Overview
- New species arise through reproductive isolation—when populations can no longer interbreed and exchange genes, genetic divergence accumulates until distinct species are formed. Geographic separation (allopatric speciation) is the most common route, but speciation can also occur within a continuous population when ecological or genetic barriers prevent gene flow.
- The genetics of speciation is well understood: incompatible gene combinations (Dobzhansky–Muller incompatibilities), chromosomal rearrangements, and differences in gene expression accumulate between isolated populations and eventually prevent them from producing fertile hybrids. Polyploidy—whole-genome duplication—can produce new plant species instantly, within a single generation.
- Speciation has been directly observed in the laboratory and in nature: apple maggot flies began diverging onto hawthorn hosts within 150 years, cichlid fishes diversified into hundreds of species in African rift lakes over tens of thousands of years, and allopolyploid plants such as Spartina anglica have been watched emerging as reproductively isolated species in real time.
Speciation—the evolutionary process by which one species divides into two or more reproductively distinct lineages—is the mechanism that generates the diversity of life on Earth. Every species alive today is the product of countless splitting events stretching back to the origin of life, and speciation continues to occur in populations under observation right now. Understanding how new species arise requires examining both the ecological and geographical conditions that trigger divergence and the genetic mechanisms that render that divergence permanent. The study of speciation has been one of the most productive areas of evolutionary biology since Ernst Mayr formalized the biological species concept in 1942, and advances in genomics have transformed what was once largely a theoretical field into one with a rich experimental literature.1, 3
The species concept
Before asking how species form, one must ask what a species is. The answer is less straightforward than it might appear, and biologists have proposed more than two dozen formal species concepts, each emphasizing different biological properties. The most influential of these is the biological species concept (BSC), formulated by the ornithologist and evolutionary biologist Ernst Mayr. In his 1942 monograph Systematics and the Origin of Species, Mayr defined species as "groups of actually or potentially interbreeding natural populations that are reproductively isolated from other such groups."1 The BSC focuses on reproductive isolation as the defining criterion: what holds a species together as a coherent unit is the exchange of genes through interbreeding, and what separates two species is the absence of that exchange. Mayr elaborated and defended this concept extensively in Animal Species and Evolution (1963), which remains a foundational text in the field.2
The BSC has been enormously productive, but it has recognized limitations. It cannot be applied to asexual organisms, which reproduce without interbreeding, and it is impractical for fossil taxa, which cannot be observed mating. It also struggles with the many cases of hybridization between organisms considered to be separate species, including numerous well-documented plant hybrids and hybrid zones in animals.3 The phylogenetic species concept (PSC), proposed by Joel Cracraft in 1983, addresses some of these difficulties by defining a species as the smallest diagnosable cluster of organisms sharing a unique combination of derived character states—that is, the smallest monophyletic group distinguishable by molecular or morphological data.6 The PSC tends to recognize more species than the BSC because it applies to any reproductively distinct population without requiring evidence of reproductive isolation.
The ecological species concept, championed by Dolph Schluter and others, defines species in terms of adaptive zones: a species is a lineage occupying a distinct niche and maintained by natural selection against gene flow from other lineages.7 Alan Templeton's cohesion species concept combines elements of the BSC and the ecological concept, requiring both genetic exchangeability (the potential for interbreeding) and ecological interchangeability (shared adaptive properties).4 Despite these debates, in practice the different concepts agree in the vast majority of cases, and the disagreements are most pronounced at the margins, where populations are in the early stages of divergence and the boundary between "population" and "species" is genuinely fuzzy. This fuzziness is itself biologically informative: it reflects the fact that speciation is a continuous process, not a discrete event.3
Modes of speciation
Biologists classify speciation primarily by the geographical relationship between diverging populations. Allopatric speciation, in which a physical barrier divides a population into geographically separated subpopulations, is the most widely documented and theoretically well-supported mode.1, 3 When a mountain range rises, a river changes course, a land bridge floods, or a small group of colonizers crosses a barrier to found a new population, the subpopulations that result are cut off from gene exchange. In isolation, each population accumulates genetic changes through mutation, drift, and the action of local natural selection. If the barrier persists long enough, these changes pile up to the point where the two populations can no longer interbreed even if the barrier is removed. The divergence of the North American red squirrel (Tamiasciurus hudsonicus) and the Kaibab squirrel (S. kaibabensis), isolated on opposite rims of the Grand Canyon, is one of many textbook examples of allopatric divergence in progress.3
Peripatric speciation is a variant of allopatric speciation in which the isolated subpopulation is very small—a handful of colonists founding a population on an island, for example. Small founder populations experience intense genetic drift, which can rapidly fix novel allele combinations that would remain rare in a large population. Ernst Mayr called this the "founder effect" and proposed it as an especially rapid pathway to speciation.2 The extraordinary diversity of Hawaiian Drosophila—more than 800 species on an archipelago that has existed for fewer than six million years—is often attributed in part to peripatric speciation events as flies colonized new islands in the Hawaiian chain.3
Parapatric speciation occurs when populations diverge across a geographical gradient without complete physical separation, as when a species ranges across a sharp environmental boundary such as a transition from grassland to forest or from polluted to unpolluted soil. Selection along the gradient can favor different genotypes in different zones, and if the selection pressure is strong enough to overcome the homogenizing effect of gene flow between zones, genetic divergence can accumulate until reproductive isