Overview
- Human skin color varies along a latitudinal gradient that closely tracks ultraviolet radiation intensity, the result of natural selection balancing two opposing pressures: protection of folate from UV-induced photodegradation near the equator versus the need for sufficient UVB penetration to synthesize vitamin D at higher latitudes.
- The genetic architecture of skin pigmentation involves dozens of genes, with independent evolution of light skin in European and East Asian populations through different sets of mutations, demonstrating convergent evolution driven by shared selective pressures after the dispersal of Homo sapiens out of Africa.
- Ancient DNA from Mesolithic European hunter-gatherers reveals that light skin pigmentation in Europe is surprisingly recent, with alleles for depigmentation at key loci such as SLC24A5 reaching high frequency only within the last 5,000 to 8,000 years, likely accelerated by the adoption of agriculture and reduced dietary vitamin D.
Human skin color is one of the most visibly variable traits in our species, ranging from deeply pigmented dark brown and black in equatorial populations to pale, lightly pigmented skin in populations at high latitudes. This variation is not random. It correlates closely with the intensity of ultraviolet (UV) radiation at different latitudes, reflecting the action of natural selection over tens of thousands of years as Homo sapiens dispersed from tropical Africa into environments with dramatically different UV regimes.1, 2 The evolutionary story of human skin color involves a delicate balance between two vital functions: protecting the body from the damaging effects of UV radiation and allowing enough UV penetration to synthesize vitamin D, an essential nutrient for skeletal health and immune function.
Research over the past several decades, particularly the work of Nina Jablonski and George Chaplin, has established that this balance is the primary selective force shaping the global distribution of skin pigmentation. The genetic architecture underlying skin color variation has also been substantially clarified through genome-wide association studies and ancient DNA analysis, revealing that light skin evolved independently in European and East Asian populations through different genetic pathways, and that the transition to light pigmentation in Europe occurred far more recently than previously assumed.5, 6, 10
The biology of melanin
Skin color in humans is determined primarily by the amount, type, and distribution of a pigment called melanin, produced by specialized cells called melanocytes in the basal layer of the epidermis. All humans, regardless of skin color, possess roughly the same number of melanocytes per unit area of skin. The difference in pigmentation arises not from the number of melanocytes but from the quantity and type of melanin they produce and the size and distribution of the organelles called melanosomes in which melanin is packaged and transferred to surrounding keratinocytes.7
Melanin exists in two primary forms. Eumelanin is a brown-to-black pigment that absorbs UV radiation very efficiently and is the dominant form in darkly pigmented skin. Pheomelanin is a yellow-to-red pigment that is less effective at UV absorption and is associated with lighter skin, red hair, and freckling. The ratio of eumelanin to pheomelanin in an individual's skin is under genetic control and varies among populations. Individuals with predominantly eumelanin-producing melanocytes have darker skin that provides greater protection against UV radiation, while those producing more pheomelanin have lighter skin that is more susceptible to UV damage but more efficient at vitamin D synthesis under low-UV conditions.7, 8
The melanin production pathway, called melanogenesis, is regulated by a cascade of enzymes and signalling molecules. The enzyme tyrosinase catalyses the initial rate-limiting steps of melanin synthesis, converting the amino acid tyrosine into intermediates that are then processed into either eumelanin or pheomelanin depending on the availability of cysteine and the activity of downstream enzymes. The melanocortin 1 receptor (MC1R), located on the surface of melanocytes, plays a central role in determining which type of melanin is produced: activation of MC1R by alpha-melanocyte-stimulating hormone promotes eumelanin synthesis, while reduced MC1R signalling favours pheomelanin production.7, 15
The vitamin D-folate hypothesis
The prevailing explanation for the global distribution of human skin pigmentation is the vitamin D-folate hypothesis, developed in its modern form by Jablonski and Chaplin. This hypothesis identifies two opposing selective pressures related to UV radiation that have shaped skin color as human populations spread across the globe.1, 2
