Overview
- Placoderms (class Placodermi) were armored jawed fishes that dominated the world's oceans, rivers, and lakes during the Devonian period (~420–359 Ma), representing the earliest jawed vertebrates known from articulated fossils and encompassing an extraordinary range of body forms from the six-meter apex predator Dunkleosteus to tiny bottom-dwelling antiarchs.
- Their anatomy — dermal bone plates sheathing the head and trunk, a unique craniothoracic joint, and gnathal plates functioning as cutting blades rather than true teeth — provides the primary fossil evidence for how jaws, paired fins, and the gnathostome body plan first evolved in vertebrate history.
- Once classified as a monophyletic evolutionary dead end, placoderms are now widely regarded as a paraphyletic grade of stem gnathostomes from which all modern jawed vertebrates (sharks, bony fishes, and tetrapods including humans) ultimately descended, a view reinforced by the discovery of the Silurian placoderm Entelognathus bearing 'modern' jaw bones.
Placoderms (class Placodermi) are an extinct group of armored jawed fishes that first appeared in the Late Silurian period, approximately 430–420 million years ago, and dominated the world's aquatic ecosystems throughout the Devonian period before disappearing entirely at the Devonian–Carboniferous boundary around 359 million years ago.1, 2 The name, derived from the Greek plax (flat plate) and derma (skin), refers to the group's defining characteristic: a heavy external armor of interlocking dermal bone plates that encased the head and anterior trunk. With more than 300 genera described from every continent, placoderms represent one of the most morphologically diverse vertebrate radiations in the [fossil record](/paleontology/fossil-record-and-deep-time), ranging from the six-meter apex predator *Dunkleosteus terrelli* to diminutive bottom-dwelling antiarchs barely ten centimeters long.2, 3 Their significance extends far beyond the Devonian seas they once ruled: as the earliest jawed vertebrates known from articulated skeletal remains, placoderms provide the foundational fossil evidence for understanding how jaws, teeth, paired fins, and internal fertilization first evolved in the vertebrate lineage that ultimately gave rise to all living gnathostomes — sharks, bony fishes, and tetrapods including humans.1, 14, 15
Anatomy and body plan
The most immediately recognizable feature of placoderms is their dermal armor, which consists of a series of interlocking bony plates covering the head and the anterior portion of the trunk. These plates are divided into two principal units: the head shield (cranial armor), which encases the skull and branchial region, and the trunk shield (thoracic armor), which protects the pectoral region and anterior body cavity. The two shields are connected by a craniothoracic joint, a hinge-like articulation that allowed the head to tilt upward relative to the trunk — a mechanism that, in arthrodires such as *Dunkleosteus*, enabled rapid opening of the mouth for suction feeding and prey capture.1, 2, 5 Posterior to the trunk shield, the body was typically covered only by small scales or was entirely naked, giving placoderms a distinctive front-heavy appearance.
