Overview
- Planetary atmospheres originate through volcanic outgassing, gravitational capture of nebular gas, and delivery of volatiles by comets and asteroids, with each mechanism dominating at different stages of planetary formation.
- The compositions of atmospheres across the solar system vary enormously, from the thick CO₂ envelopes of Venus and Mars to Earth's uniquely oxygen-rich air shaped by the Great Oxidation Event, to the hydrogen-helium envelopes of gas giants and the methane-nitrogen haze of Titan.
- Atmospheric escape processes continuously strip gases from planets, and the study of exoplanet atmospheres through transit spectroscopy is now revealing the diversity of atmospheric compositions beyond our solar system and informing the search for habitable worlds.
A planetary atmosphere is the layer of gases gravitationally bound to a planet or large moon. Atmospheres govern surface temperature, shield surfaces from radiation, mediate chemical cycles, and in at least one known case sustain life. The diversity of atmospheres across the solar system, from the crushing carbon dioxide envelope of Venus to the tenuous remnant clinging to Mars to the vast hydrogen-helium mantles of Jupiter and Saturn, reflects the varied histories of volatile acquisition, chemical evolution, and atmospheric loss that each world has experienced since the formation of the solar system approximately 4.6 billion years ago.1, 18
Understanding how atmospheres form, evolve, and are lost has become one of the central questions in planetary science, particularly as the discovery of thousands of exoplanets and the advent of transit spectroscopy now allow astronomers to characterise atmospheres around distant worlds and assess their potential habitability.14
Origins of atmospheres
Planetary atmospheres arise through three principal mechanisms, each of which has operated to varying degrees across the solar system. The first is gravitational capture of nebular gas, in which a sufficiently massive protoplanet directly accretes hydrogen and helium from the solar nebula during the earliest phase of planet formation. This process accounts for the thick hydrogen-helium envelopes of the gas giants Jupiter and Saturn, which formed quickly enough to capture enormous quantities of nebular gas before the solar wind dispersed the protoplanetary disk.8, 18
The second mechanism is volcanic outgassing, the release of volatiles from a planet's interior through volcanism and magmatic activity. As the terrestrial planets accreted, they incorporated volatile-bearing minerals. Subsequent heating from radioactive decay, gravitational differentiation, and impacts drove these volatiles to the surface, producing secondary atmospheres rich in water vapour, carbon dioxide, nitrogen, and sulfur compounds.1, 2 Earth's early atmosphere was likely dominated by CO2, N2, and H2O released through sustained volcanic degassing, with relatively little free oxygen.2, 13
The third mechanism is exogenous delivery by comets and volatile-rich asteroids. Measurements of the deuterium-to-hydrogen ratio in terrestrial ocean water, compared with ratios in various classes of meteorites and comets, indicate that a significant fraction of Earth's water was delivered by carbonaceous chondrite asteroids during the late stages of accretion and the Late Heavy Bombardment.3 Comets likely contributed a smaller but non-negligible share of volatiles, including organic compounds that may have played a role in prebiotic chemistry.3
Atmospheres across the solar system
Venus, Earth, and Mars illustrate how worlds of broadly similar size and initial endowment can develop radically different atmospheres.
