Overview
- Bipedal locomotion and brain expansion are the two defining adaptive shifts in human evolution, but they followed very different timelines: upright walking appeared by at least 6 million years ago, while the major increase in brain size did not begin until roughly 2 million years ago with the emergence of the genus Homo.
- Bipedalism freed the hands from locomotion, enabling tool use, carrying, and the fine manual dexterity required for complex technology, while brain expansion supported the cognitive capacities—planning, language, social reasoning—that most distinguish humans from other primates.
- Neither adaptation evolved in isolation: dietary shifts toward higher-quality foods and cooking with fire helped fuel the metabolically expensive brain, while the social and technological consequences of larger brains drove further selection for cognitive ability in a feedback loop that accelerated across the Pleistocene.
Two adaptations above all others define the trajectory of human evolution: the transition to bipedal locomotion and the dramatic expansion of the brain. These are the anatomical and cognitive foundations upon which everything distinctively human—tool use, language, culture, and civilization—was ultimately built. Yet they did not evolve together as a single package. Bipedalism appeared early, with evidence in the fossil record by at least 6 million years ago, while the major expansion of brain size did not begin until roughly 2 million years ago with the genus Homo.1, 7 Understanding when and why each adaptation evolved, and how they interacted, is central to understanding what made us human.
The decoupling of these two defining traits overturned one of the oldest assumptions in paleoanthropology. For most of the twentieth century, scientists expected that brain expansion and bipedalism had evolved together—that our ancestors became smart and started walking upright at roughly the same time, driven by the same selective pressures. The fossil record has shown otherwise: for at least four million years, hominins walked upright with brains no larger than those of chimpanzees. Intelligence, in the human sense, was a much later development.1, 2
The evolution of bipedalism
Bipedal locomotion is the earliest defining adaptation of the hominin lineage. The oldest evidence comes from Orrorin tugenensis, whose femoral morphology at 6 million years ago shows the asymmetric cortical bone distribution characteristic of habitual bipeds, and from the contested postcranial remains of Sahelanthropus tchadensis at 7 million years ago.1, 7 By 4.4 million years ago, Ardipithecus ramidus had a pelvis restructured for upright walking, though it retained an opposable big toe for tree climbing.2 The Laetoli footprints of Australopithecus afarensis, dated to 3.66 million years ago, preserve biomechanical evidence of fully modern, human-like bipedal gait—an adducted big toe, a medial longitudinal arch, and weight transfer from heel to toe indistinguishable from that of modern humans.3

The transition to bipedalism required coordinated redesign of at least twelve anatomical regions: the pelvis shortened and broadened to support the trunk over a single stance leg; the femur angled inward to position the knee under the body's center of gravity; the foot developed a longitudinal arch and lost its grasping big toe; the spine acquired additional curvatures; and the foramen magnum shifted to the base of the skull to balance the head atop the vertebral column.1, 7 The selective pressures driving this transformation remain debated. Experimental biomechanics has demonstrated that bipedal walking in humans is roughly 75% less costly per unit distance than quadrupedal knuckle-walking in chimpanzees, suggesting that energetic efficiency may have been a key factor.4 Other hypotheses invoke thermoregulatory advantage (reducing solar exposure by standing upright), freeing the hands for carrying food or offspring, and postural feeding in woodland trees.7 The discovery that the earliest bipeds, including Ardipithecus, lived in wooded rather than open grassland environments has largely ruled out the once-popular savanna hypothesis as the sole explanation.2
The thermoregulatory hypothesis, advanced by Peter Wheeler, provides a complementary perspective. Wheeler demonstrated through biophysical modeling that an upright posture dramatically reduces the body surface area exposed to direct solar radiation at tropical latitudes, particularly during the midday hours when the sun is near zenith. A bipedal hominin standing upright exposes roughly 40% less surface area to direct sunlight than the same individual on all fours, while simultaneously raising the body into faster-moving air currents above the ground boundary layer, enhancing convective cooling. Combined with the loss of body hair and the expansion of eccrine sweat glands, bipedalism may have enabled early hominins to remain active during the heat of the day when competing predators and scavengers were forced into shade, conferring a significant foraging advantage.9, 1
Brain expansion
The human brain is roughly three times the size of a chimpanzee's, averaging approximately 1,350 cubic centimeters compared to roughly 400 cc in our closest living relatives. This expansion occurred in stages. Australopithecines had endocranial volumes of 370–550 cc, only modestly larger than those of chimpanzees. The earliest members of Homo increased to roughly 510–750 cc. Homo erectus reached 550–1,250 cc across its geographic and temporal range. And Homo sapiens averages approximately 1,350 cc.1, 5
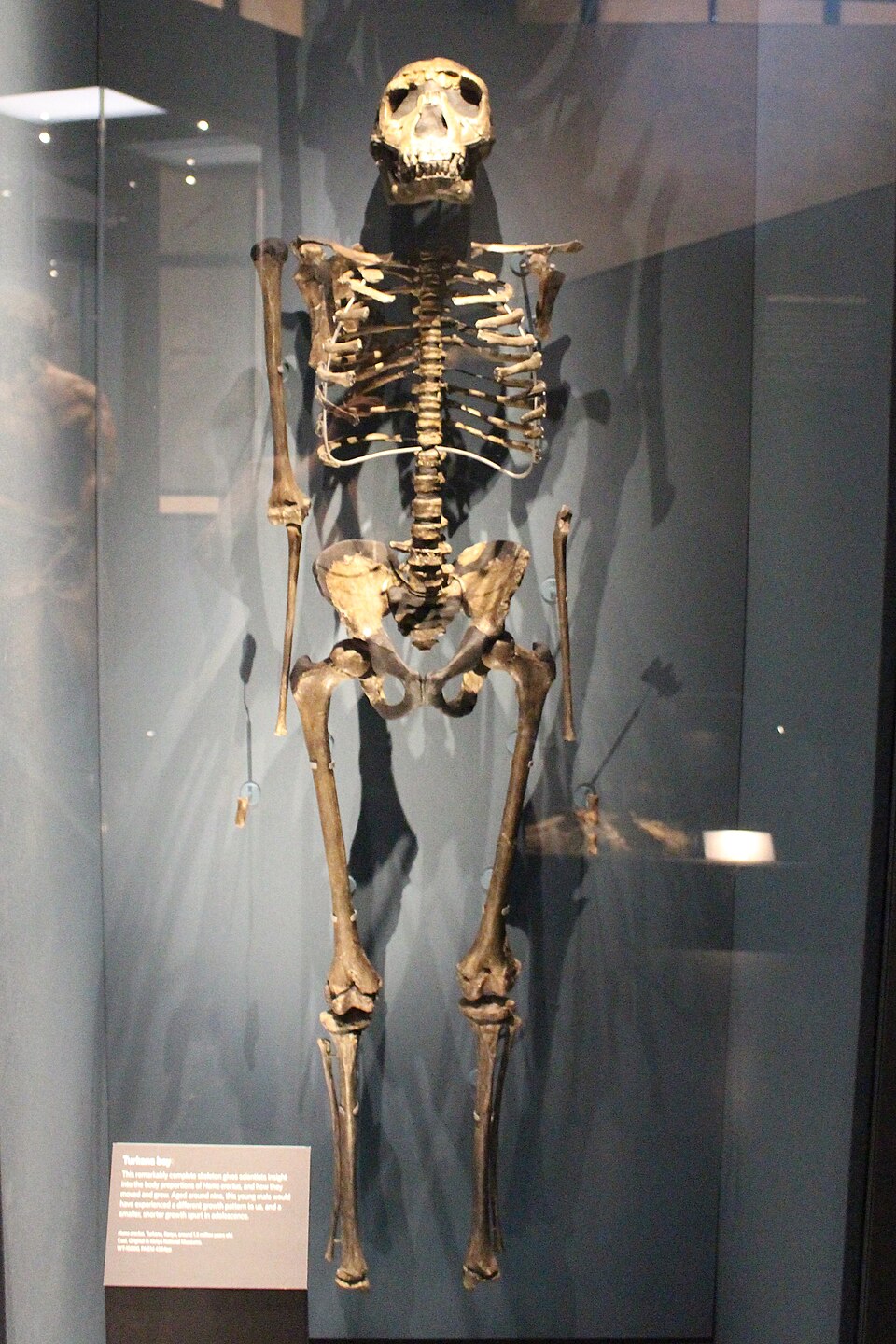
The expansion was not uniform across the brain. Endocast analyses reveal that the prefrontal cortex, temporal lobes, and parietal association areas expanded disproportionately in the human lineage, supporting the cognitive functions—planning, working memory, language, and social cognition—that most distinguish us from other primates.5, 7 Brain reorganization, including the development of asymmetric lateralization and expansion of the cerebellum, was at least as important as raw size increase. Homo naledi, with its small brain but evidence of complex behavior, demonstrates that internal reorganization could produce surprising cognitive capabilities even without the absolute brain volumes seen in H. sapiens.7
Fueling the brain
