Overview
- In 2005, Mary Schweitzer reported the discovery of flexible structures resembling blood vessels and cells inside a 68-million-year-old Tyrannosaurus rex femur (MOR 1125) — a finding that young-earth creationists claimed proved dinosaurs lived recently.
- However, the structures are not fresh, unaltered tissue: they are heavily modified, iron-cross-linked protein remnants preserved by a mechanism in which iron from haemoglobin acts as a fixative, cross-linking proteins and preventing complete decay — a process Schweitzer herself has published on extensively.
- The fossils remain dated to 68 million years by multiple independent methods, and Schweitzer — a practising Christian — has publicly criticised the misuse of her work by young-earth creationist organisations.
In 2005, palaeontologist Mary Schweitzer and colleagues at North Carolina State University reported the discovery of flexible, transparent structures resembling blood vessels and osteocytes (bone cells) inside a 68-million-year-old Tyrannosaurus rex femur, specimen MOR 1125, recovered from the Hell Creek Formation in Montana.1 The discovery was immediately sensational. The structures were flexible, retained original morphology, and appeared to contain remnants of proteins. Young-earth creationist organisations seized on the finding as evidence that dinosaurs could not be millions of years old, arguing that soft tissue cannot survive for such timescales. However, more than a decade of subsequent research by Schweitzer and other groups has elucidated the preservation mechanism, demonstrated that the structures are heavily modified chemical remnants rather than fresh tissue, and confirmed that rare long-term molecular preservation is consistent with known chemistry under favourable conditions.4, 5
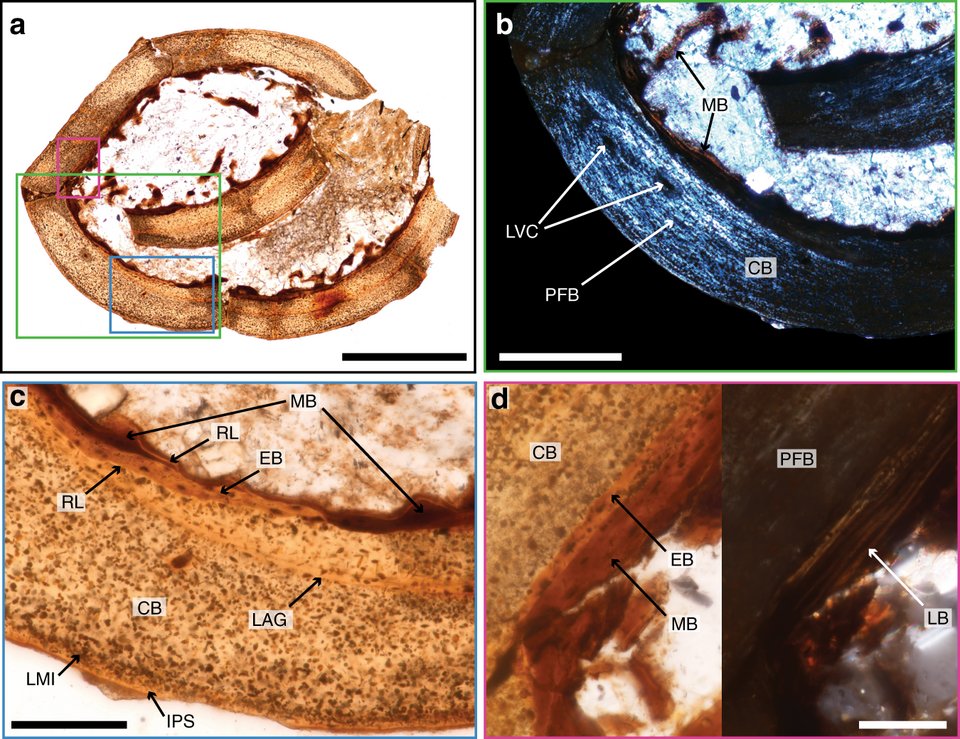
↑ Bailleul, Alida M. (20 March 2019). “An E…, Wikimedia Commons, CC BY-SA 4.0
The discovery
MOR 1125, a Tyrannosaurus rex femur from the Hell Creek Formation of eastern Montana, was excavated in 2003. The specimen was unusually well preserved and, because of its size and remote recovery location, was broken during transport — exposing the internal medullary cavity. When Schweitzer's team dissolved the mineral matrix of bone fragments in a mild acid solution (EDTA), flexible, branching structures emerged that closely resembled blood vessels in both morphology and texture. Within these vessel-like structures, small round bodies resembling red blood cells and star-shaped structures resembling osteocytes were also observed.1
Subsequent analyses using mass spectrometry identified peptide sequences consistent with collagen, the primary structural protein in bone. Schweitzer and colleagues reported collagen sequences from both MOR 1125 and a second specimen, the hadrosaur Brachylophosaurus canadensis (specimen MOR 2598, approximately 80 million years old), with peptide sequences matching those expected from archosaurs and showing phylogenetic affinity to birds, the living descendants of theropod dinosaurs.2, 6 These molecular results were initially controversial, with some researchers suggesting that the detected proteins could be modern contamination or biofilm. However, subsequent work by Schroeter and colleagues in 2017, using more stringent contamination controls and advanced mass spectrometry, confirmed the presence of endogenous collagen peptides in dinosaur bone, largely resolving the contamination debate.12
The preservation mechanism
The critical scientific question raised by the discovery was not whether the structures existed — their physical reality was confirmed by multiple laboratories — but how proteinaceous material could survive for tens of millions of years when laboratory studies of protein degradation kinetics suggest that collagen should decay to undetectable levels within a few million years under typical geological conditions. Schweitzer's research group has addressed this question through a series of experimental and analytical studies that have identified iron-mediated cross-linking as a primary preservation mechanism.4, 5
