Overview
- Amber is fossilized tree resin that preserves organisms in extraordinary three-dimensional detail through a process of polymerization and cross-linking of terpenoid compounds, with major deposits spanning from the Cretaceous to the Eocene across Baltic, Dominican, Burmese, Lebanese, and New Jersey localities.
- Inclusions in amber have revealed feathered dinosaur tails, the oldest known arthropods trapped in resin from 230 million years ago, and over 3,000 species of Eocene insects from Baltic deposits alone, providing unparalleled windows into ancient ecosystems and biodiversity.
- Despite popular fiction, DNA does not survive in amber even over thousands of years, and the scientific study of Burmese amber is complicated by serious ethical concerns over mining practices linked to armed conflict and human rights abuses in Myanmar's Kachin State.
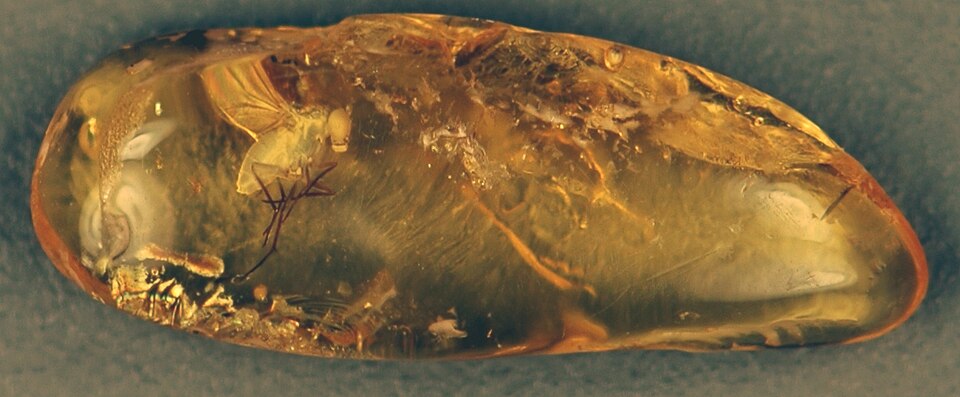
Amber is fossilized tree resin that preserves organisms and organic material in extraordinary three-dimensional detail, often retaining microscopic structures such as individual setae on insect legs, cellular architecture in plant tissues, and even the barbs and barbules of feathers. Unlike compression fossils in sedimentary rock, which flatten organisms into two dimensions, amber entombs specimens in a transparent matrix that arrests decay and maintains spatial relationships among body parts with a fidelity unmatched by any other mode of fossilization.1, 2 The study of amber and its inclusions has transformed understanding of ancient terrestrial ecosystems, revealing the diversity and ecology of organisms — particularly arthropods — that are rarely preserved through conventional taphonomic pathways. Major deposits in the Baltic region, the Dominican Republic, Myanmar, Lebanon, and New Jersey span more than 100 million years of Earth history and collectively contain hundreds of thousands of fossil specimens.5
Formation and chemistry of amber
Amber begins as resin, a viscous liquid secreted by certain trees — principally conifers and some angiosperms — as a response to injury, infection, or insect attack. Fresh resin is a complex mixture of terpenoid compounds, including volatile monoterpenoids and sesquiterpenoids that evaporate relatively quickly, and more stable diterpenoids and triterpenoids that form the structural backbone of the material.1, 2 The transformation from resin to amber is a multi-stage process spanning millions of years. In the initial phase, volatile components evaporate and the resin hardens into an intermediate substance called copal. Over geological time, the remaining terpenoid molecules undergo progressive polymerization and cross-linking, forming a dense, three-dimensional polymer network that is chemically stable and resistant to most solvents.2, 14
The chemical maturation of resin involves several concurrent reactions, including isomerization, cyclization, and the formation of ester and ether cross-links between diterpenoid molecules. Infrared spectroscopy of amber at different stages of maturation reveals a progressive reduction in the carbonyl (C=O) absorption peak near 1,700 cm−1, reflecting the consumption of free carboxylic acid groups during cross-linking reactions.3, 14 Experimental studies have replicated aspects of this process by heating and pressurizing fresh pine resin encased in sediment, producing a hardened, translucent material whose spectral signature closely resembles that of natural Eocene Baltic amber, confirming that heat, pressure, and time are the primary drivers of amberization.3 The chemical class of the resulting amber depends on the botanical source of the original resin. Baltic amber, known mineralogically as succinite, is characterized by a high succinic acid content (3–8 percent by weight) and is derived from the resin of an extinct conifer, possibly related to the living genus Sciadopitys.1, 21
Major amber deposits of the world
Amber deposits are distributed unevenly across the geological record, with the greatest volumes concentrated in the Cretaceous and Eocene periods, an interval that has been termed the "Cretaceous resinous interval" owing to a dramatic increase in resin production by forests worldwide.7 Each major deposit preserves a distinct snapshot of the terrestrial biota of its age and geographic region.
Baltic amber constitutes the largest known amber deposit on Earth. Found primarily in upper Eocene marine sediments along the shores of the Baltic Sea in present-day Russia (Kaliningrad), Poland, Lithuania, Latvia, and northern Germany, Baltic amber is approximately 34 to 48 million years old and records the fauna and flora of a warm-temperate to subtropical forest ecosystem sometimes called the "Baltic amber forest."15, 21 More than 3,000 species of arthropods and approximately 200 species of plants have been described from Baltic amber, making it the single richest repository of fossil insects of any age.15, 18
Dominican amber originates from Miocene deposits (approximately 15–20 million years old) in the northern mountain ranges of the Dominican Republic. Produced by the leguminous tree Hymenaea protera, this amber preserves an exceptionally diverse tropical forest fauna, including ants, termites, beetles, spiders, and rare vertebrate inclusions such as lizards and frogs.5 Its relative youth and optical clarity have made Dominican amber one of the most intensively studied deposits for ecological and biogeographic analyses of Neotropical arthropod communities.
Burmese amber (burmite) from the Hukawng Valley of Kachin State in northern Myanmar has emerged as one of the most scientifically important amber deposits of the twenty-first century. Radiometrically dated to approximately 99 million years ago (Cenomanian, mid-Cretaceous), burmite preserves organisms from the age of dinosaurs, including an astonishing diversity of insects, arachnids, crustaceans, molluscs, plants, fungi, and even fragments of vertebrates.9, 13 The scientific significance of this deposit is complicated by serious ethical concerns surrounding its extraction, discussed below.
Lebanese amber, dating to approximately 125–130 million years ago (Barremian–Aptian, Early Cretaceous), represents one of the oldest deposits containing abundant biological inclusions. Lebanese amber has yielded some of the earliest known representatives of several insect orders and families, providing critical data for calibrating the timing of arthropod diversification during the Cretaceous terrestrial revolution.17
New Jersey amber from the Turonian (approximately 90 million years ago) Raritan Formation has produced a rich assemblage of Cretaceous arthropods, including the oldest known ant (Sphecomyrma), parasitic wasps, moths, and diverse Diptera, documenting the ecological complexity of Cretaceous forests in what is now eastern North America.19
Major amber deposits and their characteristics5, 7
| Deposit | Age | Period | Resin source | Notable inclusions |
|---|---|---|---|---|
| Baltic (Europe) | ~34–48 Ma | Eocene | Conifer (Sciadopitys-related) | 3,000+ arthropod species |
| Dominican Republic | ~15–20 Ma | Miocene | Hymenaea protera | Tropical insects, lizards, frogs |
| Burmese (Myanmar) | ~99 Ma | Cretaceous | Conifer (Araucariaceae?) | Feathered dinosaur tail, diverse arthropods |
| Lebanese | ~125–130 Ma | Cretaceous | Conifer | Earliest insect families |
| New Jersey (USA) | ~90 Ma | Cretaceous | Conifer (Cupressaceae?) | Oldest ant, diverse Diptera |
Exceptional preservation in amber
The preservation of organisms in amber is among the most remarkable taphonomic processes known. When an organism becomes mired in fresh resin, the viscous material flows around and encapsulates it, sealing it from the external environment. As the resin hardens and undergoes polymerization, the organism is entombed in an airtight, chemically inert matrix that inhibits microbial decomposition and oxidation.1, 2 The result is preservation of a quality that is rarely achieved through any other fossilization pathway: external morphology is retained down to the micrometer scale, including surface textures, wing venation, compound eye facets, and fine sensory hairs. In some cases, internal soft tissues such as muscles, tracheae, and alimentary canals are also preserved as desiccated remains within the exoskeleton.5, 16

Insects and arachnids constitute the vast majority of amber inclusions by both abundance and diversity, owing to their small body size and ecological association with tree habitats. However, amber has also preserved a far broader range of organisms, including nematodes, annelids, gastropods, crustaceans (such as woodlice and amphipods), myriapods, feathers, plant fragments, fungi, mosses, liverworts, bacteria, and even small vertebrates including lizards, frogs, and bird wings.5, 13 Among the most spectacular discoveries from Burmese amber was a 36.7-millimetre section of a feathered dinosaur tail, described in 2016 by Xing and colleagues. The specimen, from a small coelurosaur, preserved chestnut-brown plumage with a white underside and revealed primitive feather architecture with a poorly defined rachis, providing direct evidence of feather structure in a non-avian dinosaur.8
Amber also captures ecological interactions frozen in time: parasites attached to hosts, insects carrying pollen grains, predators in the act of seizing prey, and mating pairs locked in copula. These "behavior snapshots" provide evidence for biological interactions that leave no trace in the conventional fossil record, making amber an unparalleled source of paleoecological information.5, 6
The oldest amber inclusions
The oldest known amber with biological inclusions dates to the Late Triassic (Carnian stage, approximately 230 million years ago) and was recovered from outcrops in the Dolomite Alps of northeastern Italy. Described by Schmidt and colleagues in 2012, this amber contains a nematoceran fly (order Diptera) and two species of mites (Acari), extending the record of arthropods preserved in amber by approximately 100 million years beyond the previous oldest examples from the Early Cretaceous.4 The discovery demonstrated that resin-producing trees were already generating sufficient quantities of exudate to entomb small arthropods in the Triassic, long before the major radiation of resin-producing conifers and angiosperms in the Cretaceous.
