Overview
- The earliest fossil evidence for bipedal locomotion in the hominin lineage extends to approximately 7 million years ago with Sahelanthropus tchadensis, whose anteriorly positioned foramen magnum and contested postcranial remains suggest upright posture, while Orrorin tugenensis at 6 million years ago preserves femoral morphology consistent with habitual bipedal ground locomotion.
- The Laetoli footprints, dated to 3.66 million years ago and attributed to Australopithecus afarensis, constitute the earliest direct, unambiguous evidence of human-like bipedal gait, preserving heel strike, a longitudinal arch, and a non-divergent hallux functioning as a propulsive lever.
- Multiple non-exclusive hypotheses explain why bipedalism evolved, including thermoregulatory advantage in open habitats, energetic efficiency demonstrated by the Sockol et al. 2007 finding that human bipedalism costs approximately 75 percent less energy than chimpanzee locomotion, postural feeding in woodland settings, and provisioning of mates and offspring.
The question of when, where, and why hominins first began walking upright on two legs is among the most intensely investigated problems in paleoanthropology. Bipedalism is the defining locomotor adaptation of the human lineage, yet its origins lie deep in the late Miocene, a period from which the fossil record is sparse and the available specimens are frequently fragmentary and contested. Despite these challenges, a succession of discoveries over the past three decades has pushed the earliest evidence for bipedal locomotion back to at least 6 million years ago and possibly 7 million years ago, demonstrating that upright walking preceded brain expansion, stone tool manufacture, and every other hallmark of the genus Homo by millions of years.1, 4
Understanding the origins of bipedalism requires integrating three distinct lines of inquiry: the fossil evidence that documents when and in which species bipedal traits first appear, the biomechanical analyses that reveal how early hominin locomotion differed from that of living apes, and the ecological hypotheses that attempt to explain why natural selection favoured upright walking in the first place. Each of these strands has advanced substantially since the early twenty-first century, producing a picture of bipedal evolution that is more detailed, more complex, and more mosaic in character than earlier models anticipated.6, 23
The earliest cranial evidence: Sahelanthropus tchadensis
The oldest fossil evidence potentially relevant to bipedalism comes from Sahelanthropus tchadensis, described from a nearly complete cranium (TM 266-01-060-1, nicknamed "Toumaï") recovered from the Djurab Desert of northern Chad and dated to approximately 7 million years ago. When Michel Brunet and colleagues announced the find in 2002, they noted that the foramen magnum—the opening at the base of the skull through which the spinal cord passes—was positioned more anteriorly than in any known great ape, a configuration associated with balancing the head atop a vertical spine rather than suspending it in front of a horizontal one.1
A detailed virtual reconstruction of the Toumaï cranium by Zollikofer and colleagues in 2005 confirmed that the foramen magnum orientation was closer to the human condition than to that of chimpanzees, even after correcting for taphonomic distortion of the original specimen. The reconstructed angle between the foramen magnum and the orbital plane fell closer to values typical of Homo sapiens than to those of australopiths, supporting the inference that Sahelanthropus held its head in a manner consistent with an upright posture.2 However, the foramen magnum position alone cannot confirm habitual bipedalism, because the relationship between foramen magnum angle and locomotor mode shows substantial overlap among extant primates, and postcranial evidence is required for a definitive assessment.2
In 2022, Daver and colleagues described postcranial material associated with Sahelanthropus—a left femur and two ulnae from the same locality—and argued that the femoral morphology was most parsimonious with habitual bipedality, while the ulnar morphology indicated substantial arboreal capability.3 Their interpretation suggested that hominins were already combining bipedal ground locomotion with arboreal clambering at approximately 7 million years ago. However, this interpretation has been contested: subsequent analyses by other researchers concluded that the femoral morphology does not unambiguously support habitual bipedalism and may instead reflect quadrupedal or climbing adaptations comparable to those of Miocene apes.3 The locomotor status of Sahelanthropus therefore remains an active area of debate, with the anteriorly positioned foramen magnum representing the most widely accepted, though still inconclusive, indicator of upright posture in this taxon.
Orrorin tugenensis and the 6-million-year-old femur
More compelling postcranial evidence for early bipedalism comes from Orrorin tugenensis, discovered in the Tugen Hills of central Kenya by Brigitte Senut and Martin Pickford in 2000. The original description, published in 2001, reported more than a dozen fossil fragments dated to approximately 6 million years ago, including two partial femora that would become central to debates about the antiquity of bipedal locomotion.4
The femoral morphology of Orrorin displays several features associated with bipedal hip biomechanics. The femoral neck is elongated and anteroposteriorly compressed, with cortical bone that is thicker on the inferior surface than the superior surface—a distribution pattern that reflects the bending forces generated when the full body weight passes through a single hip joint during the stance phase of bipedal walking. An obturator externus groove on the posterior femoral neck and a well-developed gluteal tuberosity on the proximal shaft further suggest adaptations for bipedal muscle function.4, 5
A comprehensive morphometric analysis by Richmond and Jungers in 2008 confirmed that the Orrorin femur differs substantially from those of both extant apes and Homo, and most closely resembles the femora of Australopithecus and Paranthropus. This finding indicated that Orrorin was indeed bipedal but shared distinctive hip biomechanics with the australopiths, suggesting that this locomotor complex evolved early in hominin evolution and persisted for nearly four million years before modifications appeared in early Homo during the late Pliocene.5 The Orrorin evidence thus establishes that some form of habitual bipedal ground locomotion was present in the hominin lineage by at least 6 million years ago, substantially earlier than the australopith radiation.
Ardipithecus ramidus and the mosaic of facultative bipedalism
The most informative early hominin for understanding the transitional stage between ape-like locomotion and committed bipedalism is Ardipithecus ramidus, represented by the partial skeleton ARA-VP-6/500 ("Ardi"), dated to approximately 4.4 million years ago. When White and colleagues published the comprehensive description in 2009, they revealed a locomotor anatomy unlike that of any living primate: the pelvis showed a distinctly broadened, laterally flared ilium consistent with bipedal gluteal function, yet the foot retained a fully opposable, divergent great toe capable of grasping branches.6, 7

Lovejoy and colleagues interpreted this combination as evidence for a novel form of locomotion—bipedal on the ground but with powerful above-branch arboreal capability. They argued that the lateral toes, rather than the divergent hallux, provided ground push-off during terrestrial walking, and that Ardipithecus represents a locomotor stage in which pelvic remodeling for bipedalism had already occurred while the foot retained a grasping function for climbing.7 This discovery overturned earlier assumptions that foot specialization for bipedalism necessarily preceded or accompanied the pelvic changes, demonstrating instead that the bipedal body plan was assembled in a piecemeal, mosaic fashion over millions of years.6, 7
The environmental context of Ardipithecus is equally significant. Paleoenvironmental reconstructions of the Aramis locality indicate that Ar. ramidus lived in a woodland habitat rather than an open savanna, challenging earlier models that linked the origin of bipedalism exclusively to grassland expansion.6 The Ardipithecus evidence suggests that bipedalism first evolved not as an adaptation for traversing open landscapes but as a locomotor strategy used in woodland environments, a finding with implications for evaluating the various hypotheses about why bipedalism evolved.
The Laetoli footprints: direct evidence at 3.66 million years
The most direct and unambiguous evidence for bipedal locomotion in any fossil species is not a bone but a set of footprints. In 1978, Mary Leakey's team excavating at Laetoli in northern Tanzania uncovered hominin footprint trails preserved in volcanic ash that radiometric methods dated to approximately 3.66 million years ago. Two trails side by side—designated G1 and G2/3—extended for approximately 27 metres and recorded the tracks of at least two individuals walking in the same direction.8 Leakey and Hay described them in 1979 as unmistakably human-like in their overall morphology, noting the absence of any knuckle impressions and the alignment of all five toes pointing forward.8
Experimental biomechanical analysis has confirmed several hallmarks of modern bipedal gait in the Laetoli prints. Raichlen and colleagues, using a comparative design in which human subjects walked with both extended-limb and bent-knee gaits across pressure-sensitive surfaces, found that the weight distribution pattern in the Laetoli footprints most closely matches that produced by economical, extended-limb bipedalism rather than the bent-knee, bent-hip gait characteristic of chimpanzees walking upright. The prints preserve evidence of heel strike, a medial longitudinal arch, and weight transfer from the lateral midfoot to the hallux at toe-off—a sequence that mirrors the mechanics of modern human walking.9 These results provide the earliest direct evidence of kinematically human-like bipedalism currently known and demonstrate that extended-limb bipedalism evolved long before the appearance of the genus Homo.9
Subsequent discoveries at Laetoli have enriched the picture of locomotor variation among the trackmakers. In 2016, Masao and colleagues reported additional footprints at Laetoli Site S that revealed marked body size variation, consistent with the pronounced sexual dimorphism documented in Australopithecus afarensis skeletal material.24 In 2021, McNutt and colleagues re-excavated the enigmatic Site A prints, originally discovered in 1976 and long dismissed as bear tracks, and demonstrated through detailed comparative analysis that they were produced by a bipedal hominin employing a distinctive cross-stepping gait different from the locomotion recorded at Site G. This finding established that at least two different bipedal gaits were present among hominins at Laetoli 3.66 million years ago, raising the possibility that more than one hominin species inhabited the landscape simultaneously.10 Because A. afarensis is the only hominin species known from contemporaneous deposits at Laetoli, the Site G prints are universally attributed to that species.8, 9
Lucy, the Dikika child, and obligate bipedalism in Australopithecus afarensis
The skeletal evidence for committed, obligate bipedalism in the hominin lineage reaches its fullest Pliocene expression in Australopithecus afarensis. The partial skeleton AL 288-1, discovered by Donald Johanson at Hadar, Ethiopia, in 1974 and popularly known as "Lucy," combines a suite of features that leave no doubt about habitual bipedal locomotion: a short, broad, laterally flared ilium, a femur angled inward toward the knee with a valgus angle within the modern human range, a forward-positioned foramen magnum, and robust bipedal knee anatomy.11 At approximately 3.2 million years old and preserving roughly 40 percent of the skeleton, Lucy provided the first comprehensive view of an early hominin body plan built for upright walking. Her brain, however, was scarcely larger than a chimpanzee's, confirming that bipedalism predated encephalization by millions of years.11
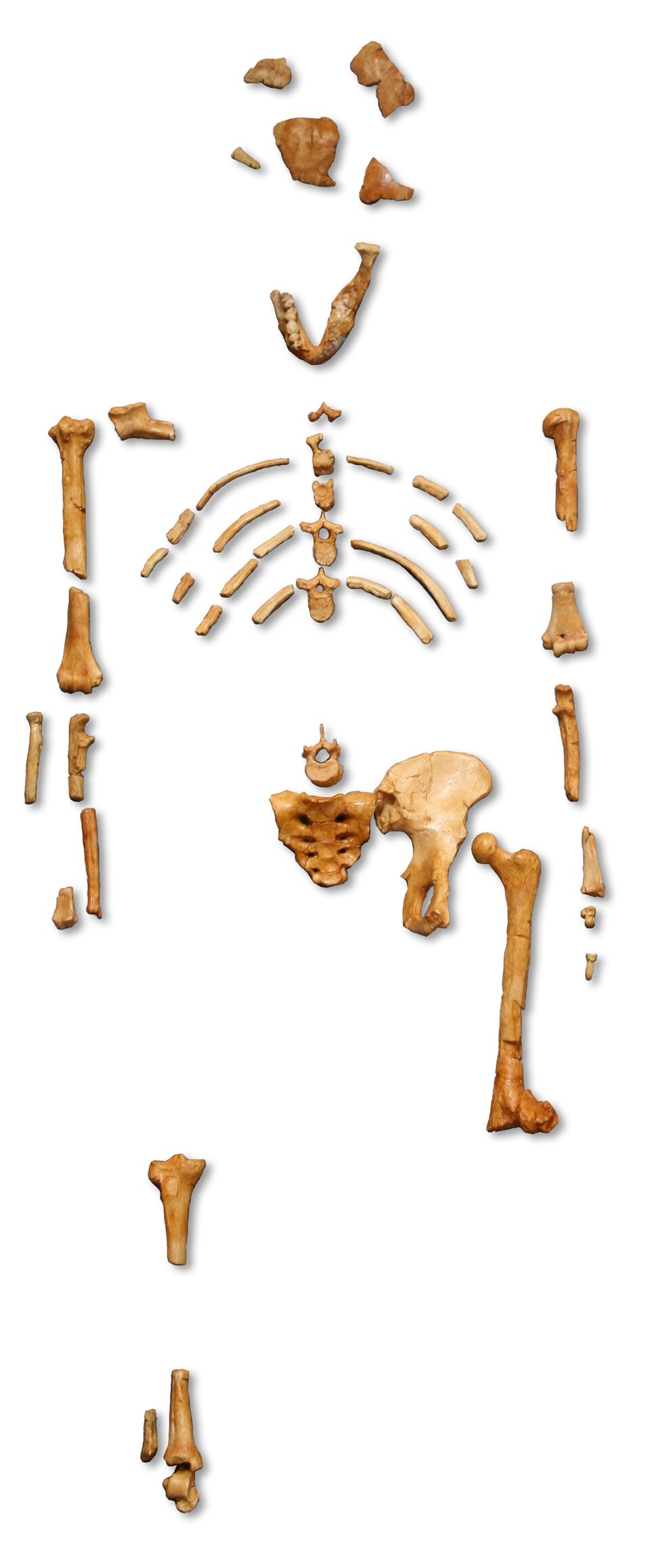
The foot of A. afarensis displays adaptations consistent with efficient terrestrial bipedalism. Latimer and Lovejoy's 1989 analysis of the calcaneus from the Hadar assemblage showed a large, robust calcaneal tuberosity—a heel-strike platform comparable to that of modern humans—along with a stiffened midfoot indicative of a longitudinal arch capable of storing and returning elastic energy during the gait cycle.21 The great toe in A. afarensis is adducted (non-divergent), meaning it was aligned with the other toes rather than being opposable as in Ardipithecus, and functioned as a propulsive lever during toe-off.21
The discovery of the Dikika child (DIK-1-1) by Zeresenay Alemseged in 2000, described in 2006, extended the anatomical evidence to a juvenile individual approximately 3.3 million years old. The remarkably complete skeleton of this roughly three-year-old A. afarensis child preserved lower-limb elements confirming bipedal locomotion, but the gorilla-like scapula and long, curved manual phalanges raised questions about the continued importance of arboreal behaviour in the species' locomotor repertoire.12 A subsequent detailed analysis of the Dikika child's nearly complete foot by DeSilva and colleagues in 2018 revealed that juvenile A. afarensis individuals already possessed many adult bipedal features but also retained increased hallucal mobility and a more gracile calcaneal tuber than expected from adult morphology, suggesting that young A. afarensis relied on pedal grasping to a greater degree than adults.13 This finding implies that the ontogeny of locomotion in early hominins was unique: juveniles likely spent more time in the trees than their parents, gradually transitioning to fully terrestrial bipedalism as they matured.
Anatomical foundations of bipedal locomotion
The transition from quadrupedal or arboreal locomotion to habitual bipedalism required coordinated modification of virtually every region of the postcranial skeleton. These changes did not appear simultaneously but accumulated in a mosaic pattern across multiple hominin species and millions of years. Several of the most consequential modifications are now well documented in the fossil record and provide the diagnostic criteria by which paleoanthropologists evaluate locomotor behaviour in fragmentary specimens.7, 22
The repositioning of the foramen magnum from a posterior location, as in quadrupedal apes whose spine extends horizontally from the skull, to an anteroinferior position beneath the braincase is among the earliest detectable cranial signatures of upright posture. In a biped, the head is balanced atop a vertical spine with minimal muscular effort, and the foramen magnum sits centrally beneath the skull rather than at its rear. This trait is observable in Sahelanthropus at 7 million years ago and is progressively more pronounced in later hominins.1, 2
The pelvis underwent what is arguably the most dramatic transformation. In apes, the ilium is tall, narrow, and oriented posteriorly, directing the gluteal muscles toward hip extension during quadrupedal locomotion. In bipeds, the ilium is shortened, broadened, and laterally flared, repositioning the gluteus medius and gluteus minimus as abductors that prevent the trunk from collapsing laterally during the single-leg stance phase of walking.7 This remodeling is already evident in the pelvis of Ardipithecus ramidus at 4.4 million years ago, establishing that the pelvic changes for bipedalism preceded the loss of the grasping foot.7
The femur developed an inward angulation toward the knee known as the valgus, or bicondylar, angle—approximately 8 to 11 degrees in modern humans. This angulation positions the knees beneath the body's centre of mass during the swing phase of gait, permitting a smooth, efficient stride rather than the lateral body sway that chimpanzees exhibit when walking upright. Tardieu and Trinkaus demonstrated that the bicondylar angle in modern humans develops postnatally in direct response to bipedal loading, starting at zero degrees at birth and reaching adult values between ages four and eight.22 In the fossil record, a pronounced valgus angle is present in Australopithecus afarensis (Lucy, AL 288-1), falling well within the modern human range and far outside the ape range, providing one of the most reliable osteological confirmations of habitual bipedalism.11, 22
The lumbar spine developed a lordotic (forward) curvature entirely absent in apes, which centres the upper body's mass above the pelvis and reduces the muscular effort required to maintain an upright trunk. The foot lost its opposable great toe and developed longitudinal and transverse arches that function as spring mechanisms, storing elastic energy during the stance phase and releasing it during toe-off. The ankle joint was reoriented for stable weight transmission through a compact mortise joint, a configuration that DeSilva's 2009 kinematic study demonstrated may have constrained the degree of dorsiflexion available during vertical climbing, creating a biomechanical trade-off between bipedal efficiency and arboreal capability.20
Key anatomical changes for bipedalism and their earliest fossil appearance1, 5, 7, 11, 21
| Anatomical feature | Bipedal condition | Earliest fossil evidence | Approximate date |
|---|---|---|---|
| Anterior foramen magnum | Head balanced atop vertical spine | Sahelanthropus tchadensis | ~7 Ma |
| Femoral cortical bone distribution | Thick inferior cortex resists bipedal bending | Orrorin tugenensis | ~6 Ma |
| Shortened, flared ilium | Gluteal muscles reoriented as abductors | Ardipithecus ramidus | ~4.4 Ma |
| Non-divergent hallux | Great toe aligned for push-off | Australopithecus afarensis | ~3.7 Ma |
| Longitudinal foot arch | Elastic energy storage and return | Australopithecus afarensis | ~3.2 Ma |
| Valgus (bicondylar) angle | Knees centred beneath body mass | Australopithecus afarensis | ~3.2 Ma |
| Lumbar lordosis | Upper body centred over pelvis | Australopithecus africanus | ~2.5 Ma |
The arboreal-to-terrestrial transition
A central question in the study of bipedal origins concerns the locomotor mode that immediately preceded habitual upright walking. For much of the twentieth century, the dominant view held that the last common ancestor of hominins and African apes was a knuckle-walker, and that bipedalism evolved directly from knuckle-walking quadrupedalism. This model gained apparent support from Richmond and Strait's 2000 report that wrist bones of Australopithecus anamensis and A. afarensis retained specialized morphology associated with knuckle-walking, suggesting a relatively recent transition from this locomotor mode.23
The discovery of Ardipithecus ramidus profoundly challenged this scenario. The hands and wrists of Ar. ramidus lack the specialized knuckle-walking features of chimpanzees and gorillas, and Lovejoy and colleagues argued that the Ardipithecus body plan was more consistent with an ancestor that practised careful above-branch climbing and palmigrade quadrupedalism rather than knuckle-walking. On this interpretation, knuckle-walking evolved independently in chimpanzees and gorillas after the human-ape divergence, and bipedalism did not derive from a knuckle-walking ancestor at all.6, 18 A comprehensive 2021 review by Almécija and colleagues confirmed that the morphological diversity of Miocene fossil apes supports no single model of the ancestral locomotor condition, concluding that the last common ancestor of humans and African apes likely had its own distinctive suite of locomotor traits, different from those of any living species.23
The kinematic evidence further complicates a simple arboreal-to-terrestrial narrative. DeSilva's 2009 study of ankle joint kinematics during vertical climbing in wild chimpanzees showed that apes achieve up to 45 degrees of dorsiflexion at the ankle during ascent, far exceeding the range available to humans, whose ankle morphology is optimized for stable bipedal weight transmission. The talocrural joint surface of early hominins such as A. afarensis appears to have already been remodeled for bipedal force distribution, potentially limiting the degree of ape-like vertical climbing available to these species.20 This biomechanical trade-off suggests that the adoption of bipedalism progressively constrained arboreal capability, even as features such as curved phalanges indicate that some degree of climbing persisted alongside bipedal locomotion in early hominins for millions of years.
Why bipedalism evolved: competing and complementary hypotheses
Identifying when bipedalism arose is a different problem from explaining why natural selection favoured it. Several major hypotheses have accumulated substantial empirical support, and most researchers now recognize that multiple selective pressures likely operated simultaneously or sequentially across different ecological contexts.
The thermoregulatory hypothesis, developed by Peter Wheeler in 1991, proposes that bipedalism evolved as an adaptation to the thermal challenges of foraging in sun-exposed habitats. Wheeler's heat-balance models demonstrated that an upright biped exposes roughly 60 percent less body surface area to direct overhead solar radiation compared with a horizontal quadruped of equivalent mass. Standing upright also elevates the body into faster-moving air currents above the ground boundary layer, increasing convective heat loss and the efficiency of evaporative cooling through sweating.15 A companion analysis showed that under all temperature regimes examined, bipedalism would allow a hominin to forage at higher ambient temperatures and over greater distances while consuming less water than a quadruped of comparable size.16 The hypothesis aligns with evidence for expanding woodland and savanna-mosaic habitats in East Africa during the late Miocene, though the Ardipithecus evidence from closed woodland environments suggests that thermoregulatory advantages may have been a secondary reinforcing factor rather than the initial selective pressure.6
The energetic efficiency hypothesis received its most rigorous test in the landmark 2007 study by Sockol, Raichlen, and Pontzer, who measured the metabolic cost of bipedal and quadrupedal locomotion in chimpanzees and compared it directly with human bipedal walking. They found that human bipedalism costs approximately 75 percent less energy per unit distance than chimpanzee bipedal walking and approximately 75 percent less than chimpanzee knuckle-walking.14 The critical variable explaining this difference was the degree of limb extension: humans walking with extended hips and knees employ an inverted-pendulum mechanism that recovers mechanical energy by exchanging kinetic and potential energy at each step, dramatically reducing the muscular work required. Chimpanzees, whose anatomy prevents full limb extension, cannot exploit this mechanism and must perform substantially more muscular work per stride.14 Pontzer and colleagues extended this work in 2014 by demonstrating that individual chimpanzees with hip anatomy closer to the human condition—specifically, a more lateral acetabulum and longer femoral neck—incur lower metabolic costs during bipedal walking, establishing a direct mechanistic link between the hip modifications documented in the hominin fossil record and the energetic advantages of bipedalism.25
Relative locomotor cost: human bipedalism versus chimpanzee gaits14
The provisioning hypothesis, advanced by C. Owen Lovejoy in 1981, proposed that bipedalism evolved as part of a reproductive strategy in which males freed their forelimbs to carry food to females and offspring. On this model, the primary selective pressure was not locomotor efficiency per se but the increased reproductive success conferred by pair-bonding and male parental investment, with bipedalism facilitating food transport as a critical component.17 Lovejoy extended and refined this model in 2009, incorporating the Ardipithecus evidence to argue that reduced canine sexual dimorphism and bipedalism evolved together as components of a suite of traits associated with diminished male-male competition and increased paternal investment.18
The postural feeding hypothesis, proposed by Kevin Hunt in 1994, offers a distinctly different ecological scenario. Based on 700 hours of observation of wild chimpanzees, Hunt documented that bipedal postures in chimpanzees occur overwhelmingly in the context of feeding—specifically, standing upright to reach food items in low trees and shrubs, often while stabilising with one hand on an overhead branch. He argued that selection initially favoured bipedal postural feeding in a woodland-adapted hominin, with habitual terrestrial bipedalism emerging secondarily as populations moved between feeding patches.19 This hypothesis aligns well with the Ardipithecus evidence for bipedalism in woodland environments and with the mosaic anatomy of early hominins that combined bipedal ground locomotion with persistent arboreal capacity.6, 19
