Overview
- The Red Queen hypothesis, proposed by Leigh Van Valen in 1973, holds that organisms must continuously adapt and evolve not merely to gain reproductive advantage but simply to maintain their existing fitness relative to the other species with which they interact, because the biotic environment itself is constantly changing as those other species evolve in turn.
- The hypothesis has become central to explaining the evolution and maintenance of sexual reproduction: by generating genetically diverse offspring through recombination, sexual populations can better resist rapidly coevolving parasites, whereas clonal lineages are quickly tracked and exploited by pathogens that adapt to their common genotypes.
- Empirical support comes from diverse systems including New Zealand mud snails, Daphnia-parasite archives in lake sediments, MHC diversity driven by pathogen pressure, and experimental evolution in nematodes, while alternative hypotheses such as Muller's ratchet and the role of environmental change continue to shape debate about the relative importance of biotic versus abiotic drivers of evolutionary change.
The Red Queen hypothesis is a principle in evolutionary biology which proposes that organisms must constantly adapt and evolve not to gain an absolute improvement in fitness, but merely to maintain their relative fitness in the face of coevolving competing species, parasites, predators, and prey. Named after a character in Lewis Carroll's Through the Looking-Glass, the hypothesis captures the idea that in a coevolutionary system, standing still is equivalent to falling behind.1, 2 First formally articulated by the palaeontologist Leigh Van Valen in 1973, the Red Queen hypothesis has become one of the most influential concepts in modern evolutionary theory, providing a framework for understanding the dynamics of [coevolution](/evolution/coevolution), the maintenance of [sexual reproduction](/evolution/evolution-of-sexual-reproduction), and the persistence of genetic diversity within populations.1
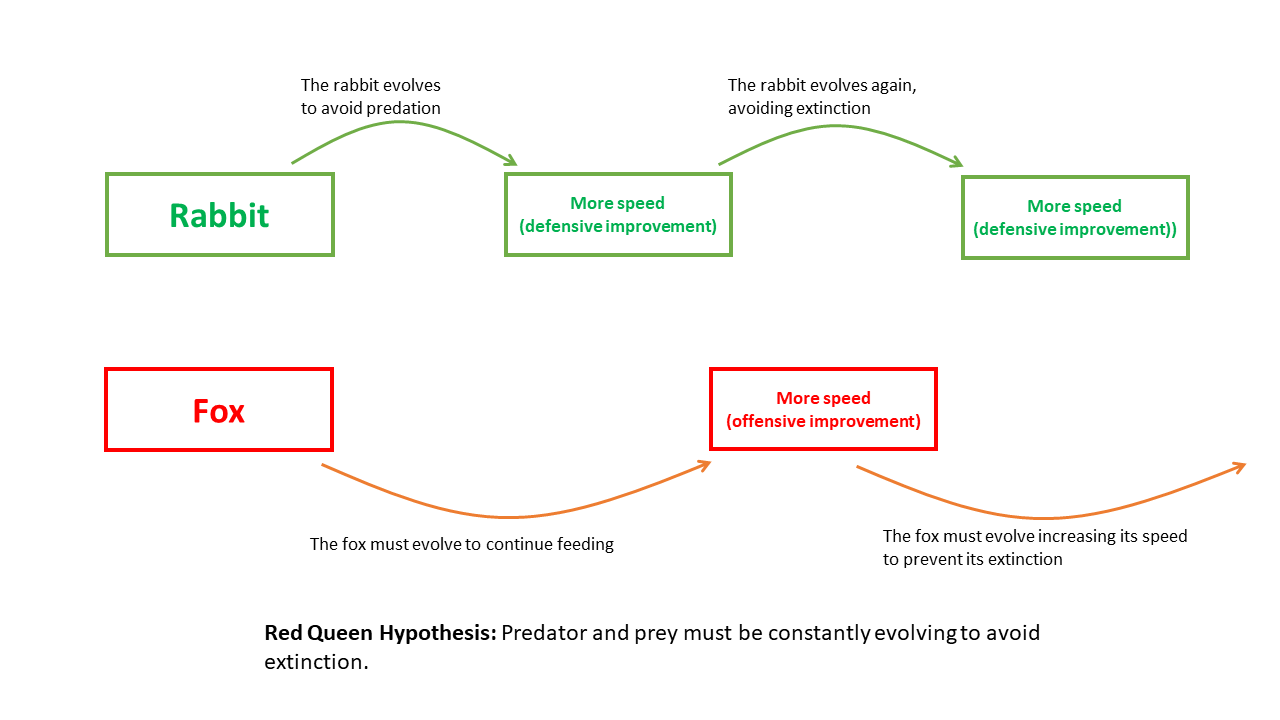
The hypothesis addresses a fundamental puzzle: if [natural selection](/evolution/natural-selection) continually improves organisms' adaptations, why do extinction rates appear roughly constant over geological time? Van Valen's answer was that the biotic environment—the community of interacting species—is itself evolving, so that any adaptive gain by one species degrades the relative fitness of others. The result is a system in perpetual motion, where evolutionary stasis is impossible and every lineage must run just to keep in place.1 This insight has had far-reaching implications, providing some of the most compelling theoretical and empirical arguments for why sex exists, why immune genes are so diverse, and why [evolutionary arms races](/evolution/evolutionary-arms-races) can persist for millions of years.
