Overview
- Domestication is artificial selection operating over human timescales, and it produces the same evolutionary phenomena—heritable variation, directional selection, adaptive change, correlated responses, and rapid morphological divergence—that Darwin identified as the operating principles of natural selection.
- The Belyaev fox experiment, initiated in 1959 in Siberia, demonstrated within 20 generations that selecting solely for tameness produced an entire suite of correlated physical changes including floppy ears, piebald coats, and curled tails, illustrating pleiotropy and the concept of domestication syndrome.
- Crop domestication has produced transformations so extreme—teosinte to modern maize, wild mustard to broccoli, cabbage, kale, and cauliflower—that the creationist claim that selection can only eliminate variation is empirically falsified: domestication demonstrably generates novel morphologies, behaviors, and gene expression patterns never present in wild ancestors.
When Charles Darwin opened On the Origin of Species in 1859, he did not begin with fossils, biogeography, or the geological record. He began with pigeons.1 The choice was deliberate and rhetorically powerful. Every Victorian gentleman familiar with the pigeon fancy—the elaborate culture of selective breeding that had produced fantails, tumblers, pouters, carriers, and jacobins from a single ancestral species, Columba livia—already understood in practice what Darwin was claiming in principle: that heritable variation, when consistently favored across generations, could reshape organisms far beyond the range seen in any wild population. Domestication was not merely an analogy for natural selection. It was evolution made visible, evolution operating on a human timescale, evolution that any reader could verify by walking into a livestock market or a poultry show. Darwin returned to domestic animals and plants throughout his career, devoting two entire volumes to the subject in 1868 with The Variation of Animals and Plants under Domestication. The evidence accumulated since his time has only deepened the case. Domestication is one of the most powerful demonstrations in all of science that evolution is not a theoretical abstraction but an empirical process—observable, repeatable, and mechanistically understood.
Darwin and the pigeon fancy
Darwin spent years studying domestic pigeons firsthand, corresponding with breeders, joining pigeon clubs in London, and maintaining his own dovecote at Down House. His central argument was that the extraordinary morphological diversity of domestic pigeon breeds—differences in beak length, body size, tail feather number, skull shape, and wing anatomy that exceed the differences between many wild bird genera—all derived from a single wild ancestor.1, 16 If a process as undirected as human preference for novelty could generate that magnitude of divergence in a few centuries, what might millions of years of consistent environmental selection produce? The logic was transparent and devastating to the argument that species were fixed.
Modern genomic analysis has confirmed Darwin’s inference completely. Whole-genome sequencing of domestic pigeon breeds and their wild relatives demonstrates that all domestic Columba livia forms descend from a common ancestor, with breed-specific phenotypes traceable to a small number of genetic loci under strong artificial selection.16 The crested head feathers found in certain breeds, for instance, map to a single regulatory mutation in the gene EphB2 that reverses follicle orientation—a novelty absent in any wild rock pigeon population that arose entirely through the accumulation of mutations and their selection by breeders.16 This is not the elimination of variation; it is the generation of variation followed by its fixation through selection. The mechanism Darwin proposed and the mechanism genomics has revealed are the same.
The Belyaev fox experiment
The most controlled and scientifically rigorous demonstration of domestication as evolution began in Novosibirsk, Siberia in 1959, when the Soviet geneticist Dmitri Belyaev initiated a long-term selective breeding program on silver foxes (Vulpes vulpes).3, 4 Belyaev selected from a fur-farm population, choosing for breeding only those individuals that tolerated human approach and contact without aggression or fear—a single behavioral criterion, tameness, applied rigorously each generation. He selected against tameness in a parallel control line. The experiment was designed to test whether domestication syndrome—the cluster of physical and behavioral traits that recurrently appears across independently domesticated species—would emerge as a correlated response to selection on behavior alone.
Within 10 to 15 generations, the selected foxes began displaying behaviors never observed in wild or fur-farm foxes: tail-wagging, whimpering, and actively seeking human attention in the manner of domestic dogs.4 Within 20 generations, the physical changes were unmistakable.3 Selected foxes showed floppy, drooping ears; shortened, curled tails; piebald black-and-white coat patterns; shortened snouts and wider skulls; and in females, earlier onset of sexual maturity. None of these traits had been selected directly. They appeared as correlated responses to selection on tameness alone—a textbook demonstration of pleiotropy, the phenomenon by which a single gene or genetic regulatory pathway influences multiple phenotypic traits simultaneously.15
Belyaev’s hypothesis, which he called destabilizing selection, proposed that tameness-related genes were linked developmentally to the same pathways controlling pigmentation, morphology, and reproductive timing.3 The modern explanation, articulated most fully by Adam Wilkins, Richard Wrangham, and Tecumseh Fitch in 2014, centers on neural crest cells—the embryonic cell population that migrates from the dorsal neural tube to give rise to adrenal glands, pigmentation cells, craniofacial cartilage, and parts of the peripheral nervous system.15 Reduced reactivity of the hypothalamic-pituitary-adrenal axis (selecting for tameness) correlates with reduced neural crest cell numbers or migration, which simultaneously affects pigmentation (producing white patches where melanocytes fail to reach), craniofacial morphology (producing shortened snouts), and ear cartilage stiffness (producing floppy ears). The suite of domestication syndrome traits across species—dogs, cattle, pigs, horses, sheep, and the experimental foxes alike—reflects a shared developmental architecture, not independent selective histories.15
Genomic analysis of the Belyaev foxes has identified specific regions of the genome differentiating the tame and aggressive lines, including loci near genes involved in serotonin and glutamate signaling, and a region on chromosome 15 strongly associated with tame behavior.5 The experiment continues to the present day at the Institute of Cytology and Genetics in Novosibirsk and remains the only controlled replicated experiment in which a wild animal has been actively domesticated under documented laboratory conditions. Its conclusions are unambiguous: selection on a single behavioral trait reshapes an entire organism through the interconnected architecture of development, producing novel morphologies within tens of generations rather than thousands.
Dogs from wolves: diversification from a single ancestor
The domestic dog, Canis lupus familiaris, is a subspecies of the gray wolf, Canis lupus.6 Every domestic dog breed—from the 1.5-kilogram Chihuahua to the 90-kilogram Saint Bernard, from the flat-faced English bulldog to the long-snouted borzoi, from the hairless Xoloitzcuintli to the heavily coated Siberian husky—descends from a single wolf population through a domestication event or events estimated to have occurred between 15,000 and 40,000 years ago.6, 7, 8 The precise geographic origin and number of founding events remain debated; ancient DNA evidence has pointed to a single domestication from a now-extinct wolf population in either East Asia or western Eurasia, with subsequent migrations accompanying human dispersal.7, 17
What is not debated is the scale of the transformation. The morphological distance between the smallest and largest dog breeds exceeds that between wolves and foxes—two species that shared a common ancestor approximately 6 to 10 million years ago—yet all of this variation arose within a single species, within a timescale representing a geological eyeblink.6 The behavioral divergence is equally dramatic. Dogs have evolved exceptional sensitivity to human social cues, including the ability to follow gaze direction and pointing gestures, capacities that wolves raised alongside humans do not develop to the same degree.6 Genetic analyses have identified signatures of selection on genes involved in starch digestion, including multiple copies of the amylase gene AMY2B in dogs relative to wolves, reflecting adaptation to the high-starch diet of human settlements.6 Dogs are not merely tamed wolves. They are evolutionarily diverged organisms whose genomes, development, behavior, and physiology have been reshaped by millennia of selection in a human ecological context—a process indistinguishable in mechanism from the natural selection operating on wild populations.
