenesis of granites and other silicic magmas.
Mid-ocean ridge basalts (MORB), which sample the depleted upper mantle, consistently have initial 87Sr/86Sr ratios in the range of 0.7020 to 0.7035, reflecting the long-term depletion of rubidium relative to strontium caused by the preferential extraction of the large-ion lithophile element rubidium into the continental crust during partial melting events over billions of years.3, 13 Ocean island basalts, which tap deeper and more heterogeneous mantle sources, show a wider range of initial ratios, from values similar to MORB to values as high as 0.707 or more, indicating the presence of recycled crustal material in their mantle source regions.3 Continental granites and other crustal melts typically exhibit initial ratios above 0.710, reflecting the high time-integrated Rb/Sr ratio of continental crustal rocks.4, 14
Initial 87Sr/86Sr ratios of major rock types3, 4, 13
The BABI and solar system initial ratio
The lowest possible initial 87Sr/86Sr ratio for any object in the Solar System is that which existed at the time of Solar System formation, before any significant decay of 87Rb had occurred. This primordial value was determined by Papanastassiou and Wasserburg in 1969 through the analysis of basaltic achondrite meteorites — a class of differentiated stony meteorites that formed very early in Solar System history and have extremely low Rb/Sr ratios, so that their present-day 87Sr/86Sr ratios have changed negligibly since formation.7
By constructing Rb-Sr isochrons for seven basaltic achondrites, Papanastassiou and Wasserburg determined an initial 87Sr/86Sr ratio of 0.69897 ± 0.00003, a value they termed BABI (Basaltic Achondrite Best Initial).7 BABI represents the strontium isotopic composition of the Solar System at approximately 4.56 billion years ago, and it serves as the fundamental reference point against which all subsequent 87Sr/86Sr evolution in terrestrial and meteoritic materials is measured.3, 7
The BABI value has profound implications. Any terrestrial rock with an initial 87Sr/86Sr ratio close to BABI must have been derived from a source that had a very low time-integrated Rb/Sr ratio — that is, from mantle material that had not been significantly enriched in rubidium by crustal recycling processes. The progressive departure of the mantle's 87Sr/86Sr from BABI over geological time provides a measure of the rate and extent of crust-mantle differentiation.3, 14 Moorbath (1975) used initial 87Sr/86Sr ratios of Precambrian igneous rocks to demonstrate that the continental crust has been extracted from the mantle progressively over Earth history, rather than being formed in a single early differentiation event.14
Suitable minerals and whole-rock analysis
The Rb-Sr method can be applied to any mineral or rock that contains measurable amounts of rubidium and strontium, but some materials are far better suited than others. The ideal chronometer is a mineral with a high Rb/Sr ratio, because the accumulation of radiogenic 87Sr is proportional to the amount of parent 87Rb present, and minerals with higher ratios develop more radiogenic strontium in a given time, yielding more precise ages.3, 4
Biotite, a common dark mica in granitic and metamorphic rocks, is one of the most widely dated minerals in the Rb-Sr system. It has Rb/Sr ratios typically in the range of 20 to 200, producing large enrichments in radiogenic 87Sr that are easily measured. However, biotite has a relatively low closure temperature for strontium diffusion, estimated at approximately 300 to 350 degrees Celsius, meaning that it is readily reset by thermal events and records cooling ages rather than crystallisation ages in rocks that have experienced post-magmatic heating.3, 8 Muscovite (white mica) has similarly high Rb/Sr ratios and a somewhat higher closure temperature of approximately 500 degrees Celsius, making it more resistant to thermal resetting than biotite.4, 8 Potassium feldspar (K-feldspar) incorporates rubidium because the Rb+ ion substitutes readily for K+ in the feldspar structure, but its Rb/Sr ratios are generally lower than those of micas, and its closure temperature is intermediate.4
Whole-rock analysis treats an entire rock sample as the system to be dated, rather than an individual mineral. Because a rock is a much larger and more internally complex system than a single mineral grain, whole-rock Rb-Sr isochrons are generally more resistant to thermal disturbance: even if individual minerals within the rock exchange strontium isotopes with one another during a metamorphic event (thereby resetting mineral isochrons), the bulk composition of the rock may remain unchanged as long as the system remains closed on the scale of the hand specimen or larger.3, 6 Whole-rock isochrons were the primary tool used in the 1960s and 1970s to date Precambrian terranes worldwide, and the method was instrumental in establishing the geological time scale for the Archean and Proterozoic eons.6, 14, 20
The success of a whole-rock isochron depends critically on the requirement that all samples had the same initial 87Sr/86Sr ratio and have remained closed systems since that time. Suites of co-magmatic volcanic or plutonic rocks, erupted or emplaced within a short time span from a well-mixed magma source, are ideal candidates. Suites of rocks with heterogeneous initial ratios, or rocks that have experienced metasomatic alteration involving the introduction or removal of rubidium or strontium by external fluids, will yield disturbed isochrons with excessive scatter.3, 9
Rb-Sr characteristics of commonly dated minerals3, 4, 8
| Mineral | Typical Rb/Sr | Closure temperature (°C) | Common rock types |
|---|---|---|---|
| Biotite | 20–200 | ~300–350 | Granites, gneisses, schists |
| Muscovite | 20–500 | ~500 | Granites, pegmatites, schists |
| K-feldspar | 1–20 | ~350–400 | Granites, syenites, gneisses |
| Whole rock | 0.1–10+ | ~600–700+ | Igneous and metamorphic suites |
Applications in geochronology and mantle evolution
The Rb-Sr method has been applied to an exceptionally wide range of geological problems over the past six decades. Its principal applications fall into three broad categories: dating igneous and metamorphic events, tracing crust-mantle evolution, and determining the provenance and thermal history of deformed rocks.
In geochronology, Rb-Sr mineral isochrons are routinely used to date the cooling of plutonic and metamorphic rocks through the closure temperature of the dated mineral. Because different minerals close to strontium diffusion at different temperatures, a suite of Rb-Sr mineral ages from a single rock can define a cooling path through the temperature range of approximately 300 to 500 degrees Celsius, complementing higher-temperature chronometers such as U-Pb zircon and lower-temperature systems such as fission-track dating.3, 8 This multi-system approach to thermochronology has been used extensively to reconstruct the exhumation histories of mountain belts, including the Himalayas, the Alps, and the Appalachians.
The combined use of Rb-Sr and samarium-neodymium (Sm-Nd) isotope systematics revolutionised understanding of mantle geochemistry in the 1970s and 1980s. Because rubidium is more incompatible than strontium during mantle melting (meaning it preferentially enters the melt phase), whereas samarium is more compatible than neodymium, the two systems fractionate in opposite directions during crust-mantle differentiation. DePaolo and Wasserburg (1976) showed that the 143Nd/144Nd and 87Sr/86Sr ratios of mantle-derived rocks are inversely correlated, defining the so-called mantle array on a neodymium-strontium isotope correlation diagram.13 This relationship provides a powerful constraint on models of mantle convection, crustal recycling, and the chemical differentiation of the Earth. McCulloch and Wasserburg (1978) used combined Rb-Sr and Sm-Nd data to estimate the rate of continental crust extraction from the mantle over geological time, concluding that a substantial fraction of the present continental mass had been generated by the end of the Archean eon.17
Recent advances in laser ablation inductively coupled plasma tandem mass spectrometry (LA-ICP-MS/MS) have opened a new chapter for Rb-Sr geochronology. Rösel and Zack (2022) demonstrated that single laser spots on mica grains can yield geologically meaningful Rb-Sr isochron ages, allowing in situ dating of deformation fabrics and hydrothermal alteration zones in polished thin sections without the need for mineral separation or dissolution chemistry.18 Olierook and colleagues (2020) applied in situ Rb-Sr geochronology to resolve multiple generations of mica growth in an Archean gold deposit in Western Australia, demonstrating that the method can distinguish superimposed geological events separated by hundreds of millions of years within a single hand specimen.19
Strontium isotope stratigraphy
One of the most important applications of strontium isotope geochemistry lies outside traditional geochronology: the use of the seawater 87Sr/86Sr ratio as a stratigraphic and dating tool for marine sedimentary rocks. The oceans are a well-mixed reservoir with a residence time for strontium of approximately 2 to 4 million years, far longer than the ocean mixing time of roughly 1,000 years. As a result, the 87Sr/86Sr ratio of seawater is globally uniform at any given time, but it varies through geological time in response to changes in the relative fluxes of strontium from continental weathering (which contributes radiogenic strontium with high 87Sr/86Sr) and from hydrothermal alteration of oceanic crust at mid-ocean ridges (which contributes mantle-derived strontium with low 87Sr/86Sr).10, 12
Burke and colleagues (1982) published the first comprehensive curve of seawater 87Sr/86Sr variation through the Phanerozoic, based on the analysis of 786 marine carbonate, evaporite, and phosphate samples of known age.10 This curve revealed systematic oscillations between values as low as approximately 0.7068 during the Late Permian and Jurassic and as high as approximately 0.7092 at the present day, reflecting the changing balance of continental and mantle strontium inputs driven by variations in seafloor spreading rates, continental configuration, and mountain-building episodes.10, 12 Veizer (1989) extended the analysis to the Precambrian, showing that strontium isotope variations can be traced back at least two billion years, although the record becomes increasingly fragmentary and difficult to interpret in older rocks.12
The practical application of this secular variation is strontium isotope stratigraphy (SIS). If the 87Sr/86Sr ratio of a marine carbonate or phosphate fossil (such as a belemnite, brachiopod, or conodont) is measured, it can be compared with the global seawater curve to determine the numerical age of the sample. McArthur, Howarth, and Bailey (2001) constructed a statistically optimised LOWESS (locally weighted scatterplot smoothing) fit to the marine strontium isotope data for the interval 0 to 509 million years ago, providing a look-up table that converts measured 87Sr/86Sr ratios directly to numerical ages with uncertainties that, in the most favourable intervals (such as the Cenozoic and parts of the Cretaceous), are as low as ±0.5 to 1 million years.11 This precision rivals and in some cases exceeds that of biostratigraphy, and SIS has become an indispensable correlation tool in petroleum exploration, paleoceanography, and sequence stratigraphy.
The temporal resolution of SIS depends on the steepness of the seawater 87Sr/86Sr curve in the interval of interest. During periods when the ratio changed rapidly (such as the Cenozoic rise or the Late Permian decline), small differences in 87Sr/86Sr correspond to short time intervals, and the dating precision is high. During periods when the curve is flat or oscillating (such as parts of the Carboniferous), the same measurement uncertainty corresponds to a much larger age range, and SIS is less useful as a dating tool.11, 12
Limitations and open-system behaviour
The most significant limitation of the Rb-Sr method is the susceptibility of the system to open-system behaviour during metamorphism, hydrothermal alteration, and weathering. Both rubidium and strontium are relatively mobile alkali and alkaline-earth elements that can be transported by hot aqueous fluids, particularly those associated with metamorphic dehydration reactions and hydrothermal circulation. When external fluids introduce or remove rubidium or strontium, the isotopic systematics are disturbed and the resulting isochron may yield an age that has no simple geological meaning.3, 16
Lanphere and colleagues (1964) provided one of the earliest documented examples of large-scale Rb-Sr redistribution during metamorphism in the World Beater Complex of California, demonstrating that strontium isotopes were homogenised among minerals over distances of metres during amphibolite-facies metamorphism, completely resetting mineral isochrons while leaving whole-rock compositions relatively undisturbed.16 This observation established the critical distinction between mineral and whole-rock isochrons: mineral isochrons are reset at relatively low temperatures (300 to 500 degrees Celsius for micas) because strontium diffusion operates efficiently on the scale of individual grains, whereas whole-rock isochrons require strontium redistribution on a much larger scale and are therefore more resistant to thermal disturbance, though they too can be reset by pervasive fluid infiltration.3, 8
Paradoxically, the susceptibility of the Rb-Sr system to metamorphic resetting can be turned to advantage. Because biotite and muscovite are reset at well-characterised temperatures, their Rb-Sr ages record the time at which the host rock cooled through the mineral's closure temperature following the last thermal event. In polymetamorphic terranes, a whole-rock isochron may preserve the protolith age while mineral isochrons record the timing of the most recent metamorphism, providing a two-stage history from a single analytical dataset.3, 9
