Overview
- Sexual reproduction imposes a severe theoretical cost because sexual females produce both sons and daughters while asexual females convert all reproductive effort into clonal daughters, yet sex is nearly universal among eukaryotes, a discrepancy that constitutes one of the deepest puzzles in evolutionary biology.
- The leading hypotheses for the maintenance of sex invoke its role in generating genetic variation that accelerates adaptation, clears deleterious mutations, and enables organisms to coevolve with rapidly changing parasites, with the Red Queen hypothesis receiving strong experimental support from systems such as New Zealand mud snails and laboratory nematodes.
- The fossil record indicates that sexual reproduction originated in eukaryotes more than one billion years ago, and the rarity of truly ancient asexual lineages suggests that sex provides long-term benefits that asexual populations cannot match.
Sexual reproduction, in which offspring arise from the fusion of gametes produced by two parents through the specialized cell division called meiosis, is the dominant mode of reproduction among eukaryotic organisms. It is found in animals, plants, fungi, and most protists, and its near-universality has long struck biologists as remarkable because sexual reproduction carries severe theoretical costs relative to asexual alternatives. An asexual female that produces clonal daughters transmits her entire genome to every offspring, whereas a sexual female shares only half her genes with each child and must invest resources in producing males that do not themselves bear young.1, 2 This disparity, known as the twofold cost of sex, implies that asexual mutants should rapidly outcompete sexual populations whenever they arise. Yet sex persists across nearly the entire eukaryotic tree of life, and truly ancient asexual lineages are exceedingly rare. Explaining why organisms pay the cost of sex rather than reproducing clonally is one of the most enduring problems in evolutionary biology, sometimes called the "queen of problems" in the field.3
Over the past half-century, a rich body of theoretical and experimental work has produced several compelling hypotheses for the maintenance of sex, each highlighting a different advantage of the genetic recombination that sex makes possible. These hypotheses are not mutually exclusive, and current thinking favours a pluralistic view in which multiple mechanisms operate simultaneously to sustain sexual reproduction across the diversity of life.4, 5

The paradox of sex
The modern formulation of the problem dates to the 1970s, when George C. Williams and John Maynard Smith independently drew attention to the enormous cost that sex imposes on individual fitness. Williams, in his 1975 book Sex and Evolution, argued that a female who reproduces asexually transmits twice as many of her genes to the next generation as a female who reproduces sexually, because the sexual female "wastes" half her reproductive effort on sons who do not directly produce offspring of their own.2 Maynard Smith formalised this logic in his 1978 monograph The Evolution of Sex, coining the phrase "twofold cost of males" to describe the demographic disadvantage: in a population with an equal sex ratio, an asexual mutant that produces only daughters should double in frequency each generation relative to the sexual population, rapidly driving the sexuals to extinction.1
The cost applies most directly to species with separate sexes (termed dioecious in plants and gonochoristic in animals) and an equal sex ratio. In species that are hermaphroditic or that reproduce through self-fertilization, the cost is reduced but not eliminated, because meiosis itself entails a "cost of meiosis" identified by Williams: each parent contributes only half its genome to each offspring, breaking up favourable gene combinations that have been tested by natural selection.2 The theoretical costs of sex are thus twofold in their broadest sense: sex dilutes the parent's genetic representation in the next generation and disrupts co-adapted gene complexes that have proven their worth. Any explanation for the maintenance of sex must identify benefits large enough to offset these considerable costs.4
Experimental confirmation that the twofold cost is not merely a theoretical abstraction came from work on the freshwater snail Potamopyrgus antipodarum in New Zealand, where sexual and asexual lineages coexist in the same populations. Gibson, Delph, and Lively demonstrated in 2017 that asexual females did indeed produce approximately twice the number of offspring as sexual females under controlled conditions, directly measuring the demographic cost that Maynard Smith had predicted four decades earlier.16
The variation hypotheses: Weismann, Fisher, and Muller
The oldest class of explanations for the advantage of sex centres on the idea that recombination generates genetic variation, providing raw material for natural selection to act upon. August Weismann proposed in 1886 that the function of sex was to create individual differences among offspring, allowing populations to adapt more rapidly to changing environments. This intuition was developed formally in the twentieth century by Ronald Fisher and Hermann Muller, who independently argued that sex is advantageous because it allows beneficial mutations arising in different individuals to be combined into the same genome.9
In an asexual population, two beneficial mutations that arise in different lineages cannot combine: each must compete with the other for fixation in a process called clonal interference. Only one lineage can ultimately prevail, and the beneficial mutation in the losing lineage is lost. In a sexual population, recombination can bring the two beneficial mutations together in a single individual, allowing both to spread simultaneously and accelerating the rate of adaptation.9 This is sometimes called the Fisher-Muller hypothesis.
Felsenstein unified these ideas in a landmark 1974 paper by showing that the Fisher-Muller advantage and a related phenomenon, Muller's ratchet (discussed below), are both manifestations of a single underlying principle: Hill-Robertson interference, the reduction in the efficacy of selection at one locus caused by selection or drift at linked loci. In finite populations, random associations between alleles at different loci (linkage disequilibrium) are constantly generated by genetic drift, and these associations interfere with selection at each individual locus. Recombination breaks down these random associations, allowing selection to operate more efficiently on each locus independently.9
Experimental tests have confirmed that sex accelerates adaptation. Goddard, Godfray, and Burt used genetically engineered strains of the yeast Saccharomyces cerevisiae that differed only in their ability to undergo sexual recombination, and found that sexual populations adapted significantly faster than otherwise identical asexual populations in a challenging environment.10 McDonald, Rice, and Desai extended this work in 2016, showing that sex not only speeds adaptation but alters its molecular signature, because recombination allows natural selection to more efficiently sort beneficial from deleterious mutations that would otherwise be locked together on the same chromosome.11
Muller's ratchet
A complementary argument focuses not on the accumulation of beneficial mutations but on the clearance of deleterious ones. Hermann Muller proposed in 1964 that asexual populations are subject to an irreversible accumulation of harmful mutations, a process he termed the "ratchet." In any finite population, the class of individuals carrying the fewest deleterious mutations can be lost by chance through genetic drift. In a sexual population, recombination can recreate mutation-free genotypes by combining the least-mutated portions of different parental genomes. In an asexual population, however, once the least-loaded class is lost, it cannot be regenerated: the ratchet has clicked forward one notch, and the minimum mutation load in the population has irreversibly increased.9, 5
Over many generations, the repeated operation of Muller's ratchet drives a relentless accumulation of slightly deleterious mutations in asexual lineages, progressively eroding their fitness. The rate at which the ratchet turns depends on the population size, the mutation rate, and the severity of mutational effects: it operates fastest in small populations with high mutation rates. Muller's ratchet provides a compelling explanation for the observed degeneration of non-recombining genomic regions, including the Y chromosome in mammals and the W chromosome in birds, both of which have lost the vast majority of their functional genes over evolutionary time.5
Alexey Kondrashov extended the mutational argument in 1988 with what is sometimes called the deterministic mutation hypothesis or "Kondrashov's hatchet." Kondrashov showed that if deleterious mutations interact synergistically, meaning that the fitness cost of carrying multiple mutations is greater than the sum of their individual effects (a form of epistasis), then sexual reproduction is advantageous even in infinite populations because recombination concentrates mutations into some individuals while producing others with few mutations.8 Selection can then efficiently remove the heavily loaded individuals, purging multiple mutations simultaneously. The hypothesis requires that the genomic deleterious mutation rate exceeds approximately one per generation and that epistasis among deleterious mutations is synergistic. Evidence on whether these conditions are met in natural populations remains mixed, with some organisms showing the required synergistic epistasis and others showing no epistasis or even antagonistic interactions.5, 8
The Red Queen hypothesis
The Red Queen hypothesis, named after the character in Lewis Carroll's Through the Looking-Glass who must keep running just to stay in the same place, proposes that sex is maintained by the coevolutionary arms race between hosts and parasites. The hypothesis was developed most influentially by W. D. Hamilton and colleagues, who argued that parasites evolve to exploit the most common host genotypes in a population. In an asexual population, all individuals are genetically identical or very similar, making the entire population vulnerable to a parasite adapted to that genotype. Sexual reproduction, by generating genetically diverse offspring through recombination, produces rare genotypes that parasites have not yet evolved to exploit, conferring a frequency-dependent advantage on sexual lineages.6

The Red Queen hypothesis predicts that sex should be most common where parasite pressure is greatest, and that the relative frequency of sexual versus asexual reproduction should fluctuate in response to changes in parasite prevalence. Both predictions have received support from studies of the New Zealand freshwater snail Potamopyrgus antipodarum, a species in which sexual diploid and asexual triploid females coexist in the same lakes. In populations where infection by sterilizing trematode parasites (Microphallus spp.) is common, sexual snails predominate; in populations with low parasite pressure, asexual snails are more frequent.7, 16 Long-term tracking of these populations has shown that common asexual clones are disproportionately infected, consistent with the hypothesis that parasites track common genotypes and that sex generates the rare genotypes needed to escape infection.7
The most direct experimental test of the Red Queen hypothesis was conducted by Morran, Schmidt, Gelarden, Parrish, and Lively in 2011 using the nematode Caenorhabditis elegans and its bacterial pathogen Serratia marcescens. C. elegans normally reproduces primarily by self-fertilization but can also outcross (mate with males). When populations were coevolved with a virulent pathogen, the rate of outcrossing increased dramatically, from less than 20 percent to over 80 percent within just 30 generations. Obligately selfing populations, which could not outcross, were driven to extinction by the coevolving pathogen, whereas outcrossing populations persisted through reciprocal adaptation.7 This experiment provided powerful evidence that parasite coevolution can directly select for sexual reproduction.
The DNA repair hypothesis
A distinct class of explanations proposes that the original function of the recombination machinery underlying sex was not to generate genetic variation but to repair damage to DNA. Harris Bernstein, Frederick Hopf, and Richard Michod argued that meiotic recombination, the process by which homologous chromosomes exchange segments during the production of gametes, originated as a mechanism for repairing double-strand breaks and other severe forms of DNA damage. A damaged DNA molecule can be accurately repaired using the homologous chromosome as a template, and recombinational repair is the only known mechanism capable of correcting double-strand breaks, which are lethal to a cell if left unrepaired.12
The DNA repair hypothesis draws support from the observation that the molecular machinery of meiotic recombination is homologous to the machinery used for recombinational DNA repair in bacteria, suggesting a deep evolutionary continuity between prokaryotic DNA repair processes and eukaryotic sex. Furthermore, many organisms increase their rates of sexual reproduction under conditions of environmental stress that cause elevated DNA damage, such as exposure to ultraviolet radiation or desiccation, consistent with the idea that sex functions in part as a DNA repair mechanism.12, 15
The DNA repair hypothesis is not necessarily in conflict with the variation-based hypotheses. It is possible that sex originated primarily as a DNA repair mechanism and was subsequently maintained because of the additional advantages conferred by the genetic variation it produces. The hypothesis does, however, offer a plausible account of why the recombination machinery evolved in the first place, a question that the variation hypotheses do not directly address.12
Origin and antiquity of sex
The fossil and molecular evidence indicates that sexual reproduction is ancient, originating in the common ancestor of all eukaryotes over one billion years ago. The oldest direct fossil evidence of sexual reproduction comes from the filamentous red alga Bangiomorpha pubescens, described by Nicholas Butterfield from approximately 1.05-billion-year-old rocks of the Hunting Formation in Arctic Canada. The fossil preserves differentiated reproductive structures, including distinct spore and gamete types, that are diagnostic of sexual reproduction in modern bangiacean red algae.14
Molecular evidence suggests that sex is even older than the Bangiomorpha fossils. Genes encoding the core components of the meiotic machinery, including the recombinase Dmc1, the synaptonemal complex proteins, and the meiosis-specific cohesin Rec8, are found across all major eukaryotic supergroups, implying that the last eukaryotic common ancestor (LECA) was already capable of meiosis and sexual reproduction. The deep conservation of these genes suggests that sex originated during the early radiation of eukaryotes, possibly in the Mesoproterozoic era more than 1.5 billion years ago.15
The evolutionary pathway from asexual prokaryotic ancestors to sexual eukaryotes likely involved several intermediate stages. Speijer, Lukes, and Elias have proposed that the earliest form of sex may have involved an increase in ploidy through endoreplication followed by a rudimentary parasexual cycle with gradual chromosome loss, and that three major innovations, cell-cell fusion, meiosis, and the evolution of distinct mating types or sexes, subsequently gave rise to the modern sexual cycle.15 Proteins with central functions in meiosis share structural and functional similarities with proteins involved in natural transformation in bacteria and DNA transfer in archaea, hinting at an evolutionary connection between prokaryotic horizontal gene transfer and eukaryotic sexual recombination.12, 15
Timeline of key events in the evolution of sexual reproduction14, 15
Ancient asexuals and their lessons
If sex provides such important advantages, then lineages that abandon it should be evolutionary dead ends, doomed to extinction by the accumulation of deleterious mutations, vulnerability to parasites, or inability to adapt to changing environments. This prediction is broadly supported by the observation that most known asexual lineages are of recent origin, representing the tips rather than the deep branches of phylogenetic trees. However, a small number of groups appear to have persisted for millions of years without sex, posing a challenge to theories that predict the inevitable demise of asexual lineages.
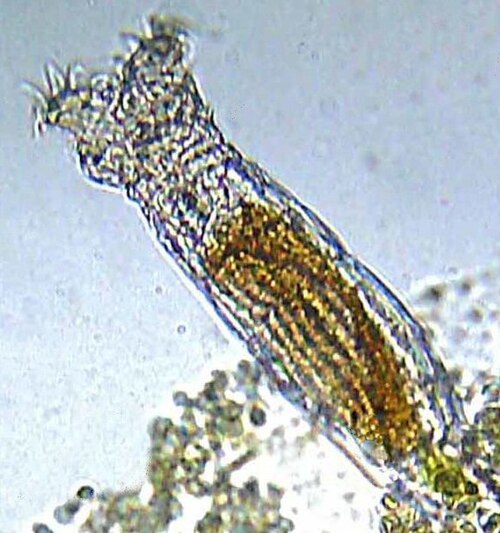
The most celebrated of these so-called "ancient asexuals" are the bdelloid rotifers, a class of microscopic freshwater invertebrates comprising approximately 460 described species. John Maynard Smith referred to them as an "evolutionary scandal" because they appear to have radiated into a diverse clade while remaining entirely asexual. No males, meiosis, or any form of sexual reproduction has ever been observed in any bdelloid species. Molecular analysis by Mark Welch and Meselson in 2000 revealed that the genomes of individual bdelloid rotifers contain divergent copies of genes that appear to have been diverging independently since before the bdelloid radiation, consistent with the absence of recombination for tens of millions of years.13
The bdelloid rotifers may survive without sex in part because they have evolved alternative mechanisms for generating genetic variation and purging deleterious mutations. Bdelloids are extraordinarily resistant to desiccation and ionizing radiation, and their genomes contain an unusually high proportion of genes acquired by horizontal transfer from bacteria, fungi, and plants, suggesting that they may compensate for the absence of sexual recombination through non-sexual genetic exchange.13 Other putative ancient asexuals, including the darwinulid ostracods and certain oribatid mites, are less well-studied but similarly challenge the expectation that all asexual lineages are short-lived.
Nevertheless, the rarity of ancient asexual lineages relative to their sexual counterparts supports the general conclusion that sex confers long-term evolutionary advantages. The vast majority of asexual lineages in animals and plants appear to be recently derived from sexual ancestors, and few persist long enough to undergo significant diversification. This pattern is consistent with the theoretical prediction that sex, despite its short-term costs, provides benefits over evolutionary timescales that asexual reproduction cannot match.3, 5
Toward a pluralistic explanation
No single hypothesis has proven sufficient to explain the maintenance of sex across all organisms and all environments. The Red Queen hypothesis provides a compelling explanation for species under intense parasite pressure but is less obviously applicable to organisms with few parasites. The mutational hypotheses (Muller's ratchet and Kondrashov's hatchet) are most powerful in species with large genomes and high mutation rates but depend on specific assumptions about epistasis that may not hold universally. The Fisher-Muller mechanism accelerates adaptation in changing environments but requires that the environment change sufficiently rapidly relative to the generation time. The DNA repair hypothesis explains the origin of recombination but does not by itself account for why outcrossing (the mating of genetically different individuals) is so common.4, 5
Otto and Lenormand, in an influential 2002 review, argued that a combination of mechanisms is likely responsible for the prevalence of sex, with different factors being more or less important in different taxa and environments. They emphasised that many of the hypotheses are conceptually related, all ultimately invoking the ability of recombination to break down linkage disequilibrium and allow natural selection to act more efficiently on individual loci. From this perspective, the various hypotheses represent different sources of the linkage disequilibrium that recombination eliminates: drift generates random associations (the Hill-Robertson effect), parasites generate negative frequency-dependent selection on linked loci (the Red Queen), and deleterious mutation accumulation generates associations between harmful alleles (Muller's ratchet and Kondrashov's hatchet).4
Hartfield and Keightley reinforced this pluralistic view in 2012, noting that the conditions under which any single hypothesis can fully account for the twofold cost of sex are restrictive, but that the combined effects of multiple mechanisms acting simultaneously can much more readily offset the cost. They argued that the problem of sex may be less paradoxical than it appears once the cumulative weight of multiple small advantages is considered.5
Major hypotheses for the evolution and maintenance of sex4, 5
| Hypothesis | Key proponents | Mechanism | Key requirement |
|---|---|---|---|
| Fisher-Muller | Fisher (1930), Muller (1932), Felsenstein (1974) | Recombination combines beneficial mutations from different lineages | Finite population, multiple beneficial mutations |
| Muller's ratchet | Muller (1964) | Sex prevents irreversible accumulation of deleterious mutations | Finite population, high mutation rate |
| Kondrashov's hatchet | Kondrashov (1988) | Sex purges deleterious mutations efficiently under synergistic epistasis | Genomic mutation rate >1, synergistic epistasis |
| Red Queen | Hamilton (1980, 1990), Lively | Sex generates rare genotypes that escape adapted parasites | Virulent, coevolving parasites with genotype-specific infection |
| DNA repair | Bernstein, Hopf, Michod (1987) | Meiotic recombination repairs double-strand DNA damage | Significant double-strand DNA damage in germline |
| Environmental variation | Williams (1975), Bell (1982) | Variable offspring hedge bets against unpredictable environments | Spatially or temporally variable selection |
Experimental approaches and evidence
The theoretical richness of the sex-evolution literature has been matched in recent decades by increasingly sophisticated experimental tests. Three experimental systems have been particularly informative: yeast, nematodes, and freshwater snails.
In yeast (Saccharomyces cerevisiae), Goddard, Godfray, and Burt created otherwise isogenic strains that differed only in their ability to undergo meiotic recombination and compared their rates of adaptation to a novel stressful environment. Sexual populations consistently achieved higher fitness than asexual populations after several hundred generations, directly demonstrating that sex speeds adaptation in a eukaryotic organism.10 McDonald, Rice, and Desai extended this approach in 2016, tracking the molecular dynamics of adaptation in sexual and asexual yeast populations through whole-genome sequencing. They found that sexual populations fixed beneficial mutations more rapidly and accumulated fewer deleterious "hitchhiker" mutations that were linked to beneficial alleles, confirming that recombination enhances the efficacy of selection at the molecular level.11
The C. elegans experiments of Morran and colleagues provided the most direct test of the Red Queen hypothesis, demonstrating that coevolution with a virulent pathogen selects for increased outcrossing in real time. The rapidity with which obligately selfing populations went extinct under pathogen coevolution, typically within 20 generations, underscored the vulnerability of genetically uniform populations to adapted enemies.7
Field studies of Potamopyrgus antipodarum in New Zealand have provided some of the strongest ecological evidence for the Red Queen. In lakes where trematode parasites are abundant, sexual snails outnumber asexual clones; in lakes with few parasites, asexual clones predominate. Over time, the clones that are most common are also those most heavily parasitized, consistent with the prediction that parasites track and exploit common genotypes.7, 16 The direct measurement of the twofold cost in this system, combined with the ecological correlation between parasitism and sexual frequency, makes Potamopyrgus one of the most complete natural test cases for the evolution of sex.
Collectively, these experimental and observational studies have moved the field from a primarily theoretical enterprise to one grounded in empirical evidence, though no single study has definitively resolved the question. The emerging picture is one in which multiple selective pressures interact to maintain sex, with their relative importance varying across species, populations, and environments.4, 5
Open questions
Despite decades of theoretical and experimental progress, several fundamental questions about the evolution of sex remain unresolved. The precise conditions under which sex first originated in eukaryotes are still debated. While the DNA repair hypothesis provides a plausible molecular scenario for the origin of meiotic recombination, the selective pressures that drove the transition from facultative to obligate sexuality in many lineages remain unclear.15 Why some eukaryotic lineages, such as bdelloid rotifers and certain fungi, have successfully abandoned sex while the vast majority have not is a question that touches on the fundamental architecture of genomes and the population biology of mutation and selection.13
The relative importance of the various hypotheses remains actively debated. The Red Queen hypothesis has received the most direct experimental support, but it is unclear whether host-parasite coevolution alone is sufficient to maintain sex in organisms that face little parasite pressure. The mutational hypotheses depend on parameters, particularly the genomic deleterious mutation rate and the nature of epistatic interactions, that are difficult to measure in most organisms and may vary widely across taxa.5, 8 Whether the pluralistic view, in which multiple small advantages of sex combine to overcome its costs, is the correct resolution or whether some deeper unifying principle underlies all the individual hypotheses remains to be determined.
The evolution of the elaborate machinery surrounding sex, including the evolution of two distinct sexes (anisogamy), the evolution of mate choice and sexual conflict, and the evolution of the remarkable diversity of mating systems observed across the tree of life, represents a separate set of questions that build upon the more fundamental question of why sex exists at all.4 The evolution of sex remains a field in which theoretical insight, natural history, and experimental ingenuity continue to interact productively, and the coming decades will likely see new hypotheses and tests emerge as genomic tools make it possible to study recombination, mutation, and parasite interactions at unprecedented resolution.5, 11
References
Evidence for the evolution of bdelloid rotifers without sexual reproduction or genetic exchange
Bangiomorpha pubescens n. gen., n. sp.: implications for the evolution of sex, multicellularity, and the Mesoproterozoic/Neoproterozoic radiation of eukaryotes