The first pressure acts in high-UV environments near the equator, where intense ultraviolet radiation poses a direct threat to a critical B vitamin: folate (vitamin B9). Folate is essential for DNA synthesis and repair, cell division, and neural tube development in embryos. Branda and Eaton demonstrated in 1978 that human blood plasma exposed to simulated sunlight loses 30 to 50 percent of its folate within one hour, and that patients undergoing UV phototherapy for skin conditions develop abnormally low serum folate concentrations.4 Folate deficiency during pregnancy is strongly associated with neural tube defects such as spina bifida and anencephaly. Heavily melanized skin absorbs up to 99.9 percent of incident UVB radiation in the outer layers of the epidermis before it can penetrate to the deeper vascularized dermis where folate circulates, thereby protecting this vital nutrient from photodegradation. In equatorial environments, darkly pigmented skin thus provides a strong selective advantage by maintaining adequate folate levels for reproductive success.1, 2
The second pressure acts in low-UV environments at higher latitudes. Vitamin D3 (cholecalciferol) is synthesized in the skin when UVB radiation converts 7-dehydrocholesterol in epidermal cells into previtamin D3. Vitamin D is essential for calcium absorption and bone mineralization; its deficiency causes rickets in children and osteomalacia in adults, conditions that deform the skeleton and, in severe cases, can obstruct the birth canal, directly reducing reproductive fitness.3, 9 W. Farnsworth Loomis first proposed in 1967 that the lighter skin of high-latitude populations evolved to allow sufficient UVB penetration for vitamin D synthesis under conditions of reduced annual UV radiation.3 Heavily melanized skin at high latitudes would block so much UVB that individuals would be unable to produce adequate vitamin D, particularly during winter months when UV intensity is minimal. Selection therefore favoured depigmentation as populations moved into higher latitudes, reducing melanin production to permit vitamin D synthesis at lower UV intensities.1, 2
Jablonski and Chaplin tested this hypothesis by mapping the global distribution of skin reflectance data against satellite-derived UV radiation measurements. They found a remarkably strong correlation: populations that had lived for thousands of years at a given latitude possessed skin pigmentation closely matching the level predicted by a model balancing folate protection against vitamin D production needs. The correlation held across all continents and was the strongest predictor of skin color variation, exceeding other environmental or cultural variables.1, 2
Ultraviolet radiation and the latitudinal gradient
Ultraviolet radiation reaching the Earth's surface varies dramatically with latitude, altitude, season, and cloud cover. The UV spectrum is divided into three bands: UVA (315 to 400 nanometres), UVB (280 to 315 nanometres), and UVC (100 to 280 nanometres). UVC is absorbed entirely by the ozone layer and does not reach the surface. UVB, the band most relevant to both vitamin D synthesis and DNA damage, varies by a factor of approximately five to six between the equator and latitudes above 45 degrees. At the equator, UVB intensity is sufficient for vitamin D synthesis year-round, while at latitudes above approximately 40 degrees North or South, UVB intensity drops below the threshold for cutaneous vitamin D production during winter months, sometimes for six months or more.2, 9
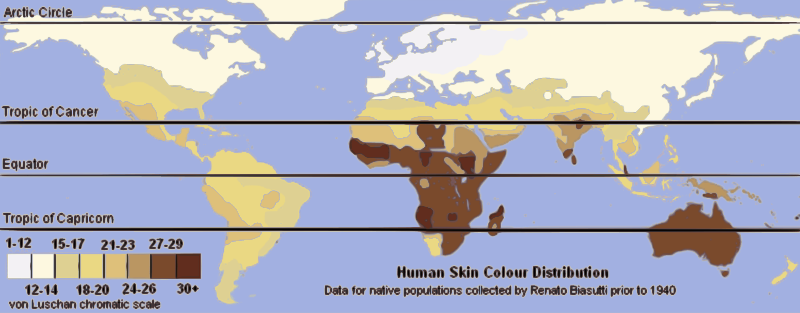
Average annual UVB radiation and predominant skin pigmentation by latitude zone1, 2
This latitudinal UV gradient maps directly onto the distribution of human skin pigmentation in populations that have occupied their regions for thousands of years. The darkest skin pigmentation is found among equatorial populations in sub-Saharan Africa, southern India, Melanesia, and Aboriginal Australia. Intermediate pigmentation characterizes populations in subtropical and warm-temperate zones, including the Mediterranean, the Middle East, Central Asia, and Southeast Asia. The lightest pigmentation occurs in populations of northern Europe and northeastern Asia that have lived at high latitudes for many millennia. There are exceptions: the Inuit of the Arctic have moderately dark skin despite living at very high latitudes, a fact explained by their traditional diet, which is exceptionally rich in vitamin D from marine fish and mammal fat, relaxing the selective pressure for depigmentation.1, 2, 12
Genetic basis of pigmentation variation
The genetic architecture of human skin pigmentation has been substantially elucidated through genome-wide association studies (GWAS), candidate gene analyses, and functional studies in model organisms. Skin color is a polygenic trait influenced by variants in dozens of genes, though a relatively small number of loci account for a large proportion of the variation between populations.7, 8
One of the most important genes is SLC24A5, which encodes a cation exchanger involved in melanosome biogenesis. Lamason and colleagues identified a single amino acid substitution (A111T, from alanine to threonine) in SLC24A5 that accounts for approximately 25 to 38 percent of the difference in skin pigmentation between Europeans and West Africans. The derived threonine allele is found at a frequency of nearly 100 percent in European populations but is virtually absent in sub-Saharan African and East Asian populations. The discovery arose from the study of the zebrafish golden mutant, which has reduced pigmentation due to disruption of the zebrafish ortholog of SLC24A5, and injection of human SLC24A5 mRNA rescued the pigmentation phenotype in mutant zebrafish, confirming the gene's role in melanogenesis.5