When red blood cells break down after death, haemoglobin releases iron in a reactive form (Fe2+/Fe3+). This iron generates hydroxyl radicals through Fenton chemistry, which in turn initiate cross-linking reactions that bond adjacent protein molecules together into a highly stable, insoluble polymer. Schweitzer and colleagues demonstrated this mechanism experimentally by soaking modern ostrich blood vessels in concentrated haemoglobin solution. After two years at room temperature, the treated vessels remained intact and retained their original morphology, while untreated control vessels decayed completely. The iron-treated tissues also showed the same dark brown colouration and resistance to enzymatic digestion observed in the dinosaur specimens.4, 5
This mechanism explains why the preserved structures are not "fresh" tissue in any meaningful sense. They are heavily modified chemical remnants: the original proteins have been cross-linked into a stable polymer that resists further degradation, analogous to the chemical tanning of leather. The flexibility and vessel-like morphology are preserved, but the molecular composition has been profoundly altered. The structures are, in Schweitzer's terminology, "molecular fossils" — they retain the shape and some of the original molecular signatures of the tissues, but are chemically distinct from living tissue.3, 4
Additional specimens and broader pattern
The T. rex discovery was not an isolated anomaly. Schweitzer's group subsequently reported similar preservation in the Brachylophosaurus canadensis specimen, including vessel-like structures, osteocyte-like cells, and collagen peptide sequences.6 Molecular evidence of preserved proteins has also been reported from Late Cretaceous sauropod dinosaur eggshells.9 In 2015, Bertazzo and colleagues at Imperial College London reported fibrous structures and cell-like bodies in eight Cretaceous dinosaur specimens from the Natural History Museum in London, some approximately 75 million years old, suggesting that molecular preservation in Mesozoic bone may be more common than previously assumed.7
The emerging pattern is consistent with rare but possible long-term preservation under specific taphonomic conditions: rapid burial, early diagenetic mineralisation of the bone, and the presence of iron from haemoglobin breakdown as a cross-linking agent. These conditions are not met in most fossils, which is why molecular preservation remains exceptional rather than routine. But their occurrence in multiple specimens from different formations, continents, and geological periods indicates that the Oklo-like rare-but-real phenomenon is a genuine feature of the fossil record, not a measurement artefact.7, 8
Independent dating of the specimens
The geological age of the Hell Creek Formation, from which MOR 1125 was recovered, is established by multiple independent dating methods. 40Ar/39Ar radiometric dating of volcanic ash beds (bentonites) interbedded with the Hell Creek sediments yields ages consistent with the latest Cretaceous, approximately 66–68 million years ago.10, 11 The formation's biostratigraphy (the characteristic assemblage of dinosaur, mammal, and plant fossils) is consistent with other Late Cretaceous deposits worldwide, and its position immediately below the Cretaceous–Paleogene boundary is confirmed by the iridium anomaly and shocked quartz that mark the asteroid impact event.11, 13
The soft tissue discovery does not challenge these dates. The specimens are embedded within a geological and geochronological framework established by decades of independent work using radiometric dating, biostratigraphy, magnetostratigraphy, and sequence stratigraphy. To reject the 68-million-year age of MOR 1125 on the basis of soft tissue preservation would require simultaneously rejecting all of these independent lines of evidence — a position that is not supported by any published scientific research.11, 13