None of these hypotheses is mutually exclusive. The energetic advantages of extended-limb walking, the thermoregulatory benefits of an upright posture, the reproductive advantages of provisioning, and the feeding efficiency of upright posture in woodland settings may each have contributed selective pressure at different stages of the transition and in different ecological contexts. The emerging consensus treats bipedalism not as the product of a single selective force but as a complex adaptation shaped by multiple reinforcing pressures acting across several million years of hominin evolution.14, 15, 17
The mosaic nature of early locomotor evolution
Perhaps the most significant insight to emerge from the past two decades of research on bipedal origins is that the transition from arboreal or quadrupedal locomotion to obligate bipedalism was not a single discrete event but a prolonged, mosaic process in which different components of the bipedal body plan were acquired at different times and in different combinations across multiple hominin lineages. Sahelanthropus at 7 million years ago may have had an anteriorly positioned foramen magnum consistent with upright head carriage while retaining a postcranial skeleton adapted primarily for arboreal or quadrupedal locomotion.1, 3 Orrorin at 6 million years ago possessed femoral features associated with bipedal hip mechanics while presumably retaining substantial climbing ability.4, 5 Ardipithecus at 4.4 million years ago had a pelvis remodeled for bipedal gluteal function alongside a fully opposable great toe for arboreal grasping.6, 7 Australopithecus afarensis by 3.7 to 3.2 million years ago had committed to bipedalism with a non-divergent hallux, a longitudinal arch, and a human-like valgus angle, yet still retained curved phalanges and shoulder morphology suggesting that climbing remained part of the behavioural repertoire.11, 12
This mosaic pattern has two important implications. First, it demonstrates that bipedalism did not arise as a single coordinated package but was assembled gradually, with different selective pressures acting on different body regions at different times. The pelvis was restructured before the foot lost its grasping function; the femoral neck was adapted for bipedal loading before the knee developed its full valgus angulation; the foramen magnum may have shifted forward before any postcranial changes occurred.7, 23 Second, it indicates that for millions of years, early hominins were not exclusively bipedal in the way modern humans are. They practised what is best described as facultative bipedalism—walking upright on the ground as their primary terrestrial locomotor mode while retaining significant capacity for arboreal locomotion. The shift from facultative to obligate bipedalism was itself a drawn-out process, with the latest persistent arboreal features disappearing only in the genus Homo.12, 20
The locomotor diversity documented at Laetoli, where at least two different bipedal gait patterns are preserved in 3.66-million-year-old volcanic ash, reinforces the picture of a hominin lineage experimenting with multiple variations on the bipedal theme rather than converging rapidly on a single optimal solution.10 The origins of bipedalism, in this view, are not a moment but a process—one that unfolded across at least four million years of late Miocene and Pliocene hominin evolution and that established the anatomical and ecological foundation upon which every subsequent chapter of human evolution was built.
References
Laetoli footprints preserve earliest direct evidence of human-like bipedal biomechanics
A nearly complete foot from Dikika, Ethiopia and its implications for the ontogeny and function of Australopithecus afarensis
The thermoregulatory advantages of hominid bipedalism in open equatorial environments
The calcaneus of Australopithecus afarensis and its implications for the evolution of bipedality
New footprints from Laetoli (Tanzania) provide evidence for marked body size variation in early hominins