Overview
- The human brain exhibits pronounced structural asymmetries, including a larger left planum temporale, a leftward bias in Broca's area, and left-dominant arcuate fasciculus connectivity, that underpin the overwhelming left-hemisphere specialization for language found in roughly 95% of right-handers and 70% of left-handers.
- Human handedness, at approximately 90% right-hand dominance compared to roughly 65–70% in chimpanzees, is detectable in the fossil record through cut mark directionality, dental striations, and flake scar patterns on stone tools, placing the emergence of strong population-level lateralization at least as early as Homo habilis some 1.8 million years ago.
- The language–tool co-evolution hypothesis proposes that language and complex tool manufacture share overlapping left-hemisphere neural substrates for hierarchical action sequencing, with neuroimaging studies of stone knapping activating Broca's area and adjacent premotor cortex, suggesting that selection pressures on one capacity may have driven elaboration of the other.
The human brain is not symmetrical. Although the two cerebral hemispheres appear broadly similar in external form, they differ substantially in their internal organization, their patterns of cortical folding, and the cognitive functions they preferentially support. In the vast majority of humans, the left hemisphere dominates for language — both production and comprehension — while the right hemisphere plays a greater role in spatial attention, prosody, and certain aspects of social cognition. This functional asymmetry, known as cerebral lateralization, is among the most distinctive features of the human brain, and its evolutionary origins are deeply intertwined with the emergence of language, tool use, and the population-level right-handedness that characterizes our species.1 Understanding when, why, and how lateralization evolved requires evidence from neuroanatomy, comparative primatology, paleoanthropology, and the archaeological record — lines of inquiry that converge on a picture of lateralization as an ancient feature of the primate brain that was dramatically amplified in the human lineage.
Cerebral asymmetry in modern humans
The structural foundation of language lateralization was first documented systematically by Norman Geschwind and Walter Levitsky in 1968, who examined 100 post-mortem human brains and found that the planum temporale — a triangular region of the superior temporal lobe that forms part of Wernicke's area, a key zone for language comprehension — was significantly larger on the left side in 65% of specimens, larger on the right in 11%, and approximately equal in 24%.3 This leftward asymmetry of the planum temporale has since been confirmed in numerous imaging studies and is now considered one of the most robust structural correlates of language lateralization in the human brain.3, 1
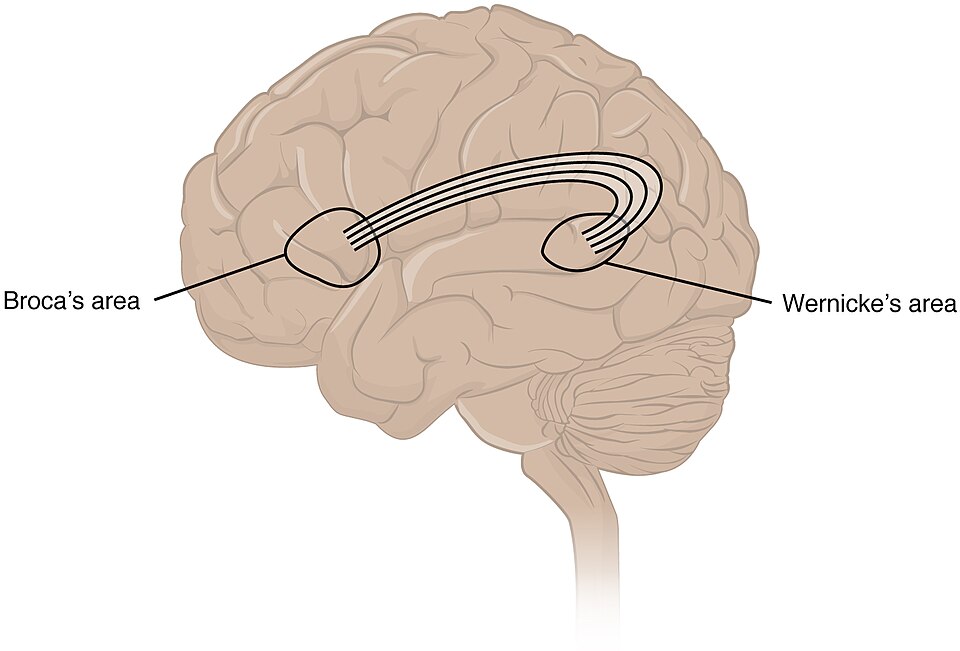
The asymmetry extends beyond the planum temporale. Broca's area, located in the inferior frontal gyrus of the left hemisphere and critical for speech production and syntactic processing, shows a leftward volumetric bias in most individuals, with the pars opercularis (Brodmann area 44) and pars triangularis (area 45) both tending to be larger on the left.1, 21 The arcuate fasciculus, the major white matter tract connecting Wernicke's area in the posterior temporal lobe with Broca's area in the frontal lobe, is substantially larger and more densely connected in the left hemisphere than the right. Catani and Mesulam documented that this leftward dominance of the arcuate fasciculus is a defining feature of the human language network, absent or greatly reduced in non-human primates, and that damage to it produces the conduction aphasias that helped establish the classical neurological model of language circuitry.21
At the gross morphological level, the human brain displays what is known as the Yakovlevian torque — a pattern in which the right frontal lobe protrudes slightly anterior to the left (right-frontal petalium) while the left occipital lobe extends slightly posterior to the right (left-occipital petalium). LeMay demonstrated this torque pattern in CT scans of living humans in the 1970s and showed that it correlates, imperfectly but consistently, with handedness and language dominance.2 Holloway and de Lacoste-Lareymondie subsequently identified the same pattern of petalial asymmetry on endocasts of fossil hominins, suggesting that the torque pattern — and, by inference, some degree of hemispheric specialization — extends deep into the hominin fossil record.22