Origins and formulation
The Red Queen hypothesis emerged from Van Valen's analysis of extinction data across a wide range of taxa. In his 1973 paper "A new evolutionary law," Van Valen demonstrated that within a given taxonomic group, the probability of a lineage going extinct appeared to be roughly constant over time—a pattern he termed the "Law of Constant Extinction."1 This finding was surprising because it contradicted the intuitive expectation that species should become better adapted over time and therefore more resistant to extinction. If organisms were progressively improving through [natural selection](/evolution/natural-selection), older lineages should have lower extinction rates than younger ones. Instead, the data suggested that no lineage, however long-lived, had achieved any cumulative advantage over the forces that caused extinction.
Van Valen explained this pattern by arguing that the effective environment of any species is dominated by other evolving species. As one species evolves an improvement—a better defence against a predator, a more efficient means of extracting resources—the species with which it interacts suffer a corresponding decline in relative fitness. Those species must then evolve compensating adaptations simply to restore their previous position, which in turn degrades the fitness of the first species, and so on in an endless cycle. The net effect is that no species ever achieves a permanent improvement in its position, because every adaptive advance is met by a counteradaptation elsewhere in the community.1
To illustrate this principle, Van Valen borrowed the metaphor of the Red Queen from Carroll's Through the Looking-Glass (1871), in which the Red Queen tells Alice: "Now, here, you see, it takes all the running you can do, to keep in the same place. If you want to get somewhere else, you must run at least twice as fast as that!"2 The analogy was apt: in a world where the biotic environment is constantly shifting because other organisms are evolving, each species must continue to evolve simply to avoid falling behind. The metaphor captured the imagination of evolutionary biologists and quickly became one of the most widely cited concepts in the discipline.
Van Valen's formulation was primarily macroevolutionary, concerned with patterns of extinction across geological time. However, subsequent researchers—most notably William Hamilton—recognized that the same logic applied powerfully at the population level, particularly to the interactions between hosts and parasites. This extension of the Red Queen from a macroevolutionary pattern to a microevolutionary mechanism transformed the hypothesis from an interesting observation about extinction rates into a central principle of evolutionary ecology.5, 6
Host-parasite coevolution
The most intensively studied application of the Red Queen hypothesis is in [host-parasite coevolution](/evolution/parasitism-and-coevolution). Parasites and pathogens exert some of the strongest selective pressures in nature, and their typically short generation times and large population sizes allow them to evolve rapidly in response to host defences. Hosts, in turn, are under intense selection to resist infection. The result is a coevolutionary dynamic in which host and parasite genotypes cycle through time: a common host genotype is targeted by parasites that evolve to exploit it, the host genotype declines in frequency, rarer genotypes enjoy a fitness advantage, and the cycle repeats.6, 9
This process is intimately connected to [frequency-dependent selection](/evolution/frequency-dependent-selection). When a particular host genotype is common, parasites adapted to that genotype have a large supply of susceptible hosts and increase in frequency. As the parasite becomes specialized on the common genotype, hosts carrying rare alleles for resistance enjoy a selective advantage—not because their defences are intrinsically superior, but simply because they are unfamiliar to the current parasite population. This negative frequency-dependent dynamic favours rare alleles and maintains genetic diversity within host populations, precisely as the Red Queen predicts.9, 16
The New Zealand freshwater snail Potamopyrgus antipodarum and its trematode parasite Microphallus sp. have become the premier model system for testing Red Queen predictions in the wild. These snails are unusual in that both sexual and asexual (clonal) individuals coexist within the same populations, providing a natural experiment for comparing the fates of sexual versus clonal lineages under parasite pressure. Lively and Dybdahl demonstrated that common clonal genotypes were disproportionately infected by parasites, while rare clonal genotypes and sexually produced offspring were less likely to be parasitized.9, 16 Over time, the frequency of particular clonal genotypes oscillated as parasites tracked common host types and drove them to low frequency, only for previously rare clones to take their place—precisely the cyclical dynamics predicted by the Red Queen.
Lively's earlier work had shown that sexual snails were more common in lake habitats where parasite infection rates were high, while asexual snails dominated in marginal habitats where parasite pressure was lower.11 This geographic pattern is consistent with the hypothesis that sexual reproduction is maintained where parasites are abundant enough to generate strong frequency-dependent selection, and that clonal reproduction succeeds where this pressure is relaxed.
The Red Queen and sexual reproduction
Perhaps the most consequential application of the Red Queen hypothesis has been to the longstanding problem of [the evolution and maintenance of sex](/evolution/evolution-of-sexual-reproduction). Sexual reproduction is costly: it requires finding mates, risks sexually transmitted diseases, breaks up favourable gene combinations, and—most fundamentally—introduces the "twofold cost of males," since asexual females transmit all of their genes to every offspring while sexual females transmit only half.3 Despite these costs, sexual reproduction is nearly ubiquitous among eukaryotes. Explaining its persistence is one of the central problems in evolutionary biology.
George C. Williams highlighted the paradox in his 1975 book Sex and Evolution, arguing that the prevalence of sex demanded an explanation in terms of short-term selective advantages powerful enough to overcome its immediate costs.3 Hamilton provided such an explanation through the Red Queen. In a series of influential papers beginning in 1980, he argued that parasites generate the frequency-dependent selection necessary to maintain sex. Because parasites evolve to exploit common host genotypes, sexual reproduction—which generates genetically variable offspring through recombination—is favoured because it produces rare genotype combinations that are less likely to be recognized and exploited by coevolving parasites.5, 6
Hamilton's argument can be summarized as follows: in a population where most individuals share a common genotype, a parasite that evolves the ability to infect that genotype has a large, vulnerable host population to exploit. Sexual reproduction shuffles alleles through meiotic recombination, producing offspring with novel genotype combinations that the current parasite population has not yet adapted to. Asexual lineages, by contrast, produce genetically identical offspring, making them predictable targets for rapidly evolving parasites. Over multiple generations, asexual lineages are driven to low frequency or extinction by parasites, while sexual lineages persist because they continually produce the genetic novelty needed to stay ahead of—or at least keep pace with—their coevolving antagonists.5, 6
Mathematical models have confirmed that the Red Queen mechanism can maintain sex under biologically realistic conditions, particularly when parasites have virulent effects on host fitness and when there is sufficient genetic variation in resistance and infectivity.6, 20 Salathé and colleagues showed that when Red Queen dynamics are coupled with directional selection, the conditions under which sex is favoured become substantially broader, suggesting that the Red Queen may work in concert with other mechanisms to maintain sexual reproduction across the diversity of conditions observed in nature.20
Empirical evidence
The Red Queen hypothesis has generated a rich body of empirical research spanning natural populations, palaeontological archives, and experimental evolution. The most direct evidence comes from systems in which the coevolutionary dynamics between hosts and parasites can be observed in real time or reconstructed from historical records.