Venus possesses a dense atmosphere of approximately 96.5 percent carbon dioxide with a surface pressure of about 92 bar, roughly ninety times that of Earth. Its extreme surface temperature of approximately 465 degrees Celsius is maintained by a powerful greenhouse effect in which the thick CO2 atmosphere, along with clouds of sulfuric acid, traps outgoing infrared radiation with extraordinary efficiency.4, 5 Early in its history, Venus may have had liquid water on its surface, but a runaway greenhouse effect, in which increasing temperatures caused ocean evaporation that further amplified warming, ultimately drove all water into the atmosphere, where it was lost to space through photodissociation and hydrogen escape.9
Mars retains a thin atmosphere of about 95 percent CO2 at a surface pressure of only 6 millibars, less than one percent of Earth's. Geological and mineralogical evidence strongly suggests that Mars once possessed a thicker atmosphere and liquid surface water, but the planet's low gravity and the absence of a global magnetic field left it vulnerable to atmospheric stripping by the solar wind over billions of years.6, 12
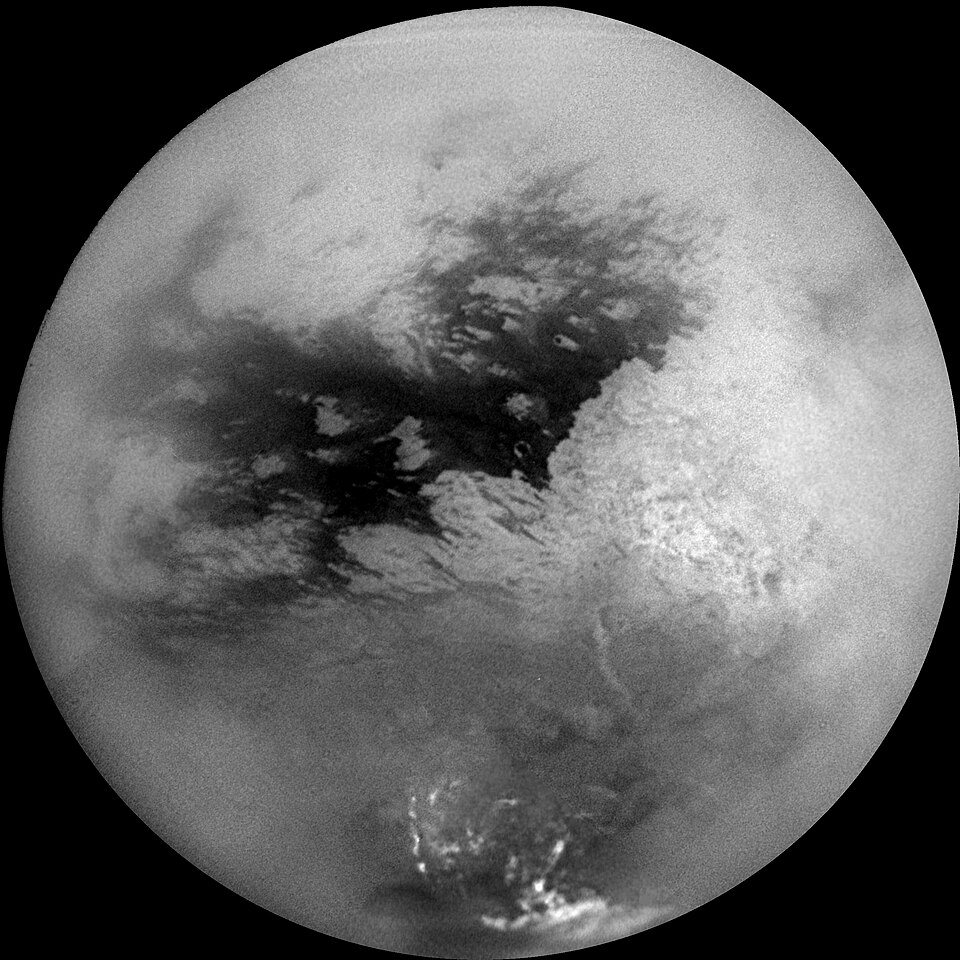
Among the outer solar system's moons, Titan stands as a remarkable outlier. Saturn's largest moon maintains a dense nitrogen-dominated atmosphere with a surface pressure of about 1.5 bar, slightly higher than Earth's, along with approximately 5 percent methane that participates in a hydrocarbon cycle analogous to Earth's water cycle, complete with rain, rivers, and lakes of liquid methane and ethane.7
The gas giants Jupiter and Saturn possess atmospheres composed primarily of hydrogen (approximately 86 and 96 percent by volume, respectively) and helium, along with trace amounts of methane, ammonia, water vapour, and phosphine that produce their characteristic banded cloud structures.8
Atmospheric composition of selected solar system bodies5, 6, 7, 8
| Body | Dominant gas | Surface pressure | Surface temp. |
|---|---|---|---|
| Venus | CO2 (96.5%) | ~92 bar | ~465 °C |
| Earth | N2 (78%) / O2 (21%) | 1.0 bar | ~15 °C |
| Mars | CO2 (95%) | ~0.006 bar | ~−60 °C |
| Titan | N2 (95%) / CH4 (5%) | ~1.5 bar | ~−179 °C |
| Jupiter | H2 (86%) / He (13%) | No solid surface | — |
The greenhouse effect
The greenhouse effect is the process by which certain atmospheric gases absorb and re-emit infrared radiation emitted by a planet's surface, thereby warming the lower atmosphere and surface above the temperature that would prevail in the absence of an atmosphere.

On Earth, the naturally occurring greenhouse gases, primarily water vapour, carbon dioxide, and methane, raise the global mean surface temperature from a theoretical blackbody equilibrium of approximately −18 degrees Celsius to the observed average of about 15 degrees Celsius, a warming of roughly 33 degrees that is essential for maintaining liquid water and habitable conditions.1, 18
Venus demonstrates the extreme end of greenhouse warming. Although Venus receives roughly twice the solar flux of Earth, its extraordinarily high surface temperature of 465 degrees Celsius cannot be explained by solar heating alone; it is overwhelmingly the product of a massive greenhouse effect sustained by the thick CO2 atmosphere. Ingersoll's 1969 analysis showed that once surface temperatures rise enough to prevent water from condensing, the resulting water vapour feedback can drive a runaway greenhouse process that ultimately vaporises all surface water.9 This mechanism is thought to explain how Venus transitioned from a potentially habitable early state to the inferno it is today.
The Great Oxidation Event
Earth's atmosphere has undergone a transformation unique among known worlds: the rise of free molecular oxygen. For the first two billion years of Earth's history (see Precambrian Earth), the atmosphere contained negligible free O2. Geological evidence including mass-independent fractionation of sulfur isotopes in sedimentary rocks older than about 2.4 billion years indicates that atmospheric oxygen concentrations were below one hundred-thousandth of present levels throughout the Archean eon.10
The Great Oxidation Event (GOE), occurring approximately 2.4 billion years ago, marks the first sustained rise of oxygen to levels above this threshold. The oxygen was produced by cyanobacteria through oxygenic photosynthesis, a metabolic innovation that may have evolved several hundred million years before the GOE itself, with oxygen initially consumed by reduced minerals and dissolved iron in the oceans before accumulating in the atmosphere.10, 11 The consequences of oxygenation were profound: the GOE triggered a global glaciation (the Huronian glaciation) as the potent greenhouse gas methane was oxidised, fundamentally restructured ocean chemistry, enabled the later evolution of aerobic metabolism and complex multicellular life, and established the ozone layer that shields the surface from ultraviolet radiation.10
Atmospheric escape processes
All planetary atmospheres lose gas to space through a variety of escape mechanisms, and the balance between volatile supply and escape determines whether a world retains a thick atmosphere or is stripped bare over geological time. Thermal escape (Jeans escape) occurs when molecules in the high-velocity tail of the Maxwell-Boltzmann distribution exceed the planet's escape velocity. Because lighter molecules reach higher velocities at a given temperature, hydrogen and helium escape preferentially, which is why Earth retains virtually no primordial hydrogen in its atmosphere despite having accreted hydrogen-bearing compounds.12
Non-thermal escape processes are equally important, particularly for planets lacking strong magnetic fields. Solar wind sputtering, in which energetic ions from the solar wind collide with atmospheric molecules and eject them to space, has been a major loss mechanism for Mars, whose global magnetic field ceased operating roughly 4 billion years ago. Measurements by NASA's MAVEN spacecraft confirmed that solar wind stripping continues to erode the Martian atmosphere at a rate of approximately 100 grams per second today, and the rate would have been substantially higher when the young Sun was more active.6, 12 Hydrodynamic escape, in which heating of the upper atmosphere by extreme ultraviolet radiation drives a bulk outflow of gas, was likely important during the early solar system when the young Sun's ultraviolet output was several times its present level, potentially stripping hydrogen-rich early atmospheres from the terrestrial planets.13
Exoplanet atmosphere studies
The characterisation of atmospheres around exoplanets has advanced rapidly since the first detection of an exoplanetary atmosphere in 2001. Transit spectroscopy, in which starlight filtered through a planet's atmosphere during transit is analysed for absorption features, has identified sodium, potassium, water vapour, carbon dioxide, and methane in the atmospheres of hot Jupiters and sub-Neptunes.14 The observation of atmospheric hydrogen escape from the hot Jupiter HD 209458b provided direct evidence that atmospheric mass loss operates on close-in exoplanets, confirming theoretical predictions.16
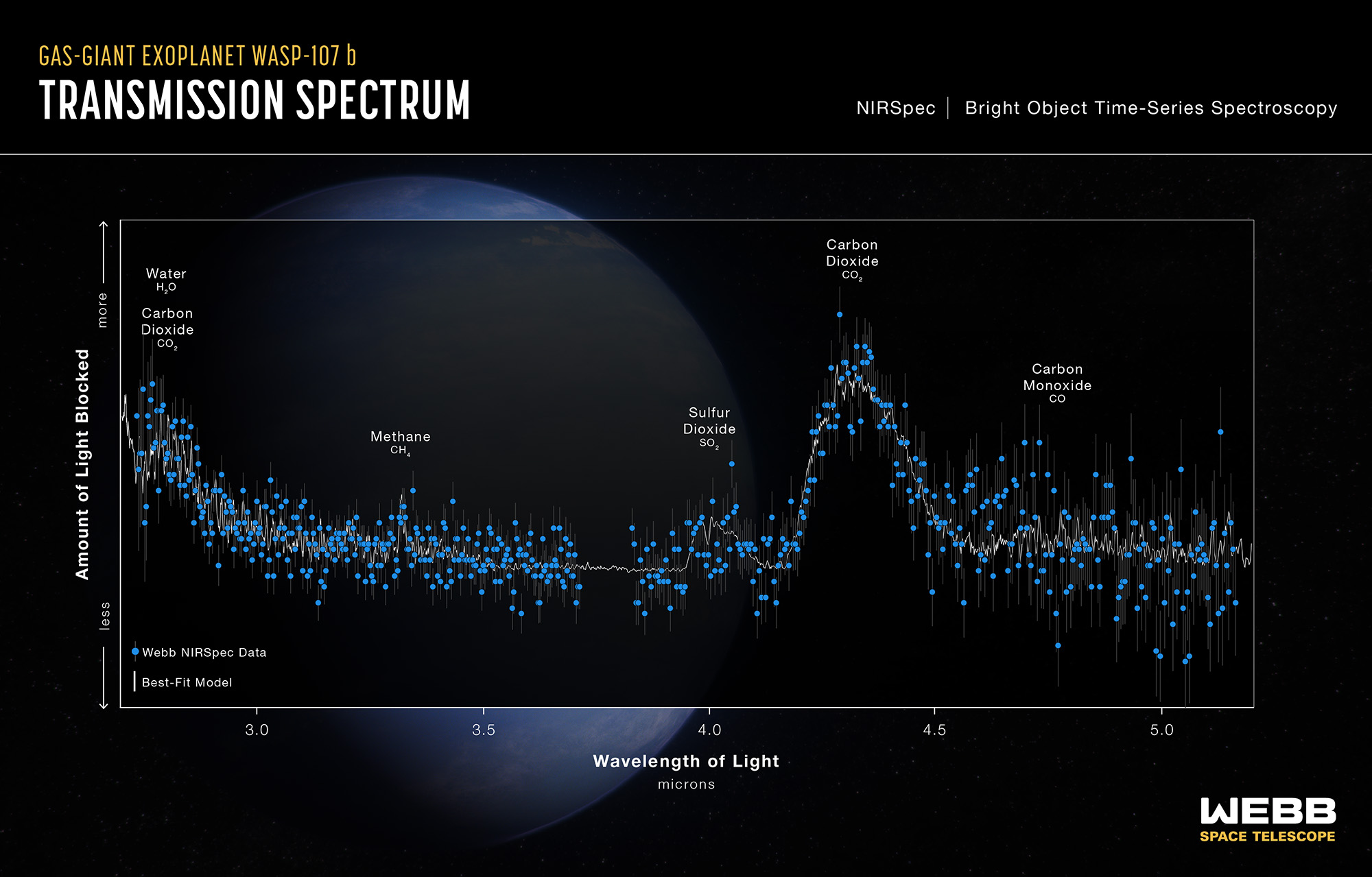
The James Webb Space Telescope (JWST) has dramatically expanded these capabilities. In 2022, JWST produced the first unambiguous detection of CO2 in an exoplanet atmosphere, identifying a clear carbon dioxide absorption feature in the transmission spectrum of the hot gas giant WASP-39b.15 JWST observations of the TRAPPIST-1 system, a compact system of seven roughly Earth-sized planets orbiting an ultracool dwarf star, have begun to constrain whether these potentially habitable worlds retain atmospheres at all. Early results for TRAPPIST-1 b suggest that the innermost planet lacks a thick atmosphere, consistent with models of atmospheric loss driven by the host star's high-energy radiation environment.17
The ultimate goal of exoplanet atmosphere studies is the detection of biosignature gases, atmospheric species such as oxygen, ozone, and methane in thermodynamic disequilibrium, that would suggest biological activity on a distant world. While no such detection has yet been made, the combination of JWST's sensitivity with next-generation ground-based extremely large telescopes is expected to bring the characterisation of Earth-sized exoplanet atmospheres within reach in the coming decade, marking a pivotal advance in the search for life beyond our solar system.14, 18
References
Volcanic outgassing of CO₂ and H₂O on Earth and the composition of the early atmosphere
Evidence for oxygenic photosynthesis half a billion years before the Great Oxidation Event