Overview
- Evolution and the origin of life are two completely different things in science. Evolution explains how life changes after it already exists. The origin of life (called 'abiogenesis') tries to figure out how dead chemicals turned into living cells in the first place.
- Scientists have proven in laboratories that the basic building blocks of life—like the ingredients for proteins and DNA—can form naturally under the harsh conditions of the early Earth.
- There is still a lot to learn about how exactly life started. But scientists today are making huge progress, studying things like underwater volcanoes and self-copying chemicals, moving this from a total mystery into a solvable puzzle of chemistry.
One of the most persistent misunderstandings in public discussions about evolution is mixing up two completely different questions: “How does life change over time?” and “How did life start?” These are separate areas of science.2, 4
Evolution explains how populations of animals and plants change through generation after generation. It only works if life already exists.1 The entirely separate question of how the very first living thing emerged from non-living chemicals is called abiogenesis. Scientists studying this aren't usually looking at fossils. Instead, they study chemistry to figure out how dead molecules managed to organize into something alive.2, 3 A theory like gravity doesn't stop working just because we don't fully understand exactly how the universe was born, and the theory of evolution does not rely on solving the exact mystery of the origin of life.
Two totally different fields
The difference between evolution and abiogenesis isn't just about words. Evolution operates on living things that have DNA, the ability to build proteins, and a way to pass traits down to their kids.1 Abiogenesis asks how something like DNA or protein came to exist in the first place.
In the 1920s, a few visionary scientists proposed an idea: maybe the very early Earth had a totally different atmosphere. In this hot soup of chemicals and water, simple non-living molecules might have clumped together over millions of years to form the very first building blocks of life.5
Creating the building blocks
This “chemical soup” idea was finally tested in a laboratory in 1953 by a young scientist named Stanley Miller.7
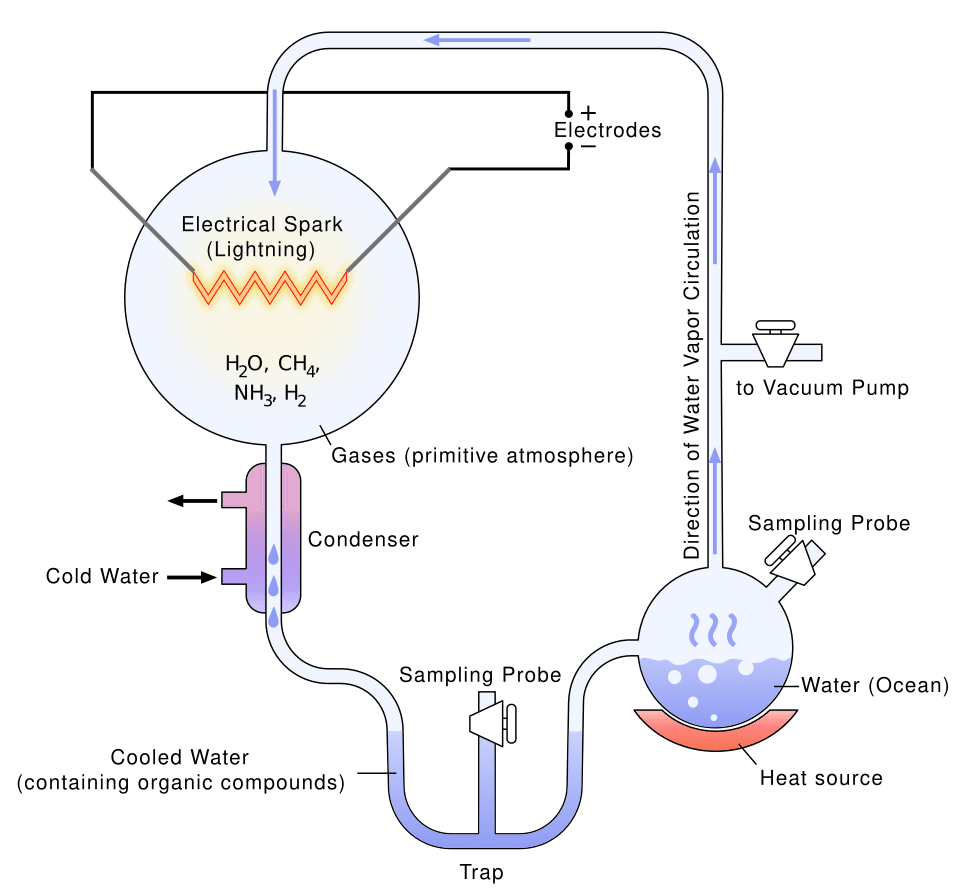
Miller took a sealed glass jar and filled it with water and simple gases that matched the early Earth (like methane and ammonia). He zapped the mixture with electricity to simulate lightning storms. Within a single week, the water turned pink and then deep red. He found that the electricity had transformed the simple gases into amino acids—the basic building blocks that make up all proteins in your body.7
This experiment shocked the world. It proved that you don't need a living creature to make the building blocks of life. They can form naturally from lightning and basic chemistry.3 Years later, scientists found even more forgotten samples from Miller's lab. Using modern, highly sensitive equipment, they realized Miller's lightning experiments actually created over 20 different types of amino acids.8
Since the 1950s, scientists have taken this much further. They have proven that almost all the basic parts of life—sugars, fats, and even the chemical letters that make up DNA and RNA—can form naturally without any living cells to build them.9, 9
Which came first: the chicken or the egg?