Lateralization and handedness
Handedness is the most visible behavioral expression of cerebral lateralization. In modern human populations across all cultures studied, approximately 90% of individuals are right-handed — a ratio that has remained stable across historical periods and geographical regions.1, 4 Right-handedness is controlled primarily by the left hemisphere, the same hemisphere that dominates for language in most individuals, and the two asymmetries are widely thought to share underlying developmental and genetic mechanisms, though the precise nature of their linkage remains debated.1
Marian Annett's right-shift theory, first articulated in the 1970s and refined over subsequent decades, provides the most influential genetic model for the human handedness distribution. Annett proposed that a single gene (which she termed the "right shift" factor, or RS+) does not determine handedness directly but instead shifts the distribution of manual skill toward the right hand while simultaneously biasing language processing toward the left hemisphere.4 In the absence of the RS+ allele, handedness would be distributed randomly, with roughly equal numbers of left- and right-handers. The RS+ allele, present in most humans, shifts this distribution such that approximately 90% of the population favors the right hand. The theory elegantly accounts for the observation that left-handedness is never eliminated from the population (because random factors continue to operate even in RS+ carriers) and that left-handers show more variable patterns of language lateralization than right-handers.4
Population-level handedness across primates5, 6
The contrast between humans and other great apes is striking. While some non-human primates show population-level hand preferences for specific tasks, these preferences are notably weaker than in humans. William Hopkins and colleagues have demonstrated through decades of research on captive chimpanzees that approximately 65–70% show a right-hand preference for certain manual tasks such as bimanual feeding, tool use, and gestural communication, but this bias is task-dependent and far less consistent across contexts than human handedness.6, 7 Gorillas show a similar right-hand bias of roughly 65% for bimanual tasks, while bonobos and orangutans display weaker and more variable population-level preferences.6 The degree of lateralization in humans — 90% right-handed with high within-individual consistency across tasks — thus represents an amplification of a pre-existing primate tendency rather than a wholly novel feature.1, 6
Handedness in the fossil record
Determining handedness in extinct hominins requires indirect evidence preserved in stone tools, skeletal remains, and tooth wear. Nicholas Toth's pioneering 1985 analysis of Oldowan stone tools from Koobi Fora, Kenya, provided the first systematic approach. By examining the direction and pattern of flake scars on cores — the remnant marks left when flakes are struck from a stone — Toth demonstrated that the orientation of flake removals could distinguish right-handed from left-handed knappers. His analysis of early Pleistocene assemblages revealed a pattern consistent with predominantly right-handed tool production, suggesting that population-level right-handedness may have been present in Homo habilis or contemporaneous hominins as early as 1.8 million years ago.5

Dental striations have provided a complementary source of evidence. When hominins used their front teeth as a "third hand" to grip food items while cutting with a stone tool held in the dominant hand, the tool occasionally struck the labial surface of the anterior teeth, leaving characteristic oblique scratch marks. The orientation of these scratches — upper-left to lower-right in right-handers, the reverse in left-handers — preserves a record of hand preference. Lozano and colleagues analyzed dental striations in Neanderthal specimens and found that the overwhelming majority of individuals showed patterns consistent with right-handedness, with left-hander frequencies comparable to those in modern human populations at roughly 10%.20 This finding pushed robust evidence for the modern human handedness ratio back to at least the Middle Pleistocene.