Amber itself, without biological inclusions, is known from even older deposits. The oldest confirmed amber comes from Carboniferous sediments approximately 320 million years old in Illinois, produced by early gymnosperms, although these early resins are found only as trace quantities and have not yielded organismal inclusions.7 A dramatic increase in both the volume and global distribution of amber occurs in the Early to mid-Cretaceous, beginning around 130 million years ago, coinciding with the diversification of resin-producing conifer families and the initial radiation of flowering plants. This "Cretaceous resinous interval" produced the vast majority of amber deposits that contain biological inclusions, including the Lebanese, Burmese, New Jersey, and numerous smaller deposits from Spain, France, Canada, and elsewhere.7
Amber and ancient ecosystems

Because amber preferentially traps small, flying or arboreal organisms in and around forest canopies, it provides a sampling window into a habitat zone that is otherwise nearly invisible in the fossil record. Compression fossils in lacustrine or fluvial sediments tend to preserve organisms associated with aquatic or riparian environments, but amber captures the inhabitants of bark, leaf litter, epiphytic vegetation, and the aerial column within and above the forest canopy.6 Statistical analyses of insect diversity in amber deposits have been used to estimate the taxonomic composition and ecological structure of ancient forests, revealing that many modern insect families and ecological guilds — including herbivores, predators, parasitoids, pollinators, and detritivores — were already well established by the mid-Cretaceous.6, 9
Baltic amber provides the most comprehensive picture of any single ancient forest ecosystem. The more than 3,000 described arthropod species include representatives of nearly every order of insects, along with spiders, pseudoscorpions, harvestmen, mites, and crustaceans.15 The botanical composition of the "Baltic amber forest" has been reconstructed from plant inclusions and pollen, revealing a mixed conifer-angiosperm forest dominated by Cupressaceae, Pinaceae, and early representatives of oak (Fagaceae), with an understorey of ferns, mosses, and liverworts in a humid, warm-temperate climate analogous to the modern forests of southeastern China or the southern Appalachian Mountains.21 Burmese amber, being Cretaceous in age, captures a fundamentally different world: a tropical or subtropical forest in which angiosperms were still in the early stages of their ecological rise and conifers of the family Araucariaceae likely produced the resin. The arthropod fauna includes lineages with Gondwanan affinities, reflecting Myanmar's position on a tectonic block that had recently rifted from the southern supercontinent.9, 13
Amber and ancient DNA
The 1993 film Jurassic Park popularized the idea that DNA could be extracted from blood-feeding insects preserved in amber and used to reconstruct the genomes of extinct organisms, including dinosaurs. This scenario was inspired by a series of studies published in the early 1990s that reported the successful extraction and sequencing of DNA from insects in amber up to 130 million years old. However, these results could not be replicated by independent laboratories, and subsequent analysis revealed that the sequences were almost certainly derived from modern contaminant DNA rather than ancient endogenous molecules.10
The definitive refutation came in 2013 when Penney and colleagues at the University of Manchester applied next-generation sequencing — far more sensitive than the methods available in the 1990s — to stingless bees preserved in Colombian copal ranging from 60 to 10,600 years old. Despite the relative youth of these specimens and the use of state-of-the-art sequencing technology, the team was unable to recover any authentic ancient DNA from the inclusions. A tiny quantity of DNA recovered from the youngest specimen (60 years old) was identified as bacterial contamination, not endogenous insect DNA.10 The authors concluded that resin entombment does not confer any special advantage for DNA preservation and that "the Jurassic Park scenario must remain in the realms of fiction."