The Potamopyrgus snail system, described above, provides the strongest field evidence. Dybdahl and Lively tracked clonal frequencies and infection rates over multiple years in New Zealand lakes, documenting the predicted time-lagged oscillations in which common clones became heavily parasitized and declined, only to be replaced by previously rare clones in a repeating cycle.9, 16 These oscillations in genotype frequency are the signature of Red Queen coevolution, and they have been documented across multiple populations and time periods.
A remarkable line of evidence comes from the Daphnia–parasite system studied by Decaestecker and colleagues. By hatching dormant Daphnia magna eggs preserved in dated sediment layers from a Belgian pond, and pairing them with parasite spores (Pasteuria ramosa) recovered from the same and different time periods, the researchers were able to reconstruct host–parasite coevolutionary dynamics spanning decades. They found that parasites were most infective to hosts from their own time period and less infective to hosts from the past or the future, demonstrating time-lagged negative frequency-dependent coevolution—the precise signature of Red Queen dynamics archived in natural sediments.10
Experimental evolution studies have provided the most controlled tests. Morran and colleagues used the nematode Caenorhabditis elegans and its bacterial pathogen Serratia marcescens to directly test whether coevolution with parasites favours sexual reproduction. Starting with C. elegans populations capable of both selfing and outcrossing (analogous to asexual and sexual reproduction), they exposed some populations to coevolving parasites, others to fixed (non-evolving) parasites, and still others to no parasites. Populations exposed to coevolving parasites evolved significantly higher rates of outcrossing, while those exposed to fixed parasites or no parasites maintained low outcrossing rates. Moreover, obligately selfing populations rapidly went extinct under coevolving parasite pressure, while obligately outcrossing populations persisted. This experiment provided the most direct experimental confirmation that host–parasite coevolution can drive the evolution of sex, exactly as the Red Queen predicts.8
Microbial systems have also yielded evidence for Red Queen dynamics. Buckling and Rainey studied coevolution between the bacterium Pseudomonas fluorescens and its bacteriophage in laboratory microcosms and observed ongoing reciprocal adaptation: bacteria evolved resistance to their contemporary phage, phage evolved to overcome that resistance, and this arms race produced directional change in both populations over many generations. When coevolution was allowed to proceed, both populations exhibited continual evolutionary change, consistent with the Red Queen prediction that neither party could afford to stop evolving.21
Hamilton–Zuk hypothesis and sexual selection
In 1982, Hamilton and Marlene Zuk extended Red Queen logic to the domain of [sexual selection](/evolution/sexual-selection), proposing that elaborate secondary sexual characters—bright plumage, complex songs, vigorous courtship displays—function as honest signals of heritable parasite resistance. Their reasoning was grounded in the Red Queen: if host–parasite coevolution maintains cycling genetic variation in resistance, and if parasites reduce the condition of infected individuals, then a male's ability to maintain bright plumage or vigorous displays despite parasite challenge signals that he carries currently favourable resistance alleles. Females choosing such males would gain indirect genetic benefits by producing offspring better equipped to resist the current parasite fauna.4
Hamilton and Zuk tested their prediction across North American passerine bird species and found a significant positive correlation between the prevalence of chronic blood parasites and the brightness of male plumage. Species with higher parasite loads had evolved more elaborate male ornamentation, consistent with the prediction that parasite pressure drives the evolution of conspicuous sexual signals.4 This finding linked the Red Queen directly to the theory of [sexual selection](/evolution/sexual-selection), suggesting that the elaborate secondary sexual characters that Darwin had struggled to explain might ultimately be maintained by host–parasite coevolution.
The Hamilton–Zuk hypothesis remains influential, though it has generated considerable debate. Some studies have replicated the interspecific correlation between parasite prevalence and ornament elaboration, while others have found weaker or nonsignificant relationships. At the intraspecific level, evidence that parasitized males are less ornamented and that females preferentially mate with less-parasitized males has been found in a variety of taxa, including birds, fish, and insects.7 The hypothesis has also been extended beyond plumage to other condition-dependent traits, including the MHC-based mate choice observed in mammals and fish, where individuals appear to prefer mates with complementary or dissimilar MHC genotypes, potentially increasing offspring resistance to a broader range of pathogens.19
Molecular evidence: MHC diversity and pathogen pressure
Some of the strongest molecular evidence for Red Queen dynamics comes from the major histocompatibility complex (MHC), a family of genes encoding cell-surface proteins that present pathogen-derived peptides to the immune system. MHC genes are among the most polymorphic in vertebrate genomes: in humans, some MHC loci (known as HLA genes) have hundreds of known alleles, far more than would be expected under neutral evolution or simple directional selection.12
The Red Queen provides a compelling explanation for this extraordinary diversity. Because different MHC alleles bind and present different pathogen peptides, the effectiveness of any given allele depends on the pathogen community to which a population is exposed. As pathogens evolve to evade recognition by common MHC alleles, rare alleles that can still detect the pathogen gain a fitness advantage—a classic example of negative [frequency-dependent selection](/evolution/frequency-dependent-selection) driven by host–parasite coevolution.12, 13 Over time, this process maintains a large number of alleles at intermediate frequencies, preventing any single allele from reaching fixation.
Multiple lines of evidence support the role of pathogen-mediated selection in maintaining MHC diversity. Population genetic analyses show signatures of balancing selection at MHC loci across diverse vertebrate species, with trans-species polymorphism—the retention of ancestral allelic lineages across speciation events—indicating that selection has maintained MHC variation for tens of millions of years.12 Comparative studies have demonstrated that MHC diversity is positively correlated with pathogen diversity across populations and species, as predicted by the Red Queen.12 And mate choice studies in both humans and other vertebrates have revealed preferences for MHC-dissimilar partners, suggesting that sexual selection may reinforce pathogen-driven balancing selection by favouring outbreeding at immune loci.19
The MHC system illustrates how Red Queen dynamics can operate at the molecular level, generating and maintaining the genetic diversity that is the raw material for continued adaptive evolution. The extraordinary polymorphism of these genes, maintained across deep evolutionary time, is among the most powerful pieces of evidence that biotic interactions—specifically, coevolution with pathogens—are a dominant force shaping the evolution of complex organisms.
Arms races and escalation
The Red Queen framework is closely related to the concept of [evolutionary arms races](/evolution/evolutionary-arms-races), in which two interacting lineages undergo reciprocal escalation of offensive and defensive traits. While the terms are sometimes used interchangeably, they describe subtly different dynamics. An arms race implies directional escalation—traits becoming progressively more extreme over time—whereas the Red Queen more broadly encompasses any situation in which reciprocal adaptation prevents either party from gaining a net advantage, including the cyclical genotype-frequency dynamics seen in host–parasite systems.1, 18
Dawkins popularized the arms race metaphor in The Selfish Gene (1976) and later works, arguing that predator–prey and host–parasite interactions generate selection for ever-more-elaborate adaptations on both sides.18 The Red Queen provides the underlying theoretical explanation for why these arms races persist: neither side can afford to stop evolving, because any pause in adaptation would be immediately exploited by the other party. The predator that stops improving its hunting technique is outpaced by prey that continue to refine their escape mechanisms, and vice versa.
At the molecular level, Red Queen arms races have been documented between primate immune genes and the retroviruses that infect them. Antiviral proteins such as TRIM5α and APOBEC3G show strong signatures of positive selection at the residues that interact directly with viral proteins, indicating ongoing adaptive evolution in response to viral counter-adaptations.15 These molecular arms races can be extraordinarily ancient: some host–pathogen conflicts at specific gene loci show evidence of reciprocal selection spanning millions of years, consistent with the Red Queen prediction of indefinite coevolutionary engagement.
Criticisms and alternative hypotheses
The Red Queen hypothesis, while widely influential, has not gone unchallenged. Several alternative hypotheses have been proposed to explain the phenomena it addresses, particularly the maintenance of sexual reproduction.
Muller's ratchet, formalized by Felsenstein in 1974, proposes that asexual populations accumulate deleterious mutations irreversibly over time because, in the absence of recombination, the class of individuals carrying the fewest mutations can only be lost (by [genetic drift](/evolution/genetic-drift)) and never reconstituted.14 Over many generations, this ratchet-like process degrades the fitness of asexual lineages, while sexual populations can purge deleterious mutations through recombination. Muller's ratchet provides a mutation-based rather than parasite-based explanation for the advantage of sex, and the two hypotheses are not mutually exclusive—both may contribute to the maintenance of recombination under different conditions.14, 17
A related class of explanations invokes the role of environmental change more broadly, including abiotic factors such as climate fluctuation, habitat disturbance, and resource availability. Under these models, sex is advantageous because it generates the genetic variation needed to adapt to unpredictable environmental shifts, not specifically to coevolving parasites. The "tangled bank" hypothesis, for example, proposes that sex is maintained because genetically diverse offspring are better able to exploit the wide range of ecological niches available in a complex environment, reducing sibling competition.3 While the Red Queen emphasizes biotic drivers of selection, the tangled bank and related hypotheses emphasize abiotic and ecological drivers.
Some critics have questioned whether the conditions required for Red Queen dynamics to maintain sex are sufficiently common in nature. The hypothesis requires that parasites exert strong fitness effects, that resistance has a significant genetic component, and that the genetic architecture of host–parasite interactions produces the frequency-dependent dynamics on which the mechanism depends. Mathematical models have shown that the Red Queen can fail to maintain sex when parasite virulence is low, when host–parasite genetic interactions are too simple or too complex, or when environmental stochasticity overwhelms the signal of frequency-dependent selection.17, 20
The pluralist view, now held by many evolutionary biologists, is that the maintenance of sex is likely explained by multiple reinforcing mechanisms rather than any single hypothesis. The Red Queen may interact with Muller's ratchet, with the Fisher–Muller effect (the advantage of bringing together beneficial mutations from different backgrounds), and with ecological variability to produce a combined advantage of sex that exceeds what any single mechanism could provide alone.17 The Red Queen remains arguably the most empirically supported of these mechanisms, particularly for systems with strong host–parasite interactions, but the full explanation for the ubiquity of sex almost certainly involves a combination of forces.
Broader implications and legacy
The Red Queen hypothesis has influenced evolutionary biology well beyond its original domains of extinction rates and sexual reproduction. Its core insight—that the biotic environment is not a static backdrop but a constantly shifting challenge driven by the evolution of other organisms—has reshaped how biologists think about [natural selection](/evolution/natural-selection), adaptation, and diversity.
In conservation biology, Red Queen thinking has highlighted the importance of maintaining genetic diversity in threatened populations. Small, genetically depauperate populations may lack the variation needed to respond to coevolving pathogens, making them vulnerable to disease outbreaks that could drive them to extinction.15 The loss of genetic diversity through bottlenecks or inbreeding may be especially dangerous precisely because it undermines the population's ability to participate in the Red Queen dynamic that keeps pace with pathogen evolution.
In epidemiology and medicine, Red Queen dynamics inform our understanding of antibiotic resistance, vaccine escape, and the evolutionary management of infectious diseases. The rapid evolution of drug-resistant pathogens is a direct manifestation of Red Queen coevolution: each new drug represents an adaptation by the "host" (in this case, human medicine), and the pathogen's evolution of resistance is the coevolutionary countermove. Recognizing this dynamic has led to strategies such as antibiotic cycling and combination therapy, which attempt to exploit the frequency-dependent nature of resistance evolution.15
More broadly, the Red Queen has become a conceptual framework for understanding why evolution never reaches a stable endpoint. In a world populated by coevolving organisms, there is no final, optimal solution to the problem of survival and reproduction. Every adaptation is provisional, effective only against the current biological landscape, and subject to obsolescence as that landscape shifts. This view has profound implications for understanding the tempo and mode of evolution: it suggests that much of evolutionary change is driven not by adaptation to a fixed physical environment but by the endless reciprocal adjustments of species locked in coevolutionary relationships.1, 15
Van Valen's original insight, now more than fifty years old, continues to generate new research and new applications. The Red Queen hypothesis stands as one of the most productive ideas in evolutionary biology—a simple metaphor drawn from a children's book that captured a deep truth about the nature of life: that in a world of coevolving organisms, the only constant is change, and the price of survival is perpetual adaptation.
References
Parasite-mediated selection against inbred Soay sheep in a free-living, island population
Male fitness increases when females are eliminated from gene pool: implications for the evolution of sex
The Red Queen and her court: rethinking the role of biotic interactions for species diversification