A further limitation is that the relatively small range of 87Rb/86Sr ratios available in some rock types can result in isochrons with short lever arms, reducing the precision of the age determination. Mafic and ultramafic rocks, which have low bulk Rb/Sr ratios, produce isochrons with gentle slopes that are particularly susceptible to analytical scatter. For such lithologies, the Sm-Nd system, in which the parent-daughter ratio is less strongly fractionated by magmatic processes but also less susceptible to disturbance, is generally preferred.3, 13
Historical significance
The Rb-Sr method holds an important place in the history of geochronology. Although the radioactive decay of 87Rb was recognised as a potential geological chronometer by Otto Hahn and colleagues in the late 1930s, and the first crude Rb-Sr age determinations were published in 1943, the method did not become a practical tool until the development of modern solid-source mass spectrometers in the 1950s, which allowed the isotopic composition of strontium to be measured with the precision required for meaningful geochronology.4, 15
The introduction of the isochron technique by Nicolaysen (1961) was a critical methodological advance.5 Prior to the isochron method, Rb-Sr ages had to be calculated assuming a value for the initial 87Sr/86Sr ratio, introducing a source of systematic error that was difficult to quantify. The isochron approach eliminated this assumption entirely and provided a graphical test of closed-system behaviour, transforming the Rb-Sr system from a semi-quantitative tool into a rigorous geochronometer. Compston and Pidgeon (1962) demonstrated that the isochron method could be applied to total-rock (whole-rock) samples, opening the way for the dating of sedimentary and metamorphic rocks that could not be dated by mineral methods.6
Throughout the 1960s and 1970s, whole-rock Rb-Sr isochron dating was the primary method used to establish the absolute chronology of the Precambrian. Moorbath and colleagues used the method extensively to date the Archean gneisses and supracrustal rocks of West Greenland, establishing ages of approximately 3.7 to 3.8 billion years for the Isua supracrustal belt and the Amîtsoq gneisses — at the time, the oldest known rocks on Earth.20 These results, combined with initial 87Sr/86Sr ratios close to the mantle evolution line, demonstrated that even in the early Archean, new continental crust was being generated from the mantle, a conclusion later reinforced by Sm-Nd isotope studies.14, 17
The method also played a central role in lunar geochronology. Papanastassiou and Wasserburg applied Rb-Sr isochron dating to Apollo mission samples, establishing the crystallisation ages of lunar basalts and determining that the Moon's surface was extensively resurfaced by volcanic activity between approximately 3.1 and 3.9 billion years ago.7, 15 The very low initial 87Sr/86Sr ratios of the oldest lunar rocks, close to BABI, confirmed that the Moon, like the Earth, formed from material with a primitive strontium isotopic composition approximately 4.5 billion years ago.7
Although the Rb-Sr method has been increasingly supplemented by U-Pb zircon geochronology for high-precision dating of igneous and metamorphic rocks, and by 40Ar/39Ar dating for thermochronological applications, it remains an active and evolving field. The development of in situ LA-ICP-MS/MS techniques has revitalised Rb-Sr dating by enabling spatially resolved analyses of mica and feldspar in thin sections, opening applications in structural geology, ore deposit characterisation, and sedimentary provenance studies that were previously impractical with conventional dissolution-based methods.18, 19
References
Subcommission on geochronology: convention on the use of decay constants in geo- and cosmochronology
Initial strontium isotopic abundances and the resolution of small time differences in the formation of planetary objects
Strontium isotope stratigraphy: LOWESS version 3: best fit to the marine Sr-isotope curve for 0–509 Ma and accompanying look-up table for deriving numerical age
Redistribution of strontium and rubidium isotopes during metamorphism, World Beater Complex, Panamint Range, California
Resolving multiple geological events using in situ Rb–Sr geochronology: implications for metallogenesis at Tropicana, Western Australia