Independent studies of DNA decay kinetics have established that even under ideal preservation conditions (cold, dry, stable temperatures), DNA undergoes hydrolytic and oxidative damage that fragments molecules into unreadable pieces within approximately 1 million years, with a half-life for the longest recoverable fragments estimated at roughly 521 years for a 242-base-pair sequence at 13°C.20 The warm, tropical conditions under which most amber deposits formed would accelerate degradation further. The oldest authenticated ancient DNA recovered to date comes from permafrost sediments, not amber, and even the most optimistic projections place an absolute upper limit for DNA survival well short of the 66-million-year minimum required to reach the age of non-avian dinosaurs.20
CT scanning and synchrotron imaging
Advances in X-ray imaging have revolutionized the study of amber inclusions over the past two decades. Conventional micro-computed tomography (micro-CT) uses laboratory X-ray sources to generate three-dimensional reconstructions of specimens still encased in amber, eliminating the need for destructive preparation techniques such as cutting and polishing that risk damaging delicate inclusions. Micro-CT is particularly well suited to amber because the density contrast between the organic inclusion and the surrounding resin matrix produces excellent image quality, revealing external morphology and, in many cases, internal anatomical structures including muscles, tracheal systems, and gut contents.16
A significant limitation of conventional micro-CT is that it struggles with opaque amber, a common condition in Mesozoic deposits where the resin has been darkened by diagenetic alteration or densely packed microscopic bubbles. A substantial proportion of Cretaceous amber is fully opaque, rendering biological inclusions invisible to the naked eye even after polishing and thereby introducing a potential bias into paleoecological surveys that rely on visual screening.11 Propagation phase-contrast X-ray synchrotron imaging, developed at facilities such as the European Synchrotron Radiation Facility (ESRF) in Grenoble, France, has overcome this barrier. Synchrotron sources produce coherent, high-brilliance X-ray beams that exploit phase shifts — subtle differences in the speed at which X-rays pass through different materials — to generate images with far greater contrast than absorption-based methods. Applied to opaque amber, synchrotron phase-contrast imaging has revealed previously invisible inclusions, including complete insects, with sufficient resolution to identify taxonomically diagnostic features such as wing venation and genitalic structures.11
Virtual "dissection" of amber specimens using synchrotron microtomography allows researchers to digitally remove layers of amber, rotate the specimen in three dimensions, and examine structures from any angle without physical contact. This non-destructive approach has become indispensable for the study of rare or unique amber fossils, including holotype specimens, where physical preparation would risk irreversible damage.11, 16
Ethical concerns around Burmese amber
The extraordinary scientific value of Burmese amber is shadowed by the circumstances of its extraction. The amber mines of the Hukawng Valley lie within Kachin State, a region of northern Myanmar that has experienced armed conflict between the Kachin Independence Army (KIA) and the Myanmar military (Tatmadaw) since the early 1960s. Control of the mines has shifted repeatedly between the two forces, and revenue from amber sales has been used to fund military operations on both sides of the conflict.12, 13 In 2017, the Myanmar military seized direct control of the major mining sites, and a United Nations fact-finding mission documented serious human rights violations in Kachin State, including forced labour, extrajudicial killings, and sexual violence.12
The paleontological community has grappled publicly with the ethics of studying specimens whose collection may be entangled with armed conflict and human suffering. In April 2020, the Society of Vertebrate Paleontology issued an open letter calling on researchers and journal editors to refrain from publishing studies based on Burmese amber specimens acquired after June 2017, the date the military assumed control of the mines. The response has been uneven: some journals and researchers have adopted the moratorium, while others have argued that a blanket prohibition would suppress important science and that ethical sourcing can be assessed on a case-by-case basis.12 A further complication is that Myanmar classifies amber as a gemstone rather than a fossil, meaning it can be legally exported under gemstone trade regulations even when the biological inclusions it contains would otherwise be subject to restrictions on the export of paleontological material. This legal ambiguity has facilitated the movement of scientifically significant specimens into international markets and private collections without oversight by Myanmar's heritage authorities.12, 13
The situation intensified following the military coup in Myanmar in February 2021, which expanded the Tatmadaw's control over extractive industries and heightened concerns about the flow of amber revenue to an internationally condemned regime. Professional societies, journal editorial boards, and funding agencies continue to debate the appropriate balance between scientific inquiry and ethical responsibility, with no consensus yet reached on a uniform policy.12 The case of Burmese amber has become a landmark example of the broader challenges facing paleontology when access to scientifically irreplaceable material is intertwined with geopolitical conflict and human rights concerns.
References
Experimental maturation of pine resin in sediment to investigate the formation of synthetic copal and amber
Absence of ancient DNA in sub-fossil insect inclusions preserved in ‘Anthropocene’ Colombian copal
Phase contrast X-ray synchrotron imaging: opening access to fossil inclusions in opaque amber
Fossils in Burmese amber offer an exquisite view of dinosaur times — and an ethical minefield
The oldest fossil insect inclusions in amber: Cretaceous Albian deposits from Lebanon
Wings of a feather stick together: functioning of the coupling structures of insect wings preserved in Baltic amber