Protein decay kinetics and theoretical limits
A central question in the debate over dinosaur soft tissue concerns the theoretical upper limit for protein survival in geological settings. Laboratory kinetic studies of collagen degradation predict that under typical burial temperatures of 7–10°C, collagen peptide bonds should hydrolyze to undetectable levels within approximately 2.7 million years, far shorter than the 68-million-year age of MOR 1125.14 However, these predictions assume free protein in an open aqueous system. In bone, collagen is tightly bound to hydroxyapatite mineral and is partially shielded from hydrolysis by the inorganic matrix, a phenomenon termed "mineral shielding." Buckley and Collins demonstrated that mineral-bound collagen degrades orders of magnitude more slowly than free collagen, extending the theoretical survival window into the tens of millions of years under favorable burial conditions such as low temperature, low water activity, and the absence of microbial colonization.8, 16
The iron-mediated cross-linking mechanism described by Schweitzer adds a second layer of stabilization beyond mineral shielding. When proteins are cross-linked into an insoluble polymer by Fenton chemistry, they become resistant to both enzymatic digestion and hydrolysis, effectively removing them from the normal degradation pathway.4, 5 Cleland and colleagues confirmed that endogenous collagen I peptides could be identified in dinosaur bone even after rigorous demineralization and contamination controls, and that the recovered sequences were phylogenetically consistent with archosaurian (bird-line) affinity, ruling out mammalian or microbial contamination as a source.15 The emerging consensus is that protein survival over deep time is not a single mechanism but a combination of mineral shielding, iron cross-linking, and favorable diagenetic conditions, each of which extends the preservation window beyond what any single factor alone would permit.4, 8, 17
Young-earth creationist misuse
Young-earth creationist organisations have prominently cited Schweitzer's discoveries as evidence that dinosaurs are only thousands of years old. This claim misrepresents both the findings and the scientific context. The preserved structures are not fresh, unaltered tissue: they are chemically modified remnants whose preservation is explained by iron-mediated cross-linking chemistry. The claim that "soft tissue cannot survive millions of years" is an argument from incredulity that ignores the published experimental evidence demonstrating the preservation mechanism.4, 5
Schweitzer herself, a practising Christian, has repeatedly and publicly criticised the appropriation of her work by young-earth organisations. In a 2006 interview with Discover magazine, she stated: "One thing that does bother me is that young-earth creationists take my research and use it for their own message, and I think they are completely wrong." She has emphasised that her research is conducted within the framework of mainstream geology and that nothing in her findings contradicts the established age of the specimens.1 The discovery of preserved molecular remnants in Mesozoic fossils is a significant advance in taphonomy and palaeobiology, expanding our understanding of how biological molecules can persist under favourable conditions. It does not, however, provide any evidence for a young Earth.4, 12
References
Molecular analyses of dinosaur osteocytes support the presence of endogenous molecules
A role for iron and oxygen chemistry in preserving soft tissues, cells and molecules from deep time
Biomolecular characterization and protein sequences of the Campanian hadrosaur B. canadensis
Collagen survival and its use for species identification in Holocene-lower Pleistocene bone fragments
Molecular taphonomy of animal and plant cuticles: selective preservation and diagenesis