Other major pigmentation genes include SLC45A2 (also called MATP), which carries a derived light-skin allele (L374F) found at high frequency in Europeans; TYRP1, which influences pigmentation in both European and Melanesian populations through different variants; OCA2 and HERC2, which are major determinants of eye color and also influence skin pigmentation; and KITLG, which carries a derived allele associated with lighter skin that is shared between European and East Asian populations.7, 8
The melanocortin 1 receptor gene MC1R shows a distinctive pattern of variation across human populations. In African populations, MC1R is strongly conserved, with very little nonsynonymous variation, consistent with purifying selection to maintain the ancestral dark pigmentation phenotype. Outside of Africa, and particularly in Europe, MC1R harbours an unusually high number of functional variants, several of which are associated with red hair, fair skin, and freckling. This pattern has been interpreted as evidence of relaxed functional constraint on MC1R as populations moved to higher latitudes where the selective pressure for dark skin was reduced or reversed.15
A landmark 2017 study by Crawford and colleagues, working with Sarah Tishkoff's group, examined skin pigmentation genetics in diverse African populations. The study identified variants in or near SLC24A5, MFSD12, DDB1, TMEM138, OCA2, and HERC2 that are significantly associated with skin pigmentation within Africa, revealing that the genetic architecture of pigmentation is far more complex even within the continent where humans originated than previously recognized.6 Several of the alleles associated with light pigmentation in Africans are ancient and predate the out-of-Africa dispersal, challenging the assumption that light skin evolved exclusively in response to reduced UV at higher latitudes.6, 13
Convergent evolution of light skin
One of the most striking findings in the genetics of human skin pigmentation is that light skin evolved independently in European and East Asian populations through largely different sets of genetic variants. This represents a clear example of convergent evolution: the same phenotypic outcome, depigmented skin adapted to high-latitude UV environments, achieved through different molecular pathways in separate populations that experienced similar selective pressures after their ancestors dispersed from Africa.7, 8
In European populations, the derived light-skin alleles at SLC24A5 (A111T) and SLC45A2 (L374F) account for a substantial proportion of depigmentation and show signatures of strong positive selection, including long-range haplotype homozygosity and high between-population allele frequency differentiation. These alleles are found at very high frequency in Europeans (above 95 percent) but are rare or absent in East Asian populations.5, 7
In East Asian populations, different loci appear to be more important. The OCA2 gene carries a derived variant (H615R) that is common in East Asians but rare in Europeans. Variation in ADAM17 and other loci also contributes to the lighter pigmentation of East Asian populations through mechanisms distinct from the European pathway.7 The independent genetic paths to light skin in these two populations confirm that the global distribution of skin color is the product of natural selection operating on different available genetic variation in different populations, rather than the spread of a single set of depigmentation mutations from a common source.7, 8
Key pigmentation genes and their population distributions5, 6, 7
| Gene | Key variant | Effect | Highest frequency |
|---|---|---|---|
| SLC24A5 | A111T (rs1426654) | Major depigmentation effect | Europe (~99%), South Asia (~50%) |
| SLC45A2 | L374F (rs16891982) | Depigmentation | Europe (~95%) |
| MC1R | Multiple nonsynonymous variants | Red hair, fair skin, freckling | Europe (diverse variants) |
| OCA2 | H615R | Depigmentation | East Asia |
| MFSD12 | Multiple variants | Darker pigmentation when highly expressed | Africa (ancestral), East Asia (derived) |
| TYRP1 | Multiple variants | Influences eumelanin synthesis | Varies by population |
Ancient DNA and the recency of European light skin
Perhaps the most unexpected finding in the study of human skin pigmentation evolution is that light skin in Europe is much more recent than previously assumed. Ancient DNA analysis of Mesolithic and Neolithic European genomes has revealed that the alleles responsible for the lightest skin pigmentation in modern Europeans were not yet at high frequency among European hunter-gatherers as recently as 7,000 to 10,000 years ago.10, 11
In 2014, Olalde and colleagues sequenced the genome of a Mesolithic hunter-gatherer from the La Brana-Arintero site in northern Spain, dated to approximately 7,000 years before present. This individual carried the ancestral (dark-skin) alleles at SLC24A5 and SLC45A2, the two loci responsible for the largest proportion of skin depigmentation in modern Europeans, while simultaneously carrying derived alleles for blue eyes at the HERC2/OCA2 locus.10 This combination, dark skin and blue eyes, is virtually unknown among modern Europeans and demonstrates that the selective sweeps at different pigmentation loci occurred at different times and were driven by different pressures.