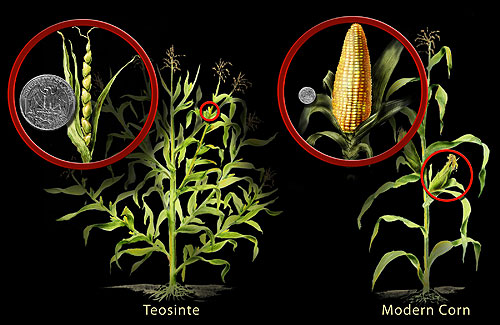
Maize from teosinte: regulatory evolution and morphological transformation
Among all the products of plant domestication, none is more morphologically radical than the transformation of teosinte into modern maize (Zea mays ssp. mays). Teosinte, the wild grass from which maize was domesticated in the Balsas River valley of southwestern Mexico approximately 9,000 years ago, is so different in appearance from domesticated corn that when botanists first encountered it in the nineteenth century they classified it as a distinct genus.12 Teosinte is a bushy, multi-stemmed plant bearing hard, triangular kernels enclosed in a stony fruit case and arranged in two-ranked spikes of five to twelve seeds. Modern maize is a single-stalked plant producing large, multi-rowed ears with hundreds of naked, starch-filled kernels exposed at maturity. The transformation of one into the other required changes in branching architecture, ear size, kernel composition, kernel exposure, and reproductive timing so comprehensive that it constitutes one of the most dramatic examples of rapid morphological change in the entire fossil and domestication record.
The genetic basis of this transformation, worked out through quantitative trait locus mapping and molecular genetics over the past three decades, reveals something unexpected: much of it is attributable to regulatory changes in a small number of key genes rather than to wholesale genomic restructuring.12 The gene teosinte branched1 (tb1), a TCP-class transcription factor, controls lateral branching in grasses. In teosinte, tb1 is expressed at moderate levels and permits the development of the numerous lateral branches that give the plant its bushy habit. In domesticated maize, a transposable element inserted approximately 60,000 base pairs upstream of the tb1 coding sequence acts as an enhancer that roughly doubles the gene’s expression level, suppressing lateral branching and channeling the plant’s energy into a single dominant stalk with a large terminal ear.10 The entire architectural revolution that distinguishes maize from teosinte traces in substantial part to this single regulatory change—a change not in the protein-coding sequence but in a cis-regulatory element controlling when and how much of the gene is expressed.
The second major transformation, the exposure of the maize kernel, is controlled by the gene tga1, which encodes a squamosa promoter binding protein. In teosinte, the hard glume (fruit case) enclosing each kernel is controlled by the wild-type tga1 allele. A single amino acid substitution in the domesticated tga1 allele converts the enclosed teosinte seed into the naked kernel of maize, fundamentally changing the plant’s suitability for human consumption and the ease of its harvest.9 Here, by contrast with tb1, the molecular change is a protein-coding substitution rather than a regulatory one. The evolution of maize thus illustrates both major categories of molecular change that drive morphological evolution: changes in gene expression through regulatory sequences and changes in protein function through coding sequence variation.11
The implications for the creationist claim that “selection can only remove information” are direct. The tb1 enhancer in maize is a new functional element—a transposable element insertion that created a novel cis-regulatory sequence at a new genomic location, expanding the functional repertoire of the locus. This is not the removal of information; it is the addition of a new regulatory module with phenotypic consequences of dramatic magnitude. The transposable element was already present in the genome as a structural entity, but its integration at this specific location, conferring this specific function, is a novel event.10 Selection then fixed this novel variant because the altered branching architecture benefited the humans who cultivated the plant. Mechanism, outcome, and timescale are all precisely what evolutionary theory predicts.