Experimental stone knapping studies have further refined the archaeological evidence. Uomini and Meyer conducted a series of experiments in which skilled modern knappers produced Acheulean bifacial tools while undergoing functional transcranial Doppler ultrasonography, a technique that measures blood flow velocity in the cerebral arteries as a proxy for hemispheric activation. They found that Acheulean tool manufacture produced significant left-hemisphere activation — the same laterality pattern seen during language tasks — providing direct neurophysiological evidence that complex stone tool production and language share overlapping neural resources in the left hemisphere.19
Evolution of Broca’s area
Broca's area occupies the posterior portion of the inferior frontal gyrus in the left hemisphere and has been central to neurolinguistic theory since Paul Broca's observations in the 1860s that damage to this region impaired speech production. On hominin endocasts, the corresponding region sometimes leaves a visible surface impression — the so-called "Broca's cap" — that has been used to track changes in inferior frontal gyrus morphology across evolutionary time.22, 1
Ralph Holloway identified what he interpreted as humanlike configurations of the Broca's cap region on endocasts of early Homo, including KNM-ER 1470 (Homo rudolfensis, approximately 1.9 million years ago), arguing that the morphology of the inferior frontal gyrus in these specimens departed from the ape pattern and approached the modern human condition.22 The interpretation remains contested because the relationship between a surface prominence on an endocast and the underlying cytoarchitectonic boundaries of Brodmann areas 44 and 45 is imprecise. An endocast cannot reveal whether the cells in the region were organized in the columnar pattern characteristic of human Broca's area, nor whether the region had the dense connectivity with temporal language areas via the arcuate fasciculus that is a hallmark of the modern human language network.21
Comparative neuroanatomy has complicated the picture further. Hopkins and Cantalupo demonstrated that chimpanzees possess a homologue of Broca's area in the inferior frontal gyrus that shows a leftward volumetric asymmetry similar in direction, though smaller in magnitude, to that observed in humans.7 This finding suggests that the structural asymmetry of Broca's area predates the human–chimpanzee divergence and that what changed during human evolution was not the presence of the region per se but its internal organization, its connectivity, and the degree of its lateralization — features that are not visible on endocasts.
Lateralization in non-human primates
The discovery that cerebral lateralization is not unique to humans but has deep primate roots has reshaped theories of language evolution. Hopkins and colleagues' extensive research program on captive chimpanzees, spanning thousands of individuals across multiple facilities, has established that chimpanzees show population-level right-hand preferences for a range of manual tasks, with the strongest and most consistent biases observed during tasks involving bimanual coordination and tool use.6, 7 Critically, these hand preferences correlate with structural brain asymmetries: right-handed chimpanzees tend to have a larger left-hemisphere motor hand area in the precentral gyrus, mirroring the contralateral relationship between handedness and motor cortex asymmetry seen in humans.7
The planum temporale, the region whose leftward asymmetry Geschwind and Levitsky documented as a language correlate in humans, also shows a leftward bias in chimpanzees and other great apes, though the asymmetry is less pronounced and more variable than in humans.1, 6 The functional significance of this asymmetry in non-human primates is not yet fully understood, but it suggests that the neural substrate for left-hemisphere auditory processing specialization was present in the common ancestor of humans and African apes and was subsequently co-opted and elaborated for language in the human lineage.