Wilde and colleagues examined pigmentation-associated alleles across a time series of ancient European genomes spanning the last 5,000 years and found direct evidence of positive selection driving the increase in frequency of light-skin alleles during this period. The derived SLC24A5 allele appears to have risen from intermediate frequency to near fixation in European populations within the last 5,000 to 8,000 years, a remarkably rapid sweep that indicates strong positive selection.11 Beleza and colleagues estimated, using population genetic modelling, that the European depigmentation alleles at SLC24A5, SLC45A2, and TYRP1 underwent selective sweeps beginning between 11,000 and 19,000 years ago, with selection intensifying during or after the Neolithic transition to agriculture.16
The acceleration of selection for light skin following the adoption of agriculture is consistent with the vitamin D hypothesis. Hunter-gatherer diets, particularly those including freshwater and marine fish, wild game, and organ meats, provide more dietary vitamin D than the cereal-based diets that replaced them during the Neolithic revolution. As agricultural populations became increasingly dependent on grains that are poor sources of vitamin D, the selective pressure to produce vitamin D through cutaneous synthesis would have intensified, driving more rapid depigmentation.12, 14
Pigmentation diversity within Africa
Africa is the continent where Homo sapiens originated, and it harbours the greatest genetic diversity of any continent, including in pigmentation genes. The traditional narrative that Africans uniformly have very dark skin is an oversimplification. While the average skin pigmentation of sub-Saharan African populations is indeed darker than that of non-African populations, there is substantial variation within Africa, from the very darkly pigmented Nilotic peoples of South Sudan and the Hadza of Tanzania to the lighter-skinned San (Bushmen) of southern Africa and some Ethiopian and Somali populations.6, 13
Crawford and colleagues (2017) and Martin and colleagues (2017) demonstrated that the genetic architecture of pigmentation within Africa is complex and ancient. Several alleles associated with light pigmentation in African populations predate the divergence of modern human populations and the out-of-Africa dispersal, suggesting that variation in skin color was already present among the earliest Homo sapiens populations. Some of the light-pigmentation alleles found in modern Europeans and East Asians were carried out of Africa by dispersing populations and subsequently underwent positive selection at higher latitudes, rather than arising as entirely new mutations outside of Africa.6, 13
The MFSD12 gene provides a particularly informative example. Crawford and colleagues found that reduced expression of MFSD12 is associated with darker pigmentation, and that derived variants at this locus are at high frequency in Nilo-Saharan and Niger-Congo populations with very dark skin as well as in East Asian and Native American populations. The presence of some of these derived alleles in both African and non-African populations suggests that the alleles are ancient, predating the divergence of these geographically distant groups, and that selection for both darker and lighter skin has operated on standing variation present in ancestral human populations.6
Signatures of natural selection
Multiple lines of genetic evidence confirm that natural selection, rather than genetic drift alone, has driven the evolution of human skin pigmentation. The signatures of positive selection at pigmentation loci are among the strongest detected anywhere in the human genome.5, 7
The SLC24A5 A111T variant in Europeans shows one of the highest levels of population differentiation (FST) of any single-nucleotide polymorphism in the human genome, with a derived allele frequency approaching 100 percent in Europeans and near zero in sub-Saharan Africans and East Asians. The region surrounding SLC24A5 also shows extended haplotype homozygosity in European populations, a hallmark of a recent selective sweep in which a beneficial allele has risen rapidly in frequency, dragging linked neutral variants along with it before recombination can break down the haplotype.5, 16
Conversely, the low nonsynonymous diversity at MC1R in African populations, compared to the high diversity at the same locus in European populations, indicates that purifying selection has maintained the ancestral, functional MC1R allele in environments with high UV radiation, where dark skin provides a fitness advantage. The relaxation of this constraint outside of Africa has allowed diverse MC1R variants to accumulate in European populations, including those associated with red hair and freckling.15