Brassica oleracea: one species, six vegetables
The wild mustard Brassica oleracea, native to the coastal limestone cliffs of western Europe and the Mediterranean, is a single species from which human selection has derived—independently, repeatedly, and in different geographic contexts—broccoli, cauliflower, cabbage, kale, Brussels sprouts, kohlrabi, and collard greens.13, 14 Each of these crops represents a different organ or developmental process that has been hypertrophied by selection: cabbage is an enlarged terminal bud; Brussels sprouts are enlarged axillary buds arranged along the stem; kohlrabi is an enlarged stem; broccoli and cauliflower are proliferating floral meristems arrested before opening; kale is an expansion of the leaf blade. The underlying developmental toolkit is identical across all forms. What differs is the allocation of growth resources, controlled by the regulatory genes governing meristem identity and organ size.14
The Brassica oleracea crop complex is particularly instructive as a refutation of the claim that selection can only reduce diversity. Starting from the same narrow wild ancestor, operating in different regions under different human preferences, selection has produced organisms so phenotypically distinct that a person encountering them without botanical knowledge would have no reason to conclude they were the same species. Comparative genomic analysis of the major B. oleracea crop groups confirms that the phenotypic divergence reflects selection at regulatory loci controlling development, and that much of the variation segregating in cultivated forms exceeds the genetic variation present in surveyed wild populations—a consequence of human cultivation exposing and fixing rare variants that would be eliminated by stabilizing selection in nature.14
Rapid evolution and the domestication syndrome
Across the full breadth of domesticated species—dogs, cats, cattle, pigs, horses, sheep, goats, chickens, rice, wheat, maize, sorghum, sunflower, and dozens of others—a consistent pattern emerges that biologists call the domestication syndrome: a suite of traits that recurrently appears in domesticated organisms regardless of the wild ancestor or the human culture doing the domesticating.15 In animals, the syndrome typically includes reduced adrenal stress response, increased docility, altered coat coloration (especially white patches and piebald patterns), floppy ears (in mammals with upright wild-type ears), shortened faces and smaller teeth, reduced brain volume relative to body mass, and earlier or more flexible breeding seasons. In plants, the syndrome includes increased seed size, loss of seed dispersal mechanisms, synchronous germination, loss of dormancy, and altered photoperiod responses.2, 12
The convergent appearance of these traits across independently domesticated species is a powerful signal of shared developmental constraint. It indicates that the traits are not independent, each requiring a separate selective event, but are instead linked through common developmental pathways—most probably the neural crest pathway in animals and analogous meristem-regulatory pathways in plants—such that selection on one trait reliably produces correlated changes across the syndrome as a whole.15 This is the mechanism Darwin intuited when he wrote about correlated variation: the organism is not a collection of independent traits but an integrated developmental system, and selection pulling on one element of the system will deform others. Domestication demonstrates this principle with a clarity and experimental replicability that no natural population can match.
The pace of domestication-driven change is also evolutionarily informative. Dog breeds have diverged in body size, skull shape, and limb proportion more rapidly than most wild mammal lineages in the fossil record.6 Maize was transformed from teosinte into a recognizably domesticated crop within a few thousand years at most.12 The Belyaev foxes displayed behavioral domestication within ten generations and morphological domestication syndrome within twenty.3, 4 These timescales demonstrate that the heritable variation present in natural populations is more than sufficient to fuel large-scale evolutionary change within spans too short to require any new mutation—standing variation, acted upon by consistent directional selection, is adequate to produce transformations that would be unrecognizable without knowledge of the starting point. This is one of the reasons that Darwin’s decision to open with domestic species was not rhetorical convenience but analytical precision: domestication is the natural experiment that proves the sufficiency of selection acting on ordinary heritable variation.
Domestication and the limits of artificial selection objections
A recurring claim in creationist and intelligent-design literature holds that while artificial and natural selection can eliminate variation and produce minor modifications within a type, they cannot generate genuinely novel structures, functions, or information. On this view, dog breeds are all still dogs, maize is still a grass, and no amount of selection will transform one fundamental type into another. The claim has rhetorical appeal but is empirically incorrect on multiple levels.
First, the factual claim that domestication produces only reduction is straightforwardly falsified. The tb1 enhancer in maize is a new genomic regulatory element created by transposable element insertion and fixed by selection—a molecular novelty not present in the ancestral teosinte population.10 The behavioral repertoire of domestic dogs, including their exceptional sensitivity to human social cues and their capacity for cooperative communication with humans, is a genuinely novel suite of capabilities not present in wolves and not explicable as the simple subtraction of wolf behaviors.6 Domestication syndrome traits like piebald pigmentation in the Belyaev foxes are new phenotypes arising as correlated responses to selection—not the removal of existing pigmentation patterns but the generation of new ones through altered neural crest migration.5, 15
Second, the implicit standard in the objection—that evolution must be observed crossing a “kind boundary” to count—is definitionally unfalsifiable and scientifically incoherent. The objection defines kinds circularly such that any change within the range that has been observed is declared insufficient by definition. But the mechanisms demonstrated in domestication are identical to those proposed in evolutionary theory: heritable variation in regulatory and coding sequences, directional selection, correlated responses through developmental integration, and the progressive accumulation of changes across generations. The observation that maize remains a grass after 9,000 years of domestication does not challenge evolutionary theory, which makes no claim that 9,000 years of directional selection on a small number of traits will transform a grass into a non-grass. What 9,000 years of selection has produced—a plant so different from its ancestor that it was classified as a distinct genus on morphological grounds—is precisely the magnitude of change that evolutionary theory predicts for that timescale.12
Third, the domestication record, viewed over archaeological timescales, documents a continuum of change in which populations that were biologically indistinguishable from their wild relatives ten thousand years ago have become, in their domesticated descendants, morphologically, behaviorally, physiologically, and genomically distinct organisms. The artificial selection driving this change differs from natural selection in direction and in the agent doing the selecting, not in mechanism. If the former produces evolution—and the evidence that it does is overwhelming—then the latter produces evolution for precisely the same reasons. Darwin’s opening gambit with the pigeon fancy was not a rhetorical trick. It was the identification of a publicly visible, empirically accessible instance of the very process whose operation on wild populations he spent the rest of his book demonstrating.
Domestication in the broader evidence for evolution
Domestication occupies a specific and irreplaceable role in the evidentiary structure of evolutionary biology. The fossil record provides the temporal depth that domestication cannot, demonstrating change across millions of years and multiple lineages. Molecular phylogenetics provides the genealogical framework that domestication presupposes. Population genetics provides the mathematical theory that makes quantitative predictions about rates of change. Domestication provides something none of these can offer independently: a controlled, documented, human-observed instance of evolution in which the agent of selection, the starting population, the selective criterion, and the outcome are all known.1, 4
The Belyaev experiment is especially significant in this regard because it is not merely an observation of change that happened to occur under human oversight, as with all agricultural domestication; it is an experiment designed with controls, replicated lines, and a specific hypothesis about mechanism.3, 4 The tame and aggressive fox lines provide the comparison that allows researchers to attribute phenotypic differences to selection on tameness rather than to drift, founder effects, or other confounds. This is as close as evolutionary biology gets to a classical controlled experiment, and its results are unambiguous: directional selection on a behavioral trait produces rapid, heritable, morphologically dramatic change through the developmental architecture of the organism, generating novel phenotypes not present in the starting population.
Domestication also makes the mechanisms of evolution concrete in a way that abstract population genetic models cannot. When a farmer selects the largest-seeded individuals from a wheat harvest to plant the following year, the logic of heritable variation and differential reproduction is tangibly present. When a dog breeder selects for a shortened face across generations and produces a brachycephalic breed, the cumulative power of selection on continuously varying traits is directly observable. Darwin grasped this communicative power in 1859 and used it to make the case for natural selection to a readership that had no molecular genetics, no population genetics, and no paleogenomics. The case is even stronger now. Every genome sequenced from a domesticated species, every regulatory element mapped, every domestication syndrome trait mechanistically explained, adds another layer of confirmation to the framework that domestication exemplifies: evolution is the change in heritable characteristics of populations across generations, driven by selection, and its products, whether wrought by nature or by human hands, are the same in kind.
References
A key developmental transition during the evolution of maize is regulated by transcriptional regulation of teosinte branched1
Domestication syndrome in mammals: A unified explanation based on neural crest cell behavior and genetics