Gestural communication in great apes provides a particularly important bridge to language evolution. Chimpanzees, bonobos, and gorillas all use intentional communicative gestures, and Hopkins's work has shown that captive chimpanzees preferentially gesture with the right hand during communicative interactions, implying left-hemisphere control of these communicative acts.6 This finding is significant because it suggests that left-hemisphere dominance for intentional, referential communication — a core feature of human language — may have its roots not in vocalization but in manual gesture, a possibility that is central to the gestural-origins theory of language evolution.
Language–tool co-evolution
One of the most consequential hypotheses in the study of language origins holds that language and complex tool manufacture co-evolved, driven by shared demands on the same left-hemisphere neural substrates. Patricia Greenfield articulated the foundational version of this hypothesis in 1991, drawing parallels between the hierarchical structure of language (words nested into phrases, phrases into clauses, clauses into sentences) and the hierarchical structure of complex manual actions (grasping embedded within assembling, assembling embedded within constructing).9 Greenfield proposed that both capacities depended on a common neural mechanism for combining elements into hierarchically organized sequences, and that Broca's area — known to be involved in both speech production and certain complex manual tasks — served as the shared substrate. On this view, selection for improved tool-making ability would have simultaneously enhanced the neural machinery available for language, and vice versa.9
Dietrich Stout and Thierry Chaminade provided critical neuroimaging evidence for this hypothesis in 2012. Using positron emission tomography (PET) to scan the brains of subjects as they produced stone tools of increasing complexity — from simple Oldowan flakes to complex Late Acheulean bifaces — they found that Acheulean tool manufacture activated a network of left-hemisphere regions that substantially overlapped with language-related areas, including the ventral premotor cortex, the inferior frontal gyrus (Broca's area), and the supplementary motor area.8 Crucially, this overlap was most pronounced for the most complex tool forms, those requiring extended sequences of hierarchically organized actions. Simple Oldowan flaking activated primarily dorsal premotor and sensorimotor regions without significant inferior frontal involvement, but the demanding bimanual, multi-step sequences required for Acheulean handaxe production recruited the left inferior frontal gyrus in a pattern indistinguishable from that activated during complex syntactic processing.8
Uomini and Meyer's 2013 study extended these findings by demonstrating that stone knapping and language production draw on the same neural resources in real time. When subjects performed a concurrent language task while knapping, both activities showed interference effects, and functional transcranial Doppler measurements revealed that both tasks produced overlapping patterns of left-hemisphere blood flow increase.19 This resource-sharing provides strong evidence that the co-evolution hypothesis is not merely a structural analogy but reflects genuine competition for shared neural circuitry.
The mirror neuron system and gestural origins
The discovery of mirror neurons in the ventral premotor cortex of macaque monkeys by Giacomo Rizzolatti and colleagues in the mid-1990s introduced a new dimension to theories of language evolution. Mirror neurons fire both when an animal performs a specific manual action and when it observes the same action performed by another individual.10 Rizzolatti and colleagues proposed that this mirror mechanism provided a neural basis for action understanding — the ability to recognize and interpret the goals and intentions behind observed actions — and noted that the mirror neuron region in macaques (area F5) is the likely homologue of Broca's area in humans.10
Michael Arbib developed the implications of this finding into a comprehensive theory of language evolution in a landmark 2005 paper. Arbib proposed a multi-stage evolutionary trajectory in which a mirror system for grasping, inherited from the common ancestor of humans and macaques, was gradually extended through a series of intermediate stages: first to imitation of complex manual actions, then to pantomime (communicating through iconic gestures), then to a conventional gestural protolanguage, and finally to spoken language with full syntax.11 On this account, language began not with vocalization but with manual gesture, and the left-hemisphere dominance of language reflects the left-hemisphere dominance of skilled manual action and its mirror-system underpinnings. The transition from gesture to speech would have occurred as vocal control came under increasing cortical regulation, eventually allowing the vocal channel to carry the communicative load that gestures had previously borne.11
The gestural-origins hypothesis is supported by several independent lines of evidence. Sign languages, which are fully expressive natural languages that use the manual-visual modality rather than the vocal-auditory modality, activate the same left-hemisphere regions as spoken languages, including Broca's area and Wernicke's area. Hickok, Bellugi, and Klima demonstrated through lesion studies and neuroimaging that deaf signers with left-hemisphere damage develop aphasias with the same symptom profiles as those seen in hearing speakers, while right-hemisphere damage impairs spatial cognition but leaves signing intact.18 This modality independence of left-hemisphere language lateralization is difficult to explain if language lateralization evolved specifically for vocal control, but follows naturally if the lateralization reflects a deeper left-hemisphere specialization for hierarchical, combinatorial, and sequential processing that is modality-independent.18, 1
When did language evolve?
