Overview
- Eurypterids, commonly known as sea scorpions, were chelicerate arthropods that dominated Paleozoic marine, brackish, and freshwater environments from their origin in the Middle Ordovician approximately 467 million years ago through their final extinction in the end-Permian mass extinction 252 million years ago, producing the largest arthropods ever to have lived.
- The giant eurypterid Jaekelopterus rhenaniae from the Early Devonian of Germany reached an estimated body length of 2.5 metres, making it the largest known arthropod in Earth's history — exceeding the size of any modern arthropod by an order of magnitude and rivaling some marine vertebrates as an apex predator of its time.
- Eurypterids are classified within Chelicerata as the likely sister group or stem lineage of arachnids, and their evolutionary history — including a broad transition from marine to freshwater and possibly semi-terrestrial habitats during the Silurian and Devonian — provides critical evidence for understanding the colonization of land by arthropods.
Eurypterids, popularly known as sea scorpions, were a diverse order of chelicerate arthropods that inhabited Paleozoic seas, lagoons, rivers, and lakes from the Middle Ordovician through the end of the Permian, a span of roughly 215 million years.2, 6 They rank among the most formidable invertebrate predators in Earth's history and include the largest arthropods ever to have lived: the giant pterygotid Jaekelopterus rhenaniae, estimated from an isolated cheliceral claw to have reached a body length of approximately 2.5 metres.1 Although their common name suggests a close relationship with true scorpions, eurypterids are more properly understood as an extinct order (Eurypterida) within the subphylum Chelicerata, representing either the sister group to arachnids or a stem lineage from which the arachnid radiation ultimately descended.3, 7 Their fossil record, spanning from the Ordovician radiation to the [end-Permian mass extinction](/paleontology/end-permian-mass-extinction), documents a remarkable evolutionary history of body plan diversification, gigantism, ecological diversification from marine to freshwater habitats, and eventual decline.2, 6
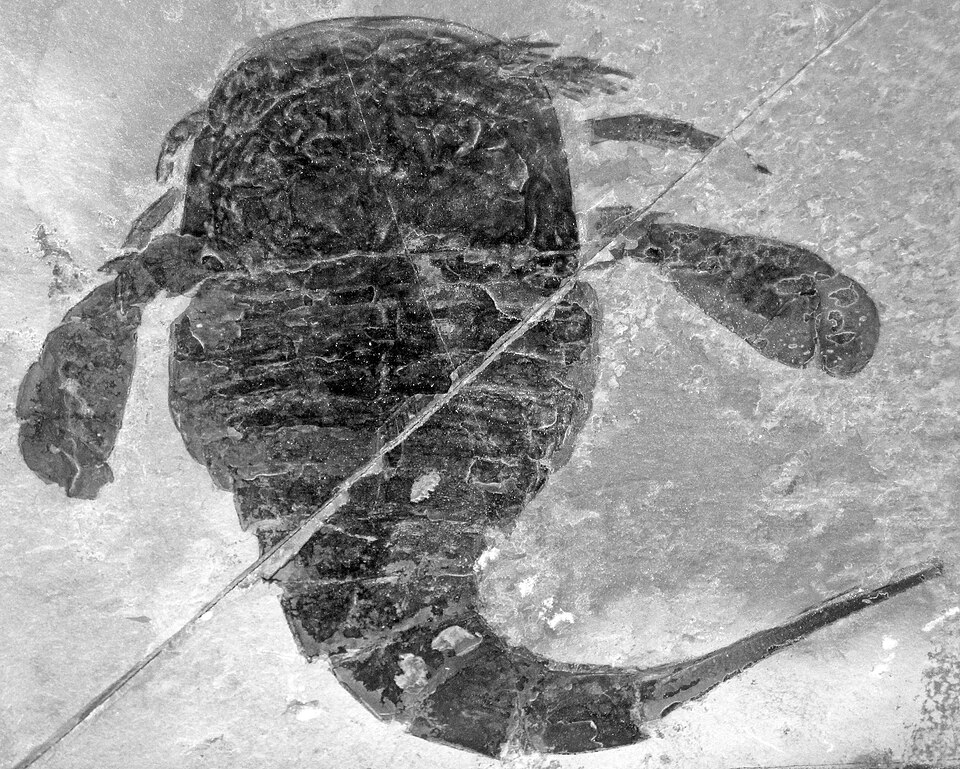
Body plan and morphology
The eurypterid body plan follows the general chelicerate ground plan: the body is divided into a prosoma (head region) bearing six pairs of appendages and an opisthosoma (trunk) composed of twelve segments plus a terminal telson.5, 6 The prosoma is covered by a dorsal carapace that bears a pair of large lateral compound eyes and a pair of smaller median ocelli, giving eurypterids excellent visual capabilities that are consistent with a predatory lifestyle.5 The first pair of prosomal appendages, the chelicerae, were small pincer-like structures in most eurypterids but became massively enlarged into toothed raptorial claws in the pterygotid family, serving as the primary prey-capture organs in these apex predators.1, 11
The remaining five pairs of prosomal appendages varied in form across different eurypterid lineages and provide the primary characters for higher-level classification. In most eurypterids, the second through fifth appendage pairs were walking legs, while the sixth pair was enlarged into broad, paddle-like swimming appendages in many aquatic species — the feature that gives the order its name (from the Greek eury-, "broad," and pteron, "wing").5, 3 Lamsdell and Braddy demonstrated that the morphology of the coxae (basal segments) of the walking legs provides reliable phylogenetic characters for resolving relationships within the order, and coxal gnathobases (toothed inner projections) indicate that the legs also functioned in food processing, passing prey items forward toward the mouth in a manner analogous to horseshoe crabs.3
