Overview
- Placoderms (class Placodermi) were the dominant jawed vertebrates of the Devonian period (~420–359 Ma), comprising a morphologically diverse radiation of armored fishes that included the apex predator Dunkleosteus, tiny bottom-dwelling antiarchs, and flattened rhenanids convergent on modern rays.
- Placoderms are the earliest jawed vertebrates known from articulated fossils and include forms that preserve the oldest known evidence of internal fertilization, live birth, and true teeth with dentine, providing critical data on the origin of gnathostome reproductive and dental systems.
- Once considered an extinct evolutionary dead end, placoderms are now recognized as a paraphyletic or stem-group assemblage from which modern jawed vertebrates (chondrichthyans and osteichthyans) likely evolved, making them central to understanding the origin and early evolution of jaws, teeth, and paired appendages.
Overview of Placodermi
Placoderms were an extraordinarily diverse group of jawed fishes that dominated aquatic ecosystems during the Devonian period, often called the “Age of Fishes.” Characterized by a bony head shield articulating with a separate trunk shield via a craniothoracic joint, placoderms encompassed a wide range of body forms, sizes, and ecological niches.2, 3 They first appeared in the Late Silurian (approximately 430–420 Ma), with early forms such as those described by Long and colleagues from fragmentary remains, and went completely extinct at or near the end-Devonian Hangenberg event (approximately 359 Ma), a span of roughly 70 million years during which they were the most species-rich group of jawed vertebrates on Earth.4, 13, 15
Traditionally classified as a monophyletic class within gnathostomes, Placodermi is now increasingly regarded as a paraphyletic assemblage — a grade of stem gnathostomes from which the two surviving jawed vertebrate lineages, Chondrichthyes (sharks and rays) and Osteichthyes (bony fishes and their descendants, including all tetrapods), ultimately arose.8, 13
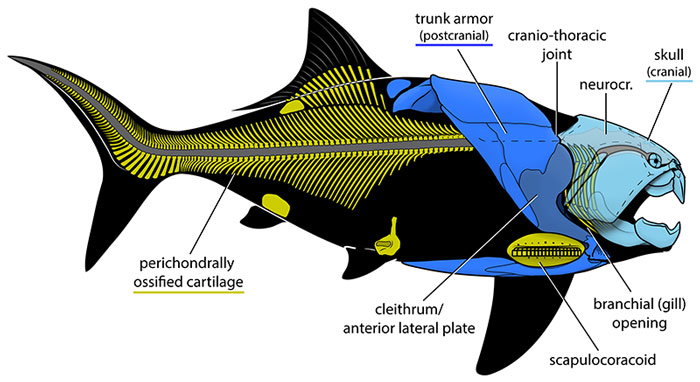
Major placoderm groups
The diversity of placoderms is traditionally divided into several major orders, each with distinctive morphologies and ecological roles.
Arthrodires were the largest and most diverse placoderm order, comprising roughly 60 percent of known placoderm species. They ranged from small, generalized forms to enormous apex predators such as Dunkleosteus terrelli, which reached estimated lengths of 6 meters or more and possessed a powerful bite mechanism with shearing bone plates rather than true teeth.1, 3 Biomechanical analysis of Dunkleosteus jaws has estimated bite forces exceeding 5,000 newtons at the tip of the jaw blades, among the highest calculated for any fish, enabling it to feed on other armored fishes and hard-shelled prey.1
Antiarchs were small, heavily armored, bottom-dwelling placoderms with distinctive box-like trunk shields and jointed pectoral appendages enclosed in bony plates. The most familiar genus, Bothriolepis, is known from hundreds of species worldwide and is one of the most common vertebrate fossils in Devonian freshwater and marginal marine deposits.3, 16 Antiarchs are notable for providing the oldest evidence of internal fertilization in vertebrates: male antiarchs possessed clasper-like intromittent organs, and the discovery of mineralized embryos within the body cavity of the ptyctodontid Materpiscis attenboroughi demonstrated live birth in the Devonian, approximately 380 million years ago.5, 6
Ptyctodontids were small, shark-like placoderms with reduced armor, crushing tooth plates, and pronounced sexual dimorphism. Males bore claspers, and females of at least one species (Materpiscis) carried developing embryos internally, providing unambiguous evidence of viviparity.5
Rhenanids were dorsoventrally flattened placoderms that converged remarkably on the body plan of modern skates and rays, with broad pectoral fins, a flattened body, and a ventral mouth, presumably adapted to a benthic lifestyle.3, 16
Petalichthyids and acanthothoracids were less diverse but phylogenetically significant groups. Acanthothoracids, in particular, include some of the most basal placoderms and have proven important for understanding the origin of the gnathostome body plan.2, 13
The origin of jaws and teeth
Placoderms occupy a critical position in the evolutionary history of jawed vertebrates because they include the earliest known gnathostomes with articulated fossils preserving jaw structure. The origin of jaws from gill arches is a foundational concept in vertebrate evolution, and placoderms provide the earliest direct fossil evidence of this transformation.2, 7
The question of whether placoderms possessed true teeth (as opposed to shearing plates or bony cusps) was long debated. In 2012, Rucklin and colleagues used synchrotron microtomography to examine the jaw plates of the arthrodire Compagopiscis and demonstrated that they contained dentine and a pulp cavity organized in a pattern consistent with true tooth development, making them the earliest known gnathostome teeth.9, 12 This finding was significant because it pushed the origin of teeth back to the base of gnathostome evolution and suggested that teeth evolved once rather than independently in multiple lineages.10
The discovery of Entelognathus primordialis, a placoderm from the Late Silurian of China with marginal jaw bones resembling those of osteichthyans (bony fishes), blurred the boundary between placoderms and modern jawed vertebrates and provided strong evidence that Placodermi is paraphyletic.8, 14 Entelognathus possesses a mosaic of placoderm features (head and trunk shields) and osteichthyan features (premaxilla, maxilla, and dentary-like bones), suggesting that the bony facial architecture of modern vertebrates was inherited from placoderm ancestors rather than evolving independently in bony fishes.8
Reproductive biology
Among the most remarkable discoveries in placoderm paleontology has been the documentation of advanced reproductive strategies. The 2008 report of a mineralized embryo with an umbilical cord-like structure preserved inside the body cavity of the ptyctodontid Materpiscis attenboroughi from the Late Devonian Gogo Formation of Western Australia provided the oldest known evidence of viviparity (live birth) in any vertebrate.5 Subsequent discoveries revealed clasper organs in male antiarchs, demonstrating that internal fertilization was widespread among placoderms and raising the possibility that it was ancestral to gnathostomes as a whole.6, 11
These findings challenged the previous assumption that the primitive gnathostome condition was external fertilization (as seen in most modern bony fishes) and suggested instead that internal fertilization may have been the ancestral state, with external fertilization in bony fishes representing a secondary loss.6
Ecology and paleogeography
Placoderms inhabited virtually all aquatic environments during the Devonian: marine shelves, reefs, open ocean, estuaries, rivers, and lakes. Arthrodires were the dominant predators in many Devonian marine ecosystems, with some forms occupying apex predator niches comparable to those of modern great white sharks.1, 16 Antiarchs were among the most geographically widespread of all Devonian vertebrates, with Bothriolepis known from every continent, providing valuable biostratigraphic correlations for Devonian continental sequences.3
The Gogo Formation of Western Australia, a Late Devonian reef complex preserved in exceptional three-dimensional detail, has yielded some of the best-preserved placoderm fossils known, with individual bones retaining cellular detail and even preserved soft tissues including muscle fibers and nerve canals.5, 16
Phylogenetic reassessment and stem gnathostome status
The traditional view of Placodermi as a monophyletic clade — a single evolutionary branch that diverged from other jawed vertebrates and went wholly extinct — has been fundamentally revised by phylogenetic analyses incorporating new fossil discoveries from China and Australia. The description of Entelognathus primordialis demonstrated that at least some placoderms possessed marginal jaw bones (premaxilla, maxilla, dentary) previously thought to be unique to osteichthyans, implying that the common ancestor of all jawed vertebrates may have had a more complex facial skeleton than previously assumed.8, 19 A second key taxon, Qilinyu rostrata, also from the Silurian of Yunnan Province, preserves a similar mosaic of placoderm-grade head armor and osteichthyan-grade jaw bones, reinforcing the paraphyletic interpretation.19
Brazeau’s reanalysis of “acanthodian” braincases further blurred the lines between placoderms and other early gnathostomes, showing that some taxa previously classified as acanthodians share braincase features with both placoderms and chondrichthyans, suggesting a complex pattern of character distributions at the base of gnathostome phylogeny.18 Under the emerging consensus, the various placoderm orders represent successive branches along the gnathostome stem lineage rather than a single radiation, with arthrodires, antiarchs, and other groups potentially diverging at different points relative to the node leading to crown gnathostomes.13, 17
Soft tissue preservation
The Gogo Formation of the Kimberley region of Western Australia has yielded placoderm fossils with three-dimensional preservation of unprecedented quality. Limestone nodules that formed rapidly around carcasses during the Late Devonian protected not only bones but also soft tissues from decay and compression. Trinajstic and colleagues reported the preservation of mineralized muscle fibers in the arthrodire Gogonasus and other Gogo placoderms, representing some of the oldest vertebrate muscle tissue known.20 Nerve canals, blood vessel traces, and cellular structures of bone (osteocyte lacunae) have also been identified in Gogo specimens, enabling detailed reconstructions of placoderm neurovascular anatomy that are impossible from compressed fossils at other localities.16, 20 The exceptional preservation at Gogo was made possible by the reef-margin environment in which the animals lived: carcasses that fell into fine carbonate mud between reef pinnacles were rapidly entombed in concretions before significant decay could occur, a taphonomic pathway analogous to the famous Solnhofen Limestone of Germany.20
Extinction
Placoderms were devastated by the late Devonian extinction, a series of extinction pulses that culminated in the Hangenberg event at the Devonian–Carboniferous boundary (approximately 359 Ma). Diversity had already been declining through the Famennian Stage, and no placoderms are known from post-Devonian rocks.15 Analysis of diversity patterns across the extinction boundary shows that placoderms did not simply fail to recover — they were actively driven to extinction during the Hangenberg crisis, which preferentially removed large-bodied, armored fish lineages while smaller, less armored forms (including early sharks and bony fishes) survived and diversified in the Carboniferous.15
The extinction of placoderms cleared ecological space that was subsequently filled by chondrichthyans and osteichthyans, fundamentally reshaping vertebrate communities and setting the stage for the fish faunas that would dominate marine and freshwater ecosystems for the remainder of the Phanerozoic.13, 15, 16
References
Copulation in antiarch placoderms and the origin of gnathostome internal fertilization
Neurocranial anatomy of an enigmatic Early Devonian fish sheds light on early osteichthyan evolution