The question of when language evolved is among the most contested in paleoanthropology, in part because language leaves no direct fossil trace. Researchers have therefore relied on a combination of anatomical, archaeological, and genetic proxies, none of which individually settles the question but which together constrain the range of plausible scenarios.
The vocal tract provides one anatomical line of evidence. Philip Lieberman argued on the basis of vocal tract reconstructions that Neanderthals and earlier hominins had a supralaryngeal vocal tract incapable of producing the full range of vowel contrasts found in modern human speech, particularly the vowel /i/, because the larynx was positioned higher in the throat than in modern Homo sapiens.12 Lieberman's reconstructions have been debated extensively, and more recent work suggests that the differences between Neanderthal and modern human vocal tracts may have been smaller than initially proposed, but the general principle that vocal tract anatomy constrains the acoustic repertoire of speech remains influential.12
The hyoid bone, a horseshoe-shaped bone in the throat that anchors muscles involved in swallowing and speech, provides complementary evidence. The Neanderthal hyoid from Kebara Cave, Israel, dated to approximately 60,000 years ago, is virtually identical in morphology to the modern human hyoid, suggesting that the muscular apparatus for fine vocal control was present in Neanderthals.14 The hypoglossal canal, which transmits the nerve controlling the tongue muscles essential for articulate speech, has been proposed as another anatomical proxy. Kay, Cartmill, and Balow reported in 1998 that the hypoglossal canal in modern humans is significantly larger relative to skull size than in chimpanzees and australopithecines, and that Homo specimens dating back at least 400,000 years show the enlarged modern condition.13 However, subsequent studies have questioned the reliability of hypoglossal canal size as a speech indicator due to high within-species variation.
Genetic evidence has focused on FOXP2, a transcription factor gene discovered in 2001 when mutations in it were found to cause severe speech and language disorders in the KE family, a large British family in which affected members suffered from verbal dyspraxia — an inability to coordinate the fine orofacial movements required for fluent speech.15 Enard and colleagues showed in 2002 that the human version of FOXP2 differs from the chimpanzee version by two amino acid substitutions that appear to have been driven by positive selection within the last 200,000 years, suggesting a recent episode of selection on a gene critical for speech motor control.16 However, Krause and colleagues subsequently demonstrated in 2007 that Neanderthals shared the modern human FOXP2 variant, pushing the selective sweep back to at least the common ancestor of humans and Neanderthals, roughly 500,000–800,000 years ago.17
Archaeological proxies provide additional constraints. The appearance of ochre use, perforated shell beads, and engraved geometric patterns at sites in Africa dating to 100,000–70,000 years ago is widely interpreted as evidence for symbolic cognition, and many researchers argue that symbolic behavior of this complexity implies language. Composite tool technologies, which require multiple components to be manufactured separately and then assembled according to a plan — hafted spear points, for example — appear in the Middle Stone Age of Africa by at least 200,000–300,000 years ago and require the kind of hierarchical planning and potentially teaching through language that the co-evolution hypothesis predicts.8 The consensus, insofar as one exists, is that the biological prerequisites for language — including the lateralized neural substrates documented by endocast evidence, the vocal anatomy indicated by hyoid and hypoglossal data, and the genetic foundations marked by FOXP2 — were assembled gradually over the course of Homo evolution, with fully modern language capacities likely present by the time Homo sapiens began producing unambiguous symbolic artifacts in Africa.
