Overview
- Allopatric speciation, in which geographic barriers divide a population and allow independent divergence, is the most common and best-documented mode of speciation in animals, supported by biogeographic analyses showing that over 70 percent of sister species pairs have non-overlapping ranges.
- Sympatric speciation, in which new species arise within a single continuously distributed population without geographic isolation, has been demonstrated in several compelling systems including cichlid fishes in small African crater lakes and host-race formation in the apple maggot fly Rhagoletis pomonella, though it remains far less common than allopatric divergence.
- Parapatric speciation along environmental gradients and peripatric speciation through founder events in small peripheral populations represent additional modes that blur the boundaries of the geographic classification, and genomic evidence increasingly reveals that gene flow during divergence is more common than the classical models assumed.
The formation of new species — speciation — can be classified by the geographic context in which populations diverge. In allopatric speciation, a physical barrier such as a mountain range, ocean, or river divides a once-continuous population into geographically separated groups that evolve independently until they can no longer interbreed. In sympatric speciation, reproductive isolation arises within a single population occupying the same geographic area, without any physical separation. Between these two extremes lie parapatric speciation, in which populations diverge along an environmental gradient while maintaining a narrow zone of contact, and peripatric speciation, in which a small peripheral population becomes isolated and diverges rapidly from the larger parent population.1, 2 The geographic classification of speciation modes, first systematised by Ernst Mayr in the mid-twentieth century, remains the dominant framework for understanding how biodiversity originates, though modern genomic evidence has revealed that the boundaries between these categories are often blurred by ongoing gene flow during divergence.1, 19
Allopatric speciation
Allopatric speciation, also called geographic speciation or vicariant speciation, occurs when a geographic barrier physically divides a population into two or more isolated subpopulations.
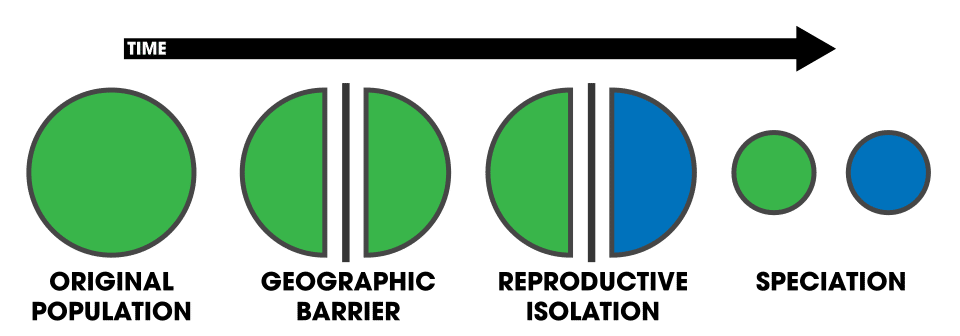
Once gene flow between the subpopulations is eliminated, genetic differences accumulate through natural selection acting on different environments, through genetic drift, and through the independent fixation of new mutations. Over time, the isolated populations diverge in morphology, behaviour, ecology, and genetics until they are reproductively incompatible — that is, even if the barrier is later removed and the populations come into secondary contact, they can no longer produce fertile hybrid offspring.1, 2
Ernst Mayr championed allopatric speciation as the predominant mode of species formation in his foundational works of 1942 and 1963, arguing that geographic isolation is almost always a necessary precondition for the evolution of reproductive barriers in animals.1 His reasoning was straightforward: in a freely interbreeding population, gene flow acts as a homogenising force that prevents the accumulation of genetic differences sufficient for reproductive isolation. Only when gene flow is physically interrupted can populations diverge to the point of speciation. This view, sometimes called the geographic model of speciation, became the default expectation of evolutionary biology for the latter half of the twentieth century.1, 2
The barriers that initiate allopatric speciation are diverse. The uplift of a mountain range can bisect a species' range, as the rise of the Isthmus of Panama approximately three million years ago separated marine organisms on either side into Caribbean and Pacific populations that subsequently diverged into distinct species.2 Glacial advances can fragment formerly continuous habitats into isolated refugia. River formation, volcanic activity, and continental drift all produce vicariant barriers that divide populations. The key requirement is that the barrier must persist long enough for sufficient genetic divergence to accumulate — typically on the order of tens of thousands to millions of years, depending on the organisms involved and the strength of divergent selection.1, 13
Darwin's finches and the allopatric model
The adaptive radiation of Darwin's finches on the Galápagos archipelago is the most celebrated example of allopatric speciation in action.

At least eighteen species of finch evolved from a single common ancestor that colonised the islands approximately one to two million years ago, diversifying into an extraordinary range of beak sizes and shapes adapted to different food sources — from the heavy crushing beaks of seed-eating ground finches to the slender probing beaks of insectivorous warbler finches.15, 16
David Lack's pioneering 1947 monograph proposed that speciation in Darwin's finches followed a cycle of geographic isolation and secondary contact. In the first phase, a population colonises a new island in the archipelago and becomes geographically isolated from the parent population on the source island. During this allopatric phase, the colonists adapt to the ecological conditions of their new island, diverging in morphology and behaviour. In the second phase, dispersal brings the now-differentiated populations back into contact on the same island. If divergence has progressed far enough that the populations are reproductively isolated, the two species can coexist; if not, hybridisation and competition may drive further divergence through character displacement, or one population may be absorbed back into the other.15, 16
Peter and Rosemary Grant's long-term field studies on the island of Daphne Major, begun in 1973 and spanning more than four decades, have provided direct observational evidence for this model. They documented natural selection acting on beak morphology in real time during drought years, showed that hybridisation between species is surprisingly common and can transfer adaptive genetic variation between lineages, and demonstrated that the secondary contact phase of allopatric speciation involves complex ecological interactions that shape the outcome of divergence.16 Whole-genome sequencing of 120 individuals spanning all Darwin's finch species confirmed the central importance of inter-island isolation for speciation, while also revealing extensive gene flow between species and identifying the ALX1 gene as a major locus underlying beak shape variation.17 The genomic data showed that divergence between species is remarkably shallow — consistent with a recent, rapid radiation facilitated by geographic isolation across multiple islands.17
Peripatric speciation and founder events
Peripatric speciation is a special case of allopatric speciation in which a small peripheral population becomes isolated at the edge of the parent species' geographic range. Because the founding population is small, it carries only a fraction of the genetic variation present in the parent population — a sampling phenomenon known as the founder effect. Mayr argued that this genetic bottleneck could accelerate divergence by altering allele frequencies, exposing previously rare alleles to selection, and disrupting co-adapted gene complexes, leading to a rapid "genetic revolution" that might produce reproductive isolation more quickly than would occur in large, stable allopatric populations.1
The concept of peripatric speciation was deeply influenced by Mayr's field observations of bird distributions in the islands of New Guinea and the Solomon archipelago during the 1920s and 1930s. He noted that small, peripheral island populations were often the most morphologically distinctive members of a species complex, precisely the pattern expected if founder events and subsequent divergence in isolation were driving speciation.1 The role of founder effects in speciation remains debated, however. Theoretical analyses have shown that genetic drift alone is generally insufficient to establish reproductive isolation unless populations are extremely small, and that strong divergent selection is still required for the rapid accumulation of incompatibilities.12, 13 Nevertheless, peripatric speciation likely contributes to diversification on island archipelagos and other fragmented landscapes where colonisation events are frequent and founding populations are small.2
Sympatric speciation
Sympatric speciation — the origin of new species from a single population without geographic isolation — was long considered theoretically implausible because gene flow within a panmictic population was thought to overwhelm any incipient divergence. If individuals in the same area can freely interbreed, how could two distinct gene pools form? The theoretical burden of proof for sympatric speciation is therefore far higher than for allopatric speciation: it must be demonstrated that reproductive isolation can evolve despite ongoing opportunities for gene exchange.2, 11
Mathematical models have shown that sympatric speciation is theoretically possible under specific conditions. Dieckmann and Doebeli's influential 1999 model demonstrated that frequency-dependent competition for resources can generate disruptive selection that splits a population into two ecologically distinct groups, and that assortative mating — the tendency for like individuals to mate with each other — can evolve to reinforce this ecological divergence and produce reproductive isolation.7 Subsequent modelling work confirmed that sympatric speciation is most likely when disruptive or divergent selection is strong, when assortative mating can be based on the same traits under ecological selection (a "magic trait" scenario), and when the fitness landscape has multiple peaks separated by a valley of low fitness.11, 12
Coyne and Orr proposed four stringent criteria that must be satisfied to demonstrate sympatric speciation in nature: the putative sister species must be shown to be each other's closest relatives (monophyly); they must be reproductively isolated; their ranges must largely overlap; and the biogeographic and evolutionary history must make periods of allopatric divergence highly unlikely.2 Very few empirical cases have met all four criteria, but several systems provide compelling evidence, as described in the sections that follow.2, 11
The apple maggot fly: sympatric host-race formation
The apple maggot fly, Rhagoletis pomonella, is the most thoroughly studied candidate for sympatric speciation in animals. Native to eastern North America, the fly originally fed exclusively on the fruits of hawthorn trees (Crataegus spp.). Sometime in the mid-nineteenth century, following the introduction of cultivated apple (Malus pumila) to the region, a population of R. pomonella shifted to infesting apple fruits — a host switch that occurred in the complete absence of any geographic barrier between the two host plants.3
Guy Bush first proposed in 1969 that this host shift represented sympatric speciation in progress, arguing that the apple-infesting population constituted a partially reproductively isolated "host race" that was diverging from the ancestral hawthorn race within the same geographic area.3 Subsequent genetic work by Feder, Chilcote, and Bush provided the critical evidence. In 1988, they demonstrated significant allele frequency differences at six allozyme loci between apple and hawthorn flies collected from the same orchards in Michigan, confirming that the two host races were genetically differentiated despite living in sympatry.4
The mechanism driving this divergence is allochronic isolation — a difference in the timing of life-history events. Apple fruits ripen approximately three to four weeks earlier than hawthorn fruits, and the flies have adapted their emergence timing to match their respective host plants. Because Rhagoletis flies mate on or near their host fruit, the difference in fruiting phenology creates a temporal barrier to gene flow between the two races. The allozyme loci that differ between the races map to chromosomal inversions and are correlated with the duration of overwintering pupal diapause, the physiological mechanism that controls adult emergence timing.4, 19 Natural selection on host-associated traits thus simultaneously produces ecological divergence and assortative mating — exactly the conditions that theoretical models predict should facilitate sympatric speciation.7, 14
Crater lake cichlids: sympatric diversification
The cichlid fishes of Africa's Great Lakes are renowned for their explosive adaptive radiations, with hundreds of species having evolved in Lakes Victoria, Malawi, and Tanganyika. While much of this diversification likely involved allopatric or microallopatric divergence within the complex topography of the large lakes, the small volcanic crater lakes of Cameroon and Nicaragua have provided some of the most compelling evidence for sympatric speciation in vertebrates.5, 6, 21

In 1994, Schliewen, Tautz, and Pääbo published a landmark study of cichlid species flocks in two Cameroonian crater lakes, Lake Barombi Mbo and Lake Bermin. Using mitochondrial DNA phylogenetics, they demonstrated that the cichlid species within each lake formed a monophyletic group — meaning that each lake was colonised by a single ancestral species, and all subsequent diversification occurred within the lake. Because the lakes are small, roughly circular, and lack internal geographic barriers, the authors argued that the diversification must have been sympatric.5
A similarly compelling case emerged from Lake Apoyo, a small volcanic crater lake in Nicaragua. Barluenga and colleagues showed in 2006 that two morphologically distinct cichlid species — the ancestral benthic Amphilophus citrinellus and the derived limnetic Amphilophus zaliosus — are sister taxa that diverged within the lake less than approximately 10,000 years ago. Population genetic analyses using mitochondrial DNA, microsatellites, and amplified fragment length polymorphisms confirmed that the lake was colonised only once and that the two species are reproductively isolated, ecologically differentiated (one is benthic and the other limnetic), and morphologically distinct in body shape.6
The broader pattern across African cichlid radiations reveals a complex interplay of speciation modes. Whole-genome sequencing of 134 Lake Malawi cichlids covering 73 species found that average sequence divergence between species pairs is only 0.1 to 0.25 percent — remarkably low, and overlapping with the genetic diversity within individual species.22 The genomic data also revealed evidence of substantial gene flow at multiple time points during the radiation, with no single species tree adequately representing all relationships. This suggests that the distinction between allopatric and sympatric speciation may be less sharp than traditionally assumed: even in systems that appear allopatric, gene flow can persist during divergence, and even in systems that appear sympatric, microgeographic structure within lakes may reduce effective gene flow between diverging populations.21, 22
Parapatric speciation and ring species
Parapatric speciation occurs when populations diverge along an environmental gradient while maintaining a narrow zone of contact and limited gene flow. Unlike allopatric speciation, there is no complete physical barrier; unlike sympatric speciation, the diverging populations are not fully overlapping in range. Instead, divergent natural selection across the gradient can steepen genetic clines — gradual changes in allele frequency across geographic space — until they become sharp enough to function as effective barriers to gene exchange. John Endler's 1977 monograph provided the theoretical framework for this process, showing that selection across environmental gradients can produce steep clines that develop into hybrid zones and, given sufficient time and selection intensity, lead to the evolution of complete reproductive isolation.8
Ring species provide a striking natural illustration of how geographic and genetic divergence can accumulate along a continuous distribution (see ring species) until populations at the terminal ends of the ring are reproductively incompatible, even though gene flow connects them through a chain of intermediate populations. The Ensatina eschscholtzii complex of plethodontid salamanders in western North America is the classic example. The complex forms a roughly horseshoe-shaped distribution around the Central Valley of California, with northern populations gradually diverging into two distinct lineages as they extend southward along the coastal ranges and the Sierra Nevada, respectively. Where the two arms of the ring meet in southern California, the terminal forms — the blotched E. e. klauberi and the large-blotched E. e. eschscholtzii — coexist without interbreeding, behaving as distinct biological species, even though they are connected by a continuous chain of intergrading subspecies around the ring.9, 10
Molecular phylogenetic analysis of the Ensatina complex using mitochondrial cytochrome b sequences confirmed the ring species interpretation by demonstrating that the phylogeny is consistent with a north-to-south range expansion down two axes, with secondary contact and reproductive isolation at the southern end of the ring.10 However, the molecular data also revealed a more complex picture than the simple ring model suggests: some subspecies are paraphyletic in their mitochondrial lineages, indicating historical gene flow and incomplete lineage sorting. Wake's comprehensive 1997 analysis concluded that the Ensatina complex includes several genetically distinct components that are at or near the species level, and that while the general biogeographic hypothesis is supported, the ring is not a perfectly continuous gradient of divergence but rather a mosaic of semi-independent lineages.9
The relative frequency debate
The question of how often speciation occurs in allopatry versus sympatry has been one of the most persistent debates in evolutionary biology. Mayr's strong advocacy for the geographic model made allopatric speciation the "null hypothesis" of the field for decades, and Coyne and Orr's influential 2004 synthesis reinforced this position with extensive biogeographic data.1, 2
The most direct approach to estimating the relative frequency of speciation modes is to examine the geographic ranges of sister species — pairs of species that are each other's closest living relatives. If speciation is predominantly allopatric, young sister species should have non-overlapping (allopatric) ranges, with range overlap increasing over time as species expand their distributions after speciation is complete. Barraclough and Vogler's 2000 analysis of range-overlap patterns across multiple animal groups found exactly this pattern: the youngest sister species pairs tended to have completely non-overlapping ranges, with the degree of range overlap increasing with the age of the species pair, consistent with an allopatric origin followed by post-speciation range expansion.18
Geographic range overlap in sister species pairs2, 18
| Range overlap category | Definition | Proportion of sister pairs | Interpretation |
|---|---|---|---|
| Fully allopatric | 0% range overlap | >70% | Consistent with allopatric origin |
| Partially overlapping | 1–89% range overlap | ~20% | Secondary contact or parapatry |
| Highly sympatric | ≥90% range overlap | <10% | Potentially sympatric origin |
These biogeographic data strongly suggest that allopatric speciation is the predominant mode in animals, with fewer than 10 percent of sister species pairs showing the degree of range overlap expected under sympatric speciation.2, 18 However, Fitzpatrick and colleagues cautioned in 2008 that the geographic classification itself is problematic because the current distributions of species may not reflect the conditions under which speciation occurred. Post-speciation range shifts can cause allopatrically formed species to become sympatric, and sympatrically formed species to become allopatric, obscuring the signal of the original speciation mode.24
In plants, the picture is different. Polyploidy — whole-genome duplication — provides a mechanism for instantaneous sympatric speciation, because a newly formed polyploid individual is immediately reproductively isolated from its diploid parent population by differences in chromosome number. A 2009 analysis estimated that approximately 15 percent of angiosperm speciation events and 31 percent of fern speciation events are accompanied by increases in ploidy, indicating that sympatric speciation through polyploidy is a major contributor to plant diversity.23 Additional cases of sympatric speciation in plants have been documented in non-polyploid systems, such as the two endemic species of Howea palm on Lord Howe Island, which diverged through adaptation to different soil types and a resulting shift in flowering time, without any geographic barrier on the tiny oceanic island.20
Genomic evidence and speciation with gene flow
The advent of whole-genome sequencing has transformed the study of speciation by revealing that the traditional dichotomy between allopatric and sympatric speciation is often an oversimplification. Genomic data from numerous study systems have demonstrated that gene flow frequently continues during the speciation process — a phenomenon termed speciation-with-gene-flow — blurring the distinction between geographic modes.19
In the Rhagoletis system, genomic analyses have revealed that divergence between the apple and hawthorn host races is concentrated in specific chromosomal regions, particularly within inversions, while the rest of the genome remains relatively undifferentiated. This pattern of heterogeneous genomic divergence is a hallmark of speciation-with-gene-flow: natural selection maintains differentiation at loci underlying adaptation and reproductive isolation, while recombination and gene flow homogenise neutral or weakly selected regions.4, 19 The concept of divergence hitchhiking — in which physical linkage to a locus under divergent selection protects nearby loci from the homogenising effects of gene flow — explains why chromosomal inversions, which suppress recombination across large genomic regions, play an outsized role in speciation-with-gene-flow.19
The genomic revolution has similarly transformed understanding of Darwin's finch radiation. Whole-genome data revealed that hybridisation and introgression have been pervasive throughout the radiation, with adaptive alleles being shared between species through interspecific gene flow.17 Far from being a simple case of allopatric divergence followed by reproductive isolation, the finch radiation appears to involve repeated cycles of divergence, secondary contact, gene flow, and further divergence — a reticulate process in which the boundary between "allopatric" and "sympatric" phases is continuously shifting.16, 17
The emerging consensus is that speciation is best understood not as a single geographic event but as an extended process during which reproductive isolation accumulates gradually, and gene flow may persist at varying levels throughout much of this process. The critical question is not whether populations are geographically separated at some instant, but whether the ecological and genetic conditions are sufficient for divergent selection to overcome the homogenising effects of gene flow and drive the accumulation of reproductive barriers.13, 14, 19 Ecological speciation — the process by which reproductive isolation evolves as a consequence of divergent natural selection between ecological environments — can operate across the entire continuum from complete allopatry to full sympatry, and its genomic signature is becoming increasingly recognisable through the pattern of "islands of divergence" in an otherwise undifferentiated genome.14, 19
Documented examples of speciation modes across study systems2, 11
Synthesis
The study of speciation modes has evolved from a polarised debate between advocates of allopatric and sympatric speciation into a more nuanced understanding of speciation as a continuous process shaped by the interplay of geography, ecology, and genetics. Allopatric speciation remains the best-supported and most common mode in animals, as demonstrated by the overwhelming preponderance of allopatric sister species pairs in biogeographic analyses.1, 2, 18 Sympatric speciation, once considered nearly impossible, has been convincingly documented in a handful of systems that meet the stringent criteria proposed by Coyne and Orr, including Rhagoletis host races, crater lake cichlids, and Howea palms, and is a routine mechanism in plants through polyploidy.3, 4, 5, 6, 20, 23 Parapatric speciation and ring species illustrate that divergence can occur along environmental gradients without complete geographic separation.8, 9
Perhaps the most important insight from the genomic era is that speciation rarely conforms neatly to any single geographic category. Gene flow during divergence appears to be the rule rather than the exception, and the critical determinants of whether speciation succeeds are the strength of divergent selection, the genetic architecture of reproductive isolation, and the degree to which ecologically important loci are protected from the homogenising effects of recombination and migration.13, 14, 19 The geographic framework pioneered by Mayr remains indispensable for organising our understanding of speciation, but the clean categorical boundaries it implies have given way to a richer, more continuous picture of how new species are born.1, 24
References
Sympatric host race formation and speciation in frugivorous flies of the genus Rhagoletis (Diptera, Tephritidae)
Genetic differentiation between sympatric host races of the apple maggot fly Rhagoletis pomonella
Evolutionary relationships within the Ensatina eschscholtzii complex confirm the ring species interpretation
Whole-genome sequences of Malawi cichlids reveal multiple radiations interconnected by gene flow