Ancient DNA time series have provided direct observation of allele frequency change at pigmentation loci over thousands of years, an exceptionally rare opportunity to measure natural selection in real time. The increase in frequency of the SLC24A5 derived allele from roughly 10 percent in Mesolithic Europeans to near fixation in modern Europeans occurred over a period of approximately 5,000 to 8,000 years, implying a selection coefficient on the order of 1 to 10 percent per generation, which is unusually strong for a human trait.11, 16
Cultural factors and modern health implications
The evolution of human skin pigmentation has not occurred in a purely biological vacuum. Cultural practices, including clothing, shelter, diet, and migration, have modified the selective pressures on skin color throughout human history and continue to do so today.2, 12
Clothing reduces UV exposure to the skin, which can be either beneficial (protecting against sunburn and skin cancer) or detrimental (reducing vitamin D synthesis). The adoption of body-covering clothing by populations migrating into cold climates would have intensified the selective pressure for depigmentation by further reducing the already limited UV radiation reaching the skin. Conversely, traditional diets rich in vitamin D, such as those of Arctic peoples who consume large quantities of fatty fish and marine mammal blubber, can relax the selective pressure for light skin, which may explain why Inuit populations maintain relatively dark pigmentation despite living at very high latitudes.2, 12
In the modern world, rapid migration, urbanization, and changes in lifestyle have created widespread mismatches between skin pigmentation and UV environment. Darkly pigmented individuals living at high latitudes, particularly in northern Europe or northern North America, are at elevated risk of vitamin D deficiency because their melanin-rich skin requires substantially more UVB exposure to produce adequate vitamin D than the skin of lighter-pigmented individuals at the same latitude.9 Vitamin D deficiency is associated with increased risk of rickets, osteoporosis, certain cancers, autoimmune diseases, and cardiovascular disease, and is particularly prevalent among dark-skinned immigrant populations in northern countries.9 Conversely, lightly pigmented individuals living in high-UV environments, such as people of northern European descent in Australia, face elevated rates of melanoma and other forms of skin cancer because their low melanin levels provide insufficient protection against UV-induced DNA damage.2
Understanding the evolutionary history of human skin pigmentation thus has direct relevance to public health. It illustrates how traits that were adaptive in ancestral environments can become sources of disease risk when populations move to new environments faster than natural selection can adjust their biology, a phenomenon sometimes described as evolutionary mismatch.2, 9, 12
Skin color and the concept of race
The evolutionary biology of skin pigmentation has important implications for the concept of biological race. Skin color has historically been the primary visible trait used to classify humans into racial categories, but the genetic evidence demonstrates that skin color is a superficial adaptation to local UV environments that provides essentially no information about the overall genetic relatedness of populations.2, 6
The convergent evolution of light skin in European and East Asian populations through entirely different genetic mechanisms illustrates that similar appearances can arise from different evolutionary histories. Two populations with similar skin color may be no more closely related genetically than two populations with very different skin color. Skin pigmentation is controlled by a small number of loci under strong directional selection and can change substantially over a few thousand years, while the vast majority of the genome, which reflects overall population history, changes far more slowly and tells a different story.6, 7, 13
The finding that substantial pigmentation variation existed within Africa before the out-of-Africa dispersal, and that some alleles associated with light skin in non-African populations are of ancient African origin, further underscores the complexity of skin color evolution and its inadequacy as a marker of deep genetic ancestry. The global distribution of skin color reflects a history of adaptation to local environments, not a hierarchy of genetic types, and the mechanisms that produce it are among the best-understood examples of recent natural selection in the human species.2, 6, 13
References
Understanding the evolution of human pigmentation: recent contributions from population genetics
Derived immune and ancestral pigmentation alleles in a 7,000-year-old Mesolithic European
Direct evidence for positive selection of skin, hair, and eye pigmentation in Europeans during the last 5,000 y
The evolution of human skin pigmentation involved the interactions of genetic, environmental, and cultural variables