Unlike modern jawed vertebrates, most placoderms did not possess true teeth in the conventional sense. Instead, the margins of their jaw bones bore gnathal plates — sharpened bony blades or crushing surfaces formed from the dermal bone of the jaw itself. In arthrodires, these plates were self-sharpening: as the upper and lower blades slid past each other during jaw closure, they maintained a keen cutting edge through continuous wear.4, 5 However, the question of whether placoderms possessed structures homologous to true teeth has been substantially revised. In 2012, Rücklin and colleagues used synchrotron microtomography to examine the gnathal plates of the arthrodire *Compagopiscis* from the Gogo Formation and demonstrated that they contained dentine and a pulp cavity organized in a pattern consistent with genuine tooth development, pushing the origin of teeth to the base of gnathostome evolution.7 Subsequent work on additional Gogo arthrodires confirmed that these dental structures added new layers of dentine throughout life, a process homologous to tooth replacement in modern vertebrates.8
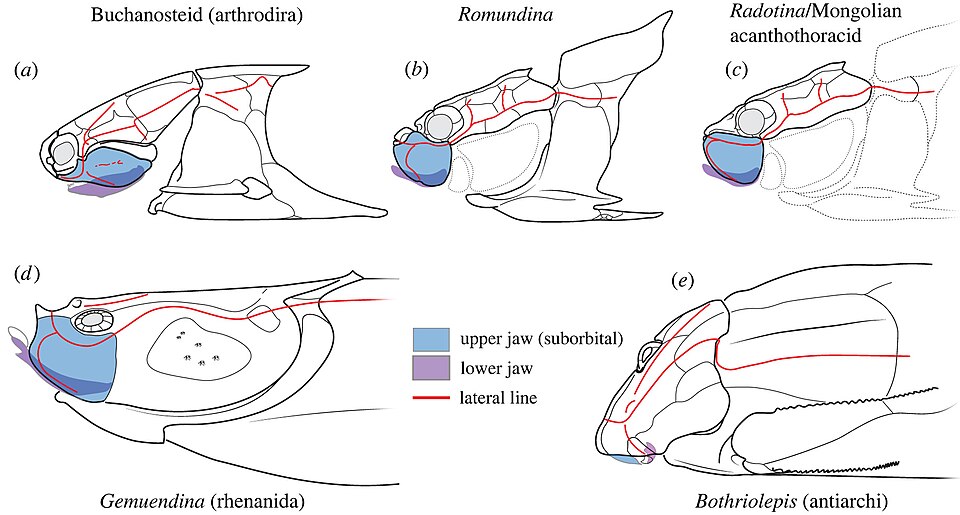
Placoderms possessed paired pectoral fins, and in some groups the pelvic fins were also present, though both were often partially or wholly enclosed in bony armor. The paired fins of antiarchs were particularly distinctive: articulated, jointed appendages sheathed in dermal bone that have been compared structurally (though not homologously) to the limbs of crustaceans.22 The internal skeleton was composed of cartilage in most placoderms, though in some lineages, such as certain arthrodires and the remarkable *Entelognathus*, endochondral ossification of internal elements has been documented.9 Sensory structures included lateral line canals running through the dermal plates of the head shield, and the orbits were typically positioned laterally or dorsally, suggesting that most placoderms had a broad visual field suited to detecting predators or prey approaching from the side or above.1, 2
Major groups
The internal classification of Placodermi has historically recognized between six and nine orders, though recent phylogenetic revisions have complicated traditional boundaries. The major groups span an extraordinary range of body forms and ecological strategies, rivaling the morphological disparity of any comparable vertebrate radiation.
The Arthrodira were by far the most diverse and ecologically dominant placoderm order, comprising roughly 60 percent of all described placoderm species. Arthrodires ranged from small, generalized forms to enormous apex predators. The largest and most famous was *Dunkleosteus terrelli* from the Late Devonian Cleveland Shale of Ohio, which reached estimated lengths of up to 6 meters and possessed one of the most powerful bite mechanisms ever calculated for any vertebrate.4, 5 Biomechanical modeling by Anderson and Westneat estimated bite forces exceeding 5,000 newtons at the tip of the gnathal blades and over 7,400 newtons at the posterior blade edge, sufficient to puncture the armor of other placoderms and crush hard-shelled prey.5 The arthrodire radiation produced a wide spectrum of body forms: small, fast-swimming hunters with reduced armor; large, heavily plated durophages; elongated eel-like forms; and deep-bodied reef dwellers, making the Arthrodira one of the most ecomorphologically diverse orders in Paleozoic vertebrate history.6
The Antiarchi were small, heavily armored, bottom-dwelling placoderms characterized by a box-like trunk shield and remarkable jointed pectoral appendages enclosed entirely in dermal bone. The most ubiquitous genus, *Bothriolepis*, is known from more than 100 species distributed across every continent, making it one of the most species-rich and geographically widespread vertebrate genera in the entire [fossil record](/paleontology/fossil-record-and-deep-time).2, 3, 22 *Bothriolepis* is particularly abundant in [Devonian](/paleontology/devonian-fish-diversity) freshwater and marginal marine deposits, where its fossils frequently occur in mass-death assemblages numbering hundreds or thousands of individuals. The jointed pectoral appendages of antiarchs were probably used to prop the animal on the substrate or to assist in locomotion along the bottom, rather than for swimming in open water.22
The Rhenanida were dorsoventrally flattened placoderms that convergently evolved a body plan strikingly similar to that of modern skates and rays, with broad, wing-like pectoral extensions, a flattened profile, and a ventrally positioned mouth. This convergence on a benthic, ray-like morphology from an entirely independent evolutionary starting point illustrates how similar ecological pressures can produce remarkably similar solutions across distantly related lineages.2, 3
The Ptyctodontida were small, shark-like placoderms with greatly reduced armor, large eyes, and tritoral (crushing) tooth plates suited to a durophagous diet. Ptyctodontids exhibited pronounced sexual dimorphism, with males possessing clasper-like pelvic structures used for internal fertilization — a discovery that proved critical for understanding the reproductive biology of early vertebrates.11, 12
The Petalichthyida were a small but phylogenetically significant order of flattened, bottom-dwelling placoderms, while the Acanthothoraci include some of the most basal known placoderms and have proven essential for rooting the gnathostome family tree. Additional minor groups, including the Brindabellaspida and Pseudopetalichthyida, are known from limited material but contribute to the picture of placoderm morphological diversity at its Devonian peak.1, 14
Dunkleosteus terrelli
No placoderm is more widely recognized than *Dunkleosteus terrelli*, a giant arthrodire from the Late Devonian (Famennian Stage, approximately 358–382 Ma) whose massive head shield and fearsome gnathal blades have made it an icon of prehistoric marine life. Known primarily from the Cleveland Shale of Ohio, with additional material from Morocco, Belgium, and Poland, *Dunkleosteus* was among the largest vertebrates of its time, with total body length estimates ranging from 4.1 to over 6 meters depending on the reconstruction used.3, 4
The biomechanics of *Dunkleosteus* feeding have been studied in detail. The craniothoracic joint allowed the head shield to rotate upward while the lower jaw depressed simultaneously, producing an extremely rapid gape cycle estimated at approximately 20 milliseconds for the initial opening phase — fast enough to generate suction forces that would have drawn prey into the mouth.5 The gnathal plates functioned as self-sharpening shears: the anterior portions were honed into blade-like edges by continuous occlusion, while the posterior portions formed broader, flatter surfaces suited to crushing. Anderson and Westneat's biomechanical analysis revealed that the bite force at the tip of the jaw blades exceeded 5,000 newtons, placing *Dunkleosteus* among the most powerful biters in vertebrate history and rivaling estimates for large theropod dinosaurs and modern crocodilians.5
Because the postcranial skeleton of *Dunkleosteus* was unarmored and composed of cartilage that rarely fossilized, the body shape behind the trunk shield remains uncertain. Early reconstructions depicted a long, shark-like body with a heterocercal tail, but more recent analyses have suggested a shorter, more compact body plan. Regardless of exact proportions, *Dunkleosteus* was unambiguously the apex predator of Late Devonian marine ecosystems, occupying an ecological role that would later be filled by large sharks after the placoderm extinction.3, 4
The origin of jaws and the gnathostome body plan
Placoderms are central to one of the most important questions in vertebrate paleontology: how did jaws evolve? The prevailing model, rooted in comparative embryology and supported by fossil evidence, holds that the vertebrate jaw arose through the modification of one or more anterior pharyngeal (gill) arches in jawless ancestors.1, 15 Placoderms, as the earliest jawed vertebrates with articulated skeletal remains, provide the primary fossil evidence for this transformation. The cranial anatomy of basal placoderms preserves transitional stages in the reorganization of the head skeleton that accompanied the evolution of functional jaws, including changes to the braincase, the arrangement of gill arches, and the configuration of the hyomandibular bone that in later vertebrates would assume roles in jaw suspension and, ultimately, in hearing.1, 17
The discovery of tooth-like structures with dentine in placoderm gnathal plates by Rücklin and colleagues fundamentally altered the understanding of dental evolution. Before this finding, placoderms were thought to possess only bony cutting plates, with "true" teeth (possessing dentine, enameloid, and a pulp cavity) evolving independently in sharks and bony fishes after the divergence of placoderms.7 The demonstration that arthrodire gnathal plates contained organized dentine pushed the origin of teeth back to the base of the gnathostome tree and suggested that teeth evolved only once, in a common ancestor of all jawed vertebrates.7, 8 This interpretation was strengthened by Chen and colleagues' analysis of additional early gnathostome jaw structures, which revealed a conserved developmental program for tooth formation shared by placoderms and crown gnathostomes.8
Beyond jaws and teeth, placoderms preserve evidence of other foundational gnathostome innovations. The paired pectoral and pelvic fins of placoderms are the earliest known paired appendages in the vertebrate fossil record, and their morphological diversity — from the armored pectoral limbs of antiarchs to the more mobile fins of arthrodires — illustrates the early experimentation with paired-appendage function that eventually led, through the sarcopterygian lineage, to the [evolution of tetrapod limbs](/paleontology/fish-to-tetrapod-transition).1, 22
Entelognathus and the phylogenetic revolution
The description of *Entelognathus primordialis* by Zhu and colleagues in 2013 was one of the most consequential discoveries in early vertebrate paleontology in decades. Recovered from Late Silurian rocks (approximately 423 Ma) of Qujing, Yunnan Province, China, this small fish possesses a striking mosaic of characteristics: the head and trunk are encased in typical placoderm-grade dermal armor, with a craniothoracic joint and plate configurations recognizable as arthrodire-like, yet the jaw margins bear bones that closely resemble the premaxilla, maxilla, and dentary of osteichthyans (bony fishes).9
This combination of features had profound implications. Under the traditional view that Placodermi was a monophyletic clade — a single branch that diverged early from the gnathostome stem and went wholly extinct — the marginal jaw bones of bony fishes were interpreted as an independent innovation of the osteichthyan lineage, unrelated to the gnathal plates of placoderms. *Entelognathus* demolished this interpretation by demonstrating that at least some placoderms possessed the very jaw bones previously thought to be unique to bony fishes, implying that these bones were inherited from a common placoderm-grade ancestor rather than invented independently.9, 14 The finding strongly supported the hypothesis that Placodermi is paraphyletic — not a single natural group, but rather a grade of stem gnathostomes arranged along the lineage leading to crown jawed vertebrates.14, 15
A second key taxon, *Qilinyu rostrata*, described in 2016 from rocks of similar age in the same region of Yunnan, reinforced the paraphyletic interpretation. Like *Entelognathus*, *Qilinyu* combines placoderm-grade cranial armor with maxillary and premaxillary jaw bones, and its inclusion in phylogenetic analyses pushes the origin of the osteichthyan-like facial skeleton even deeper into the gnathostome stem.10 Together, these Chinese Silurian fossils have redrawn the gnathostome family tree: rather than placoderms branching off as a single extinct side lineage, the various placoderm orders appear to represent successive branches along the stem leading to modern jawed vertebrates, with some orders (particularly certain arthrodires and maxillate forms) more closely related to crown gnathostomes than to other placoderms.14, 15 This interpretation is consistent with the description of *Guiyu oneiros*, the oldest known articulated osteichthyan from the Late Silurian of Yunnan, whose mosaic of primitive and derived characters demonstrates that the divergence between placoderm-grade and crown-gnathostome anatomy was already underway by approximately 419 million years ago.20
Brazeau's reanalysis of "acanthodian" braincases further complicated the picture, demonstrating that some taxa previously classified as acanthodians — spiny, shark-like fishes traditionally placed outside Placodermi — share braincase features with both placoderms and chondrichthyans. This finding suggested a complex mosaic of character distributions at the base of gnathostome phylogeny that cannot be accommodated by simple dichotomies between "placoderms" and "modern" jawed fishes.17 Detailed neurocranial studies of enigmatic Early Devonian fishes have further blurred these boundaries, revealing that braincase architecture previously considered diagnostic of osteichthyans was already present in forms that retain many placoderm-like features.21
Reproductive biology and live birth
Among the most unexpected discoveries in placoderm research was the documentation of sophisticated reproductive strategies, including the oldest known evidence of viviparity (live birth) in any vertebrate. In 2008, Long and colleagues described a specimen of the ptyctodontid *Materpiscis attenboroughi* from the Late Devonian (approximately 380 Ma) Gogo Formation of Western Australia that preserved a mineralized embryo within the body cavity of the mother, connected by a structure interpreted as an umbilical cord or yolk sac stalk.11 The species was named in honor of Sir David Attenborough, who had visited the Gogo Formation collecting locality decades earlier. This discovery pushed the first evidence of live birth back by approximately 200 million years beyond its previous oldest record and demonstrated that internal gestation of offspring was already present in Devonian vertebrates.11
Subsequent research extended these findings. Long and colleagues documented clasper-like intromittent organs in male antiarch placoderms, demonstrating that internal fertilization was not restricted to ptyctodontids but was widespread across Placodermi.12 The presence of claspers in antiarchs — one of the most basal placoderm orders — raised the provocative possibility that internal fertilization was the ancestral condition for all jawed vertebrates, with the external fertilization seen in most modern bony fishes representing a secondary reversal rather than a primitive state.12 This hypothesis remains debated, but the placoderm evidence unambiguously demonstrates that vertebrate reproductive complexity has far deeper evolutionary roots than was previously appreciated.
The Gogo Formation
The Gogo Formation of the Kimberley region of Western Australia is one of the most important fossil sites in the world for understanding placoderm biology. Deposited during the Late Devonian (Frasnian Stage, approximately 382–372 Ma) in a reef-margin environment, the Gogo sediments preserve vertebrate fossils in exceptional three-dimensional fidelity within limestone concretions that formed rapidly around carcasses on the seafloor.3, 13 The rapid encapsulation in fine carbonate mud halted decomposition and prevented the compression that destroys anatomical detail in most fossil deposits, yielding specimens in which individual bones retain cellular-level structures including osteocyte lacunae, nerve canals, and blood vessel traces.13
Trinajstic and colleagues reported the preservation of mineralized muscle fibers in Gogo placoderms, representing some of the oldest vertebrate soft tissue known. These findings have enabled reconstructions of placoderm musculature and neurovascular anatomy that would be impossible from compressed fossils at other localities.13 The Gogo Formation has also yielded the embryo-bearing *Materpiscis* specimen that documented live birth, as well as the sarcopterygian *Gogonasus*, whose exceptionally preserved postcranial skeleton shed light on the [fish-to-tetrapod transition](/paleontology/fish-to-tetrapod-transition), and some of the most complete arthrodire, ptyctodontid, and antiarch skeletons known, making it an unrivaled window into the anatomy, ecology, and reproductive biology of Devonian fishes.3, 11, 18 The taphonomic pathway responsible for this preservation — rapid concretion formation in reef-margin muds — is analogous to the mechanism that produced other famous [exceptional fossil localities](/paleontology/lagerstatten-exceptional-preservation), such as the Solnhofen Limestone of Germany and the [Burgess Shale](/paleontology/burgess-shale) of British Columbia.13
Ecology and paleogeography
Placoderms inhabited virtually every aquatic environment available during the Devonian: open-ocean pelagic zones, continental shelves, reef complexes, estuaries, rivers, lakes, and even marginal terrestrial habitats subject to seasonal drying. This ecological breadth is reflected in their morphological diversity. Large arthrodires such as *Dunkleosteus* occupied apex predator niches in marine ecosystems, analogous to the roles played today by great white sharks and orcas.3, 4 Smaller arthrodires filled mid-trophic-level predator roles, feeding on invertebrates and smaller fishes. Antiarchs were predominantly benthic detritivores or herbivores in freshwater and marginal marine settings, while rhenanids occupied ray-like benthic niches on the seafloor, and ptyctodontids were durophages that fed on hard-shelled invertebrates.1, 2
