Overview
- The Burgess Shale is a Middle Cambrian (~508 Ma) fossil deposit in the Canadian Rockies of British Columbia that preserves an extraordinary assemblage of over 200 marine species with exceptional soft-tissue detail, providing an unparalleled window into the rapid diversification of animal life during the Cambrian explosion.
- Discovered by Charles Doolittle Walcott in 1909 and reinterpreted by Harry Whittington, Derek Briggs, and Simon Conway Morris in the 1970s, the Burgess Shale revealed that early Cambrian seas harboured a far greater diversity of body plans than previously recognized, including numerous stem-group organisms that combine features of different modern phyla.
- Rapid burial by submarine mudslides near the Cathedral Escarpment, combined with low-oxygen bottom waters and early carbonate cementation, inhibited microbial decay and preserved soft tissues including guts, eyes, and neural structures as carbonaceous compressions in fine-grained shale.
The Burgess Shale is a fossil deposit of extraordinary scientific importance located in the Canadian Rocky Mountains of British Columbia. Dating to approximately 508 million years ago during the Middle Cambrian (Wuliuan Stage, Miaolingian Series), it preserves an exceptional assemblage of over 200 species of marine organisms, many of which retain exquisite detail of soft tissues that almost never survive the fossilization process — including guts, eyes, gill structures, and even traces of neural tissue.2, 5 Deposits of this calibre, in which the preservation is so complete that even non-mineralizing organisms are recorded in fine anatomical detail, are termed Konservat-Lagerstätten (German for "conservation deposits"), and the Burgess Shale is among the most celebrated examples in the entire geological record.7
Situated within Yoho National Park at an elevation of approximately 2,300 metres, the Burgess Shale forms part of the Canadian Rocky Mountain Parks UNESCO World Heritage Site, inscribed in part because of the outstanding palaeontological significance of its fossils.19 The deposit was discovered by the American palaeontologist Charles Doolittle Walcott in 1909, systematically reinterpreted by a Cambridge research group in the 1970s and 1980s, and has remained a focal point of research into the origins and early diversification of animal life ever since. It provides an unparalleled window into the Cambrian explosion — the geologically rapid appearance of most major animal phyla in the fossil record — and its organisms have fundamentally reshaped scientific understanding of how animal body plans originated, diversified, and were pruned by extinction over deep time.1, 3, 13
Discovery and early research
The Burgess Shale was discovered in the late summer of 1909 by Charles Doolittle Walcott, then Secretary of the Smithsonian Institution and one of the foremost palaeontologists in North America. According to Walcott's own accounts, he encountered the first specimens of soft-bodied fossils on a ridge between Mount Wapta and Mount Field while conducting fieldwork in the Canadian Rockies near the town of Field, British Columbia.10 The precise circumstances of the discovery have been embellished in popular retellings — a famous story involving his wife's horse stumbling over a fossiliferous slab on a trail is almost certainly apocryphal — but there is no doubt that Walcott recognized the exceptional nature of the material immediately and returned the following year to open a systematic quarry on what is now called Fossil Ridge.10

Between 1910 and 1924, Walcott and his family, who frequently assisted with fieldwork, collected over 65,000 specimens from the quarry and shipped them to the Smithsonian in Washington, D.C.2, 10 He published a series of descriptions of the fossils, assigning most of them to existing groups of living organisms. Walcott interpreted the arthropods as relatives of modern crustaceans and chelicerates, the worms as polychaetes and priapulids, and the various enigmatic forms as members of one known phylum or another. This approach of fitting Cambrian organisms into modern taxonomic categories — which Stephen Jay Gould later characterized as "shoehorning" — reflected the prevailing assumption that the Cambrian fauna was fundamentally similar to later marine communities and that its apparent strangeness was merely a consequence of incomplete preservation and insufficient study.3
Walcott's sheer volume of administrative duties as Smithsonian Secretary limited the time he could devote to detailed anatomical analysis, and after his death in 1927, the vast majority of his Burgess Shale collection sat in the Smithsonian's drawers largely unstudied for decades. The fossils were not forgotten, but they were not the subject of sustained, intensive research until the 1960s, when a new generation of palaeontologists turned fresh eyes to the material and discovered that Walcott's taxonomic assignments were, in many cases, profoundly mistaken.2, 3
Reinterpretation by Whittington, Briggs, and Conway Morris
The modern era of Burgess Shale research began in 1966–1967, when Harry Blackmore Whittington of the University of Cambridge was invited to undertake a comprehensive restudy of the collection. Whittington, accompanied by the Geological Survey of Canada, conducted new quarrying operations at the original Walcott site and several adjacent localities, recovering additional material and meticulously re-examining the Smithsonian specimens using improved preparation techniques, including photography under ultraviolet light to enhance contrast between the dark organic films and the surrounding rock matrix.1, 2
Two of Whittington's graduate students — Derek Briggs, who specialized in arthropods, and Simon Conway Morris, who focused on the non-arthropod fauna — produced a landmark series of monographs through the 1970s and 1980s that fundamentally revised the taxonomy of the Burgess Shale organisms. Whittington's 1975 redescription of Opabinia regalis, a five-eyed animal with a forward-pointing flexible proboscis, was a turning point: when he presented his reconstruction at a meeting of the Palaeontological Association, the audience reportedly burst into laughter at the sheer strangeness of the creature, which defied placement in any known phylum.1, 20 Conway Morris's redescriptions of worm-like organisms revealed further anatomical surprises, and Briggs demonstrated that many of the arthropods lacked the defining features of any living arthropod group.5, 20
The collective conclusion of the Cambridge group was that the Burgess Shale contained a wealth of organisms that could not be accommodated within the classification of modern animals — the so-called "weird wonders" that appeared to represent extinct body plans with no living descendants.3 Stephen Jay Gould brought these findings to a broad public audience in his 1989 book Wonderful Life, in which he argued that the Burgess Shale demonstrated that the Cambrian explosion had produced a far greater range of fundamental body plans (morphological disparity) than survives today, and that the winnowing of this initial diversity was governed as much by historical contingency as by adaptive superiority.3 Gould's thesis provoked vigorous scientific debate, with Conway Morris later countering in The Crucible of Creation (1998) that evolutionary convergence, rather than contingency, was the dominant pattern in the history of life, and that many of Gould's "weird wonders" were in fact ancestors or relatives of living groups when analyzed with modern phylogenetic methods.4
Exceptional preservation
The preservation quality of the Burgess Shale is exceptional even among Lagerstätten. The vast majority of Cambrian organisms had no mineralized hard parts — no shells, no bones, no teeth — and under normal conditions of fossilization they would have decayed entirely, leaving no trace in the rock record. Yet in the Burgess Shale, delicate structures including the alimentary canal, musculature, gill lamellae, compound eyes, and even the nervous system are preserved as thin films of carbonaceous material compressed between layers of fine-grained mudstone.6, 23
The geological setting provides the key to this remarkable preservation. During the Middle Cambrian, the Burgess Shale organisms lived on and near the seafloor at the base of a steep submarine escarpment known as the Cathedral Escarpment, a near-vertical wall of carbonate reef rock (the Cathedral Formation) that rose above the adjacent deeper-water basin. Periodically, masses of fine-grained mud swept off the escarpment as turbidity currents or debris flows, rapidly burying the benthic and nektonic communities in events known as obrution deposits.6, 8 The rapid entombment in fine-grained sediment was a necessary first step, as it quickly isolated the organisms from scavengers and removed them from the oxygenated water column.
However, rapid burial alone is insufficient to explain the quality of preservation. Nicholas Butterfield's taphonomic studies demonstrated that the organic tissues of the Burgess Shale fossils were preserved through a pathway distinct from typical carbonization, involving the retention of original biological polymers at early stages of diagenesis.23 More recently, Robert Gaines and colleagues showed that the mechanism depended on the specific chemistry of the early Palaeozoic ocean. Bottom waters were low in dissolved oxygen, and the global ocean contained significantly lower sulphate concentrations than today. These conditions meant that once organisms were buried, the microbial processes that normally decompose organic matter — particularly sulphate reduction — were starved of the oxidants needed to proceed. The result was an early and nearly complete shutdown of microbial decay within the sediment.8 Furthermore, pervasive early cementation by carbonate minerals sealed the sediment within days to weeks of deposition, forming a physical barrier that prevented further oxidant diffusion from overlying seawater into the burial environment. This combination of rapid entombment, low-oxygen conditions, sulphate limitation, and early cementation created a narrow taphonomic window in which soft tissues could be preserved as carbonaceous compressions with extraordinary fidelity.7, 8
Analysis of the Greater Phyllopod Bed — the principal fossil-bearing horizon within the Walcott Quarry — has revealed that it consists of at least 26 discrete obrution event beds within a vertical thickness of approximately seven metres, each representing a separate burial event that entombed a snapshot of the living community. Over 50,000 specimens belonging to 158 genera have been recovered from this interval alone.6
Iconic organisms
Anomalocaris is perhaps the most famous Burgess Shale animal and one of the largest predators of the Cambrian seas. Reaching body lengths estimated at up to one metre, it possessed a pair of large, segmented frontal appendages used for grasping prey, a circular mouth apparatus composed of overlapping plates, a series of lateral swimming lobes along its body, and large compound eyes. Remarkably, Anomalocaris was originally described as three separate organisms: the frontal appendages were mistaken for the body of a shrimp-like crustacean, the circular mouth was described as a jellyfish, and the body was identified as a sea cucumber. It was not until the 1980s that Harry Whittington and Derek Briggs demonstrated that all three "species" were parts of a single, large predatory animal.5, 9 The related genus Hurdia, described in detail by Allison Daley and colleagues in 2009, expanded knowledge of the anomalocaridid body plan by revealing a prominent anterior carapace and exceptionally well-preserved gill structures, strengthening the interpretation of anomalocaridids as stem-group euarthropods — organisms on the evolutionary lineage leading to modern arthropods but lacking the full suite of arthropod characters.9
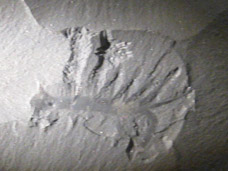
Hallucigenia is a small, worm-like animal whose history of misinterpretation has become legendary in palaeontology. When Simon Conway Morris described it in 1977, he named it for its "bizarre and dream-like appearance" and reconstructed it walking on stiff, bilaterally paired spines with a row of flexible tentacles waving along its back. This interpretation persisted for over a decade until the discovery of related fossils in the Chengjiang biota of China made it clear that Conway Morris had the animal upside down: the "tentacles" were legs and the "stilts" were dorsal protective spines.5, 12 In 2015, Martin Smith and Jean-Bernard Caron used electron microscopy to identify which end was the head, discovering a pair of simple eyes and a ring of pharyngeal teeth surrounding the mouth, along with needle-like teeth lining the throat. These features placed Hallucigenia securely among the lobopodians, a group closely related to the ancestors of modern velvet worms (Onychophora) and arthropods within the superphylum Ecdysozoa.12
Opabinia, the animal whose reconstruction provoked laughter at its first public presentation, possessed five stalked compound eyes arranged across the top of its head and a long, flexible, forward-pointing proboscis tipped with a claw-like grasping organ that it presumably used to capture food and convey it to a backward-facing mouth on the underside of its head. Its body bore a series of lateral lobes and gills but lacked the jointed legs characteristic of true arthropods. Modern phylogenetic analyses place Opabinia on the stem lineage of arthropods, below the anomalocaridids, making it a crucial form for understanding how the arthropod body plan was assembled incrementally.1, 20
Pikaia gracilens has long held iconic status as one of the earliest known chordates — the phylum that includes all vertebrates, including humans. A laterally compressed, ribbon-shaped animal roughly 4 to 5 centimetres long, it possessed a notochord (a stiffening rod along its back), V-shaped myomeres (muscle blocks), and what appears to be a vascular system, all diagnostic chordate features. A comprehensive redescription by Conway Morris and Caron in 2012, based on 114 specimens, confirmed its placement as a stem-group chordate, though its exact position within the chordate tree remains debated.11
Marrella splendens is the most abundant fossil in the Burgess Shale, accounting for over a third of all specimens recovered from the Walcott Quarry. It was a small arthropod, approximately 2 centimetres in length, with a distinctive head shield bearing two pairs of sweeping, spine-like projections and a body of 24 to 26 segments, each bearing a pair of biramous (two-branched) appendages. Despite being the first Burgess Shale fossil Walcott described, Marrella cannot be placed in any living arthropod class; it belongs to its own order, the Marrellomorpha, and is considered a stem-group arthropod.5, 21
Notable organisms of the Burgess Shale5, 9, 11, 12
| Organism | Modern affinity | Body length | Ecological role |
|---|---|---|---|
| Anomalocaris canadensis | Stem-group euarthropod (Radiodonta) | Up to ~1 m | Apex predator |
| Hallucigenia sparsa | Stem-group ecdysozoan (Lobopoda) | ~1–3 cm | Epibenthic grazer |
| Opabinia regalis | Stem-group euarthropod | ~4–7 cm | Predator / scavenger |
| Pikaia gracilens | Stem-group chordate | ~4–5 cm | Nektobenthic swimmer |
| Marrella splendens | Stem-group arthropod (Marrellomorpha) | ~2 cm | Filter feeder / detritivore |
| Wiwaxia corrugata | Stem-group mollusc or lophotrochozoan | ~2–5 cm | Epibenthic grazer |
| Hurdia victoria | Stem-group euarthropod (Radiodonta) | ~20–50 cm | Predator / suspension feeder |
| Cambroraster falcatus | Stem-group euarthropod (Radiodonta) | ~30 cm | Benthic predator (infaunal prey) |
| Titanokorys gainesi | Stem-group euarthropod (Radiodonta) | ~50 cm | Nektobenthic predator |
Significance for understanding animal evolution
The Burgess Shale's greatest contribution to science lies in what it reveals about the tempo and mode of early animal evolution. The deposit captures a moment in the Cambrian explosion when the major animal phyla were rapidly diversifying, and it demonstrates that the earliest representatives of these lineages often looked dramatically different from their modern descendants. Many Burgess Shale organisms are now understood to be stem-group members of living phyla — forms that lie on the evolutionary branch leading to a modern group but that had not yet acquired the full complement of features that define the group today.13 Anomalocaris, Opabinia, and Hurdia, for example, are stem-group euarthropods: they possessed some arthropod features (segmentation, compound eyes, lateral lobes) but lacked others (jointed legs, a hardened exoskeleton). Similarly, Pikaia is a stem-group chordate and Hallucigenia is a stem-group ecdysozoan, each combining features that in modern organisms are distributed among different phyla.11, 12, 13
The recognition of stem groups has resolved much of the controversy that surrounded the Burgess Shale in the wake of Gould's Wonderful Life. Gould had argued that many of the Burgess Shale organisms represented entirely independent evolutionary experiments — body plans so fundamentally different from anything alive today that they constituted phyla in their own right, each one an alternative version of animal life that went extinct without leaving descendants.3 If true, this would mean that the Cambrian explosion produced a far greater range of fundamental body plans (morphological disparity) than survives in the modern world, and that the subsequent history of life involved a progressive constriction of anatomical possibility. Briggs, Fortey, and Wills challenged this interpretation in a quantitative analysis of Cambrian arthropod morphospace, concluding that the range of morphological variation among Cambrian arthropods did not significantly exceed that among living arthropods, and that the appearance of extraordinary disparity was partly an artifact of the unfamiliar combinations of characters seen in stem-group forms.14
Modern cladistic and phylogenomic analyses have largely supported the stem-group interpretation, reassigning most of the erstwhile "weird wonders" to positions on the stems of recognized phyla rather than treating them as independent lineages.13 The broader lesson of the Burgess Shale, however, remains intact: the Cambrian explosion was not merely an increase in the number of species but a profound expansion of anatomical innovation, during which the modular building blocks of animal body plans — segments, appendages, eyes, guts, nervous systems — were combined and recombined in an extraordinary range of configurations. The stem-group organisms of the Burgess Shale preserve this combinatorial experimentation in exceptional detail, providing evidence for how complex body plans were assembled step by step along the branches leading to modern phyla.13, 20
Continuing research and new discoveries
Research on the Burgess Shale has continued to produce major discoveries well into the twenty-first century. In 2012, a team led by Jean-Bernard Caron of the Royal Ontario Museum discovered a spectacular new fossil locality in the Burgess Shale Formation at Marble Canyon in Kootenay National Park, approximately 40 kilometres southeast of the original Walcott Quarry. The Marble Canyon site, described in Nature Communications in 2014, yielded an arthropod-dominated assemblage with remarkably high density and diversity of soft-bodied fossils, including a substantial proportion of species new to science — approximately 22 percent of the total diversity recovered in the initial collecting seasons.15