The metabolic cost of maintaining such a large brain is enormous. The human brain consumes roughly 20–25% of the body's resting energy despite comprising only about 2% of body mass.1, 8 The "expensive-tissue hypothesis," proposed by Leslie Aiello and Peter Wheeler in 1995, argued that the evolution of a larger brain was made possible in part by a reduction in gut size, enabled by a shift to higher-quality, more easily digested foods—particularly meat and animal fat.8 Archaeological evidence of butchery by at least 2.6 million years ago and of controlled fire use by at least 1.0 million years ago (with suggestive evidence as early as 1.7 million years) is consistent with this model: cooking dramatically increases the caloric yield and digestibility of both meat and plant foods, potentially underwriting the metabolic demands of a larger brain.1, 6

The social brain hypothesis offers a complementary explanation, proposing that the primary selective pressure for brain expansion was the computational demands of navigating increasingly complex social groups. Larger brains enabled more sophisticated social cognition—the ability to track alliances, predict behavior, communicate through language, and cooperate in large groups—which in turn enabled more effective foraging, hunting, defense, and child-rearing.1, 7 These dietary and social factors likely operated in a positive feedback loop: better nutrition supported larger brains, which enabled more complex social organization and technology, which in turn improved access to high-quality food. This self-reinforcing cycle accelerated across the Pleistocene, ultimately producing the cognitive capacities that define Homo sapiens.1, 8
Other key adaptations
Beyond bipedalism and brain expansion, several additional adaptations were critical to the emergence of modern humans. The evolution of the human hand, with its fully opposable thumb and precision grip, enabled the fine motor control required for producing increasingly sophisticated stone tools and, eventually, for complex manipulation of materials in art, clothing, and construction.1, 7 The reduction of the canine teeth and the shift from honing canine-premolar complexes to a more uniform dental battery accompanied changes in diet and social behavior, including the reduced importance of intrasexual combat displays that characterize many other primate species.1
The evolution of language, though impossible to date precisely from fossil evidence, was almost certainly among the most transformative adaptations in human evolution. The descent of the larynx, the expansion of Broca's and Wernicke's areas in the brain, and the fine neurological control of the tongue, lips, and diaphragm required for articulate speech all left indirect traces in the skeletal and endocranial record. The hyoid bone of the Kebara Neanderthal, morphologically modern in form, suggests that the anatomical prerequisites for speech were present at least by the Middle Pleistocene.1, 7 Together, these adaptations formed an interlocking suite of traits that, operating across millions of years of selection, transformed a bipedal ape with a chimpanzee-sized brain into the most ecologically dominant species in Earth's history.
References
Laetoli footprints preserve earliest direct evidence of human-like bipedal biomechanics
Microstratigraphic evidence of in situ fire in the Acheulean strata of Wonderwerk Cave
The expensive-tissue hypothesis: the brain and the digestive system in human and primate evolution
The thermoregulatory advantages of hominid bipedalism in open equatorial environments: the contribution of increased convective heat loss and cutaneous evaporative cooling