The cosmopolitan distribution of genera such as *Bothriolepis* — found on every continent, including Antarctica — provides valuable biostratigraphic data for correlating Devonian sedimentary sequences across different paleocontinents. The ability of antiarchs to tolerate a range of salinities, from fully marine to freshwater, accounts for their extraordinary geographic spread and their abundance in continental deposits.2, 22 In many Devonian ecosystems, placoderms were the numerically dominant vertebrates: mass-death assemblages containing hundreds of *Bothriolepis* individuals are known from sites in Canada, Australia, and China, suggesting that these animals formed large populations comparable in density to modern schooling fishes.3
Extinction and legacy
Placoderms were eliminated by the [late Devonian extinction](/paleontology/late-devonian-extinction), a protracted series of biotic crises that culminated in the Hangenberg event at the Devonian–Carboniferous boundary (approximately 359 Ma). Placoderm diversity had already been declining through the Famennian Stage of the Late Devonian: the Frasnian–Famennian extinction pulse (the Kellwasser event, approximately 372 Ma) eliminated many reef-associated arthrodires, and overall generic richness fell substantially before the final Hangenberg crisis delivered the coup de grâce.16 No placoderm fossils are known from any post-Devonian rocks anywhere in the world, indicating that the group's extinction was complete and global.14, 16
Analysis of diversity and body-size patterns across the Devonian–Carboniferous boundary by Sallan and Coates revealed that the Hangenberg event preferentially removed large-bodied, heavily armored vertebrate lineages while smaller, less armored forms — including early chondrichthyans and bony fishes — survived and diversified in the Carboniferous.16 Sallan and Galimberti documented a dramatic reduction in vertebrate body size following the end-Devonian extinctions, a pattern they termed the "Lilliput effect," in which surviving lineages were disproportionately small-bodied and only gradually re-evolved large body sizes over millions of years.19 The ecological space vacated by placoderms was subsequently filled by sharks and bony fishes, fundamentally restructuring vertebrate communities and establishing the faunal composition that would characterize marine and freshwater ecosystems for the remainder of the Phanerozoic.14, 16
Significance for vertebrate evolution
The significance of placoderms extends far beyond their role as Devonian ecological dominants. As the earliest jawed vertebrates with a substantial fossil record, they provide the primary window into the assembly of the gnathostome body plan — the suite of anatomical innovations (jaws, teeth, paired fins, internal fertilization) that underlies the biology of more than 60,000 living species of jawed vertebrates, from sharks to humans.1, 15 The recognition that Placodermi is likely paraphyletic, rather than a monophyletic dead-end clade, has transformed their evolutionary significance: rather than representing an experiment that failed, placoderms are now understood as the ancestral stock from which all modern jawed vertebrates descended.14
The implications of this revised phylogeny are profound. Features once thought to have evolved independently in sharks and bony fishes — including marginal jaw bones, organized dentine in teeth, and clasper-based internal fertilization — may instead be ancient gnathostome inheritances lost or modified in different descendant lineages.9, 12, 15 The Silurian placoderms *Entelognathus* and *Qilinyu* demonstrate that the facial skeleton of modern vertebrates was already present in animals that otherwise looked like typical armored placoderms, collapsing the morphological gap between "primitive" and "advanced" jawed vertebrates and revealing that the evolutionary transition from placoderm-grade anatomy to modern gnathostome anatomy was far more gradual and mosaic than previously imagined.9, 10
Placoderms thus occupy a pivotal position in [life through time](/paleontology/life-through-time): they are the bridge between the jawless vertebrates of the Ordovician and Silurian — lampreys, hagfishes, ostracoderms — and the explosively diverse jawed vertebrate fauna that emerged in the Carboniferous and persists today. Every human jaw, every shark tooth, every bird wing traces its evolutionary ancestry through the armored fishes that once ruled the Devonian world. Far from an evolutionary dead end, placoderms are the foundation upon which the entire edifice of gnathostome diversity was built.14, 15
References
Feeding mechanics and bite force modelling of the skull of Dunkleosteus terrelli, an ancient apex predator
Copulation in antiarch placoderms and the origin of gnathostome internal fertilization
Neurocranial anatomy of an enigmatic Early Devonian fish sheds light on early osteichthyan evolution