The opisthosoma was typically divided into a broader mesosoma of seven segments and a narrower metasoma of five segments, though the degree of differentiation between these regions varies among lineages.6 The telson took diverse forms: a pointed spine in some groups (superficially resembling the sting of a true scorpion, though there is no evidence for a venom apparatus), a broad flattened paddle in others, and a simple rounded plate in still others.5, 6 Recent micro-CT studies of exceptionally preserved eurypterid specimens have revealed details of the respiratory structures on the ventral surface of the opisthosoma, showing gill-like organs in marine species that appear transitional toward the book lungs of terrestrial arachnids, providing a potential anatomical link between aquatic and terrestrial chelicerate respiration.15
Key genera and gigantism
The genus Eurypterus, from the Silurian of North America and Europe, is the most abundant and best-known eurypterid, with Eurypterus remipes serving as the state fossil of New York.12, 5 Eurypterus was a relatively small eurypterid, typically 15 to 25 centimetres in length, with broad swimming paddles and small chelicerae, suggesting an active swimming lifestyle and a generalized predatory or scavenging diet. Its exceptional abundance in Silurian lagoonal deposits of New York State — where hundreds of specimens have been collected from the Bertie Formation and equivalent units — makes it one of the most commonly encountered Paleozoic arthropod fossils.12, 14
Pterygotus and its relatives in the family Pterygotidae represent the ecological apex of eurypterid evolution. These were large, active predators with elongate bodies, reduced walking legs, powerful swimming paddles, and enormously enlarged chelicerae armed with denticles for seizing and processing prey.11 Pterygotus species from the Silurian and Devonian reached lengths of over one metre, making them among the largest predators in pre-vertebrate marine ecosystems.11, 2 The largest eurypterid of all, Jaekelopterus rhenaniae from the Early Devonian of the Rhineland in Germany, is known from a single cheliceral claw measuring 46 centimetres in length. Braddy, Poschmann, and Tetlie extrapolated from this claw to estimate a total body length of approximately 2.5 metres, exceeding all other known arthropods by a substantial margin.1
The question of how eurypterids achieved such extraordinary sizes — far exceeding the maximum body size of any living arthropod — has been addressed from multiple angles. Elevated atmospheric oxygen levels during the Silurian and Devonian may have relaxed the respiratory constraints that limit arthropod body size, while the absence of large vertebrate predators in early Paleozoic aquatic ecosystems may have allowed eurypterids to fill apex predator niches that would later be occupied by fish.1, 9 Lamsdell and Braddy tested Cope's Rule (the tendency for body size to increase through evolutionary time) in eurypterids and found support for a directional trend toward larger body sizes within several eurypterid lineages, suggesting that selection consistently favoured larger individuals, possibly through improved predatory efficiency or competitive advantages.9
Ecology and habitat
Eurypterids occupied a wide range of aquatic habitats over their 215-million-year history, and their ecological distribution shifted substantially through time.2 The earliest eurypterids, from the Middle and Late Ordovician, are found predominantly in marine shelf deposits, suggesting an initially marine ecology.8, 2 During the Silurian, eurypterids became particularly abundant in marginal marine settings — hypersaline lagoons, estuaries, and tidal flats — environments that are often interpreted as transitional between fully marine and freshwater habitats. The iconic Eurypterus-bearing lagerstatte of the Bertie Formation in New York State represents just such a restricted, hypersaline lagoonal environment.12, 14
By the Devonian and Carboniferous, an increasing proportion of eurypterid species are found in freshwater deposits, including river channels, lakes, and swamp environments, documenting a broad evolutionary transition from marine to continental waters that parallels the invasion of fresh water by many vertebrate lineages during the same interval.2 Tetlie demonstrated that this marine-to-freshwater transition occurred independently in multiple eurypterid lineages, suggesting that the physiological adaptations required for osmoregulation in dilute water evolved convergently across the order rather than being inherited from a single freshwater ancestor.2 Some authors have speculated that certain eurypterids may have been capable of brief terrestrial excursions, analogous to the amphibious behaviour of modern horseshoe crabs, though direct evidence for terrestrial locomotion in eurypterids remains limited.15
Phylogenetic relationships
The phylogenetic position of eurypterids within Chelicerata has been debated for over a century, but recent analyses converge on placing them as close relatives of arachnids — either as the sister group to Arachnida or as a paraphyletic grade from which arachnids evolved.3, 7 Lamsdell's comprehensive phylogenetic analyses of eurypterid morphology, incorporating coxal characters, appendage structure, and opisthosomal segmentation, supported a sister-group relationship between Eurypterida and Arachnida within a clade he termed Sclerophorata.6, 10 This placement implies that the last common ancestor of eurypterids and arachnids was an aquatic chelicerate, and that the terrestrial adaptations of arachnids — including book lungs, a waterproof cuticle, and modified reproductive structures — evolved from an aquatic ancestor rather than representing a primitive terrestrial condition.7, 13
The relationship between eurypterids and xiphosurans (horseshoe crabs) has also been the subject of extensive debate. Although eurypterids and horseshoe crabs share numerous morphological similarities — including a prosomal carapace with lateral and median eyes, coxal gnathobases, and book gills — most recent phylogenetic analyses place them in separate clades within Chelicerata, with horseshoe crabs forming a group with [trilobites](/paleontology/trilobites) and eurypterids aligning with arachnids.7, 10
Decline and extinction
Eurypterid diversity declined markedly after the Devonian, a pattern that has been linked to the rise of jawed fish — particularly the armored placoderms and the early sharks — as the dominant aquatic predators.2, 6 The giant pterygotids, which had been the apex predators of Silurian and Early Devonian seas, disappeared entirely by the end of the Devonian, possibly outcompeted or displaced by the rapidly diversifying gnathostome vertebrates that occupied similar ecological niches with greater physiological efficiency.2 Through the Carboniferous and Permian, eurypterid diversity was reduced to a handful of lineages, mostly small-bodied forms restricted to freshwater environments, a shadow of the group's former ecological prominence.2, 6
The final eurypterids disappear from the fossil record at or near the end-Permian boundary approximately 252 million years ago, victims of the most catastrophic mass extinction in Earth's history, an event that eliminated an estimated 81% of all marine species and fundamentally restructured both marine and terrestrial ecosystems.2 By the time of the end-Permian crisis, eurypterids were already reduced to low diversity and restricted geographic and ecological ranges, and the extinction event appears to have simply finished off a group that had been in long-term decline for over 100 million years. No eurypterid is known from any post-Paleozoic deposit, and the order left no direct descendants — though its close relatives the arachnids, which had diversified extensively on land during the Carboniferous, survived the Permian crisis and continue to flourish today.6, 13
References
The earliest known eurypterid: a chasmataspid-like chelicerate from the Katian (Upper Ordovician) of Ireland
A new pterygotid eurypterid from the Silurian Bertie Formation of Ontario, and a revision of the pterygotid taxa from the Bertie Formation and vicinity
An overview of the Silurian eurypterids of New York, with a description of new material from the Williamsville and Ellicott Creek Breccia members of the Fiddlers Green Formation
Breathing life into fossils: taphonomic studies using micro-CT scanning to reveal eurypterid respiratory adaptations