Modality independence of language lateralization
One of the most striking findings in the neuroscience of language is that left-hemisphere lateralization for linguistic processing is not specific to the auditory-vocal channel. The study of sign languages has been decisive in establishing this point. Deaf individuals who use American Sign Language (ASL) or other natural sign languages as their primary means of communication process sign language in the same left-hemisphere perisylvian regions — Broca's area, Wernicke's area, the arcuate fasciculus — that hearing individuals use for spoken language.18
Hickok, Bellugi, and Klima's research program on deaf signers with focal brain lesions provided the clearest evidence. Left-hemisphere damage in deaf signers produced sign-language aphasias that paralleled the classical aphasia syndromes of hearing speakers: anterior left-hemisphere lesions caused effortful, agrammatic signing (the sign-language analogue of Broca's aphasia), while posterior left-hemisphere lesions caused fluent but semantically empty signing (the sign-language analogue of Wernicke's aphasia). Right-hemisphere damage, by contrast, impaired visuospatial processing but left sign-language abilities intact — a remarkable dissociation given that sign language is produced and perceived in the spatial domain.18
This modality independence has profound implications for theories of language lateralization. If left-hemisphere dominance for language reflected an evolved specialization for auditory or vocal processing, one would expect sign language, which uses an entirely different input-output modality, to be lateralized differently. The fact that it is not suggests that the left hemisphere's role in language reflects a deeper computational specialization — for the hierarchical, rule-governed combination of discrete meaningful units into structured sequences — that operates regardless of whether those units are spoken words, manual signs, or written symbols.18, 1 This computational characterization aligns with the co-evolution hypothesis: the same left-hemisphere machinery that organizes complex manual action sequences for tool manufacture can, in principle, organize the combinatorial sequences that define language, whether those sequences are realized through the hands or the voice.
Synthesis
The evidence from neuroanatomy, comparative primatology, paleoanthropology, and genetics converges on a coherent, if still incomplete, account of how brain lateralization and language co-evolved in the human lineage. Cerebral asymmetry is not a uniquely human trait; the planum temporale asymmetry, Broca's area homologue, and population-level hand preferences documented in chimpanzees and other great apes indicate that the foundations of lateralization were present in the common ancestor of humans and African apes.6, 7 What changed during human evolution was the degree and consistency of lateralization: the shift from roughly 65–70% right-handedness in chimpanzees to 90% in humans, from modest planum temporale asymmetry to the pronounced leftward bias of the modern human language network, and from gestural communication aided by weak lateralization to full-blown language with robust left-hemisphere dominance.1, 3
The archaeological record suggests that this amplification unfolded gradually. Population-level right-handedness consistent with the modern ratio was present by at least the Middle Pleistocene, based on Neanderthal dental striations and Acheulean tool patterns.5, 20 Endocast evidence for petalial asymmetry and humanlike Broca's cap morphology extends back to early Homo roughly 1.9 million years ago.22 The co-evolution of language and tool use, mediated by shared left-hemisphere substrates for hierarchical action sequencing, provides a plausible selective mechanism: as tool technologies became more complex, requiring longer and more deeply nested action sequences, the same neural circuitry was simultaneously recruited for or co-opted by the emerging capacity for combinatorial, syntactic communication.8, 9, 19 The modality independence of language lateralization suggests that this co-evolutionary process was not specific to speech but reflected a general left-hemisphere computational specialization that could support language in any modality.18
Many questions remain open. The relative contributions of genetic drift, sexual selection, and direct selection on language ability to the amplification of lateralization are not well constrained. The degree to which Neanderthals and other archaic hominins possessed language remains debated, with the FOXP2 evidence suggesting shared genetic prerequisites but the archaeological record showing a less clear-cut pattern of symbolic behavior than in contemporaneous Homo sapiens.16, 17 The precise evolutionary relationship between the mirror neuron system and language remains a matter of theoretical reconstruction rather than direct empirical demonstration.11 What is no longer in doubt is that brain lateralization is both an ancient feature of primate neurobiology and a critically amplified one in humans, and that its evolutionary elaboration was inseparable from the emergence of the two capacities — language and complex tool manufacture — that most distinctively define our species.
References
The archaeological evidence for handedness in the Lower and Middle Pleistocene, and its implications for the evolution of laterality in hominids
Handedness in chimpanzees (Pan troglodytes) is associated with asymmetries of the primary motor cortex but not with homologous language areas
Language, tools and brain: The ontogeny and phylogeny of hierarchically organized sequential behavior
From monkey-like action recognition to human language: an evolutionary framework for neurolinguistics
The arcuate fasciculus and the disconnection theme in language and aphasia: history and current state