Among the new taxa described from recent fieldwork are Cambroraster falcatus, a large hurdiid radiodont with a horseshoe-shaped carapace that earned it the nickname "the Millennium Falcon" among the research team. Described by Moysiuk and Caron in 2019, Cambroraster reached approximately 30 centimetres in length and possessed rake-like frontal appendages adapted for sifting through seafloor sediment in search of infaunal prey, demonstrating that even early Cambrian predators had already diversified into specialized ecological niches.16 In 2021, the same team described Titanokorys gainesi, a giant hurdiid radiodont approximately half a metre long with an enormous head carapace so large relative to its body that the animal has been described as "little more than a swimming head." Titanokorys represents one of the largest animals known from the Cambrian and underscores the ecological importance of radiodonts as apex and near-apex predators in early Palaeozoic marine ecosystems.22
These ongoing discoveries demonstrate that the Burgess Shale is far from exhausted as a source of new scientific knowledge. Modern collecting techniques, improved imaging technologies including micro-CT scanning and synchrotron radiation, and the discovery of new fossiliferous horizons continue to expand the known diversity of the Burgess Shale biota and to refine understanding of the anatomy, ecology, and evolutionary relationships of its organisms.15, 16
Other Cambrian Lagerstätten
The Burgess Shale is the most famous Cambrian Lagerstätte, but it is not unique. Several other deposits around the world preserve soft-bodied Cambrian faunas of comparable quality, and together these sites provide a comprehensive picture of marine ecosystems during the Cambrian explosion.7
The Chengjiang biota of Yunnan Province, China, discovered by Hou Xianguang in 1984, is approximately ten million years older than the Burgess Shale, dating to approximately 518 million years ago (Cambrian Stage 3). It preserves a diverse assemblage of over 250 species with soft-tissue preservation comparable to the Burgess Shale, including arthropods, lobopodians, priapulid worms, sponges, ctenophores, and early chordates and vertebrates. Some taxa are shared with the Burgess Shale, but others are unique to Chengjiang, and the older age of the deposit means it captures an earlier phase of the Cambrian radiation.17 The Qingjiang biota, also from South China and described in 2019, is roughly contemporaneous with the Chengjiang (~518 Ma) but preserves a remarkably different taxonomic composition, with a high proportion of cnidarians and other non-arthropod forms, suggesting significant ecological heterogeneity among early Cambrian marine communities even at similar latitudes and time intervals.18
Other important Cambrian Lagerstätten include the Sirius Passet fauna of northern Greenland (~518 Ma), which preserves exceptionally large arthropods and lobopodians in a high-latitude setting; the Emu Bay Shale of Kangaroo Island, South Australia (~514 Ma), notable for three-dimensional preservation of soft tissues through pyritization; and the Kaili biota of Guizhou Province, China (~508 Ma), which is approximately contemporaneous with the Burgess Shale and shares several genera with it.7 The global distribution of these deposits demonstrates that Burgess Shale-type preservation was not a geographically isolated phenomenon but occurred wherever the appropriate taphonomic conditions — rapid burial, low-oxygen bottom waters, limited sulphate availability, and early cementation — coincided during a narrow interval of the early Palaeozoic.7, 8
Butterfield has argued that the temporal restriction of Burgess Shale-type preservation to the Cambrian and earliest Ordovician reflects a fundamental change in ocean chemistry: as atmospheric oxygen levels rose and marine sulphate concentrations increased through the Palaeozoic, the taphonomic window for this mode of preservation closed permanently, making the Cambrian Lagerstätten an irreplaceable record of a style of fossilization that can never be replicated in younger rocks.7
References
The enigmatic animal Opabinia regalis, Middle Cambrian, Burgess Shale, British Columbia
The Burgess Shale anomalocaridid Hurdia and its significance for early euarthropod evolution
Pikaia gracilens Walcott, a stem-group chordate from the Middle Cambrian of British Columbia
Wonderful strife: systematics, stem groups, and the phylogenetic signal of the Cambrian radiation
A new hurdiid radiodont from the Burgess Shale evinces the exploitation of Cambrian infaunal food sources
The Qingjiang biota — a Burgess Shale-type fossil Lagerstätte from the early Cambrian of South China
Extraordinary fossils reveal the nature of Cambrian life: a commentary on Whittington (1975)
A new study of Marrella splendens (Arthropoda, Marrellomorpha) from the Middle Cambrian Burgess Shale, British Columbia, Canada
A giant nektobenthic radiodont from the Burgess Shale and the significance of hurdiid carapace diversity
Organic preservation of non-mineralizing organisms and the taphonomy of the Burgess Shale