One of the biggest hurdles for figuring out the origin of life is a classic “chicken or egg” problem. In modern cells, DNA holds the instructions to build proteins, but proteins are required to read the DNA. So which one came first?10, 11
Scientists proposed a solution called the "RNA World" hypothesis. RNA is a chemical cousin of DNA. Scientists discovered that RNA can actually do both jobs. It can hold genetic instructions like DNA, but it can also fold up and perform jobs in the cell exactly like a protein.10, 11 For this discovery, scientists won the Nobel Prize.12, 13
In 2009, scientists achieved a massive breakthrough: they created a piece of RNA in the lab that actually copied itself. It didn't need any proteins to help it. This self-copying chemical proved that before DNA and complex cells existed, simple chemicals could have reproduced themselves on their own.14
The first simple cells
If a chemical can copy itself, it needs a way to protect those copies so they don't wash away in the ocean. This means the very first "creature" would need a type of skin or membrane to hold its chemicals inside. We call these primitive shells "protocells."15
Research has shown that simple fatty acids naturally form microscopic bubbles when dropped into water. Unlike the complicated walls of modern cells, these fat bubbles assemble themselves entirely naturally. They can stretch, absorb more fat from the water to "grow," and even split in half to "reproduce" if they are squeezed or pushed by waves. Best of all, they can trap self-copying RNA inside of them.15, 16
Underwater volcanoes
So where did all of this chemistry happen? While Charles Darwin once imagined a "warm little pond," modern scientists increasingly point to the bottom of the ocean. At places called alkaline hydrothermal vents, volcanic heat escapes from the earth's crust into the cold ocean water.17

These vents act like giant natural chemistry labs. They provide constant heat and energy, and the porous rocks have millions of microscopic holes that act just like the protective walls of a tiny cell. The swirling chemicals from the Earth's crust mix with ocean water to create electrical energy practically identical to how your own human cells generate energy today. Many scientists believe the very first lifelike chemistry started deep in these vents.18, 19
The oldest ancestor
Scientists aren’t just guessing about this. By comparing the DNA of all the plants, animals, and bacteria alive today, scientists can work backward to figure out what the very first common ancestor must have looked like. We call this ancient organism LUCA (the Last Universal Common Ancestor).20
By tracing the genetic family tree, scientists estimate LUCA lived more than 4 billion years ago.20 Based on the genes that all living things share, researchers believe LUCA was a single-celled organism that survived without oxygen, loved extreme heat, and probably lived exactly in those hot underwater vents. Physical evidence backs this up too—there are traces of chemical signatures in ancient crystals showing life probably existed an incredible 4.1 billion years ago.21
A mystery no more
We don't know the exact story of how life started, and there are still missing pieces to the puzzle. However, scientists are no longer blindly guessing. They have mathematically recreated the DNA of the oldest life, built self-copying chemicals in the lab, generated the building blocks of life with simulated lightning strikes, and watched simple fat bubbles behave like living cells.7, 14, 16
The origin of life is not a magical event that defies explanation. It is an intense, difficult chemical problem, and scientists are finding the chemical answers.2, 4
The chirality puzzle
One of the remaining puzzles in origin-of-life research is homochirality—the fact that biological amino acids are almost exclusively "left-handed" (L-form) while biological sugars are "right-handed" (D-form). Most chemical reactions produce equal mixtures of left- and right-handed molecules, yet life uses only one type. If early life had tried to use a mix of both, the proteins and nucleic acids would not fold properly, and biology as we know it could not function.22
Several mechanisms have been proposed to explain how this asymmetry originated. Breslow and Cheng demonstrated that L-amino acids can catalyse the production of D-sugars from simple precursors, suggesting that a small initial excess of one handedness could amplify itself through autocatalytic feedback. Meteorites such as the Murchison meteorite have been found to contain a slight excess of L-amino acids, raising the possibility that the initial symmetry-breaking event had an extraterrestrial origin, with interstellar processes such as circularly polarised ultraviolet light preferentially destroying one mirror form over the other.22
Systems chemistry: putting the pieces together
Recent work has moved beyond studying individual building blocks in isolation toward "systems chemistry"—the study of how multiple classes of prebiotic molecules could have formed together under a single set of conditions. Patel and colleagues demonstrated in 2015 that precursors to amino acids, nucleotides, and lipids can all be generated from hydrogen cyanide, hydrogen sulfide, and ultraviolet light through a connected network of chemical reactions, suggesting that the building blocks of life did not need to form separately and then somehow find each other.23
Advances in RNA replication have also continued. Attwater and colleagues engineered ribozymes capable of copying RNA templates using short triplet building blocks, achieving significantly greater accuracy and length of copied sequences than earlier systems. While still far from the efficiency of modern enzymes, these experiments demonstrate a plausible pathway from simple self-replicating RNA to more complex catalytic systems capable of sustaining rudimentary Darwinian evolution.24 Taken together, these developments reinforce the view that the origin of life is not a single event requiring miraculous coincidence but a series of chemically reasonable steps, each building on the last, bridging the gap between simple geochemistry and the first self-replicating protocells.2, 23
References
Pioneers of Origin of Life Studies—Darwin, Oparin, Haldane, Miller, Oró—And the Oldest Known Records of Life
Mechanism of Synthesis of Adenine from Hydrogen Cyanide under Possible Primitive Earth Conditions
Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions
Self-splicing RNA: autoexcision and autocyclization of the ribosomal RNA intervening sequence of Tetrahymena
The nature of the last universal common ancestor and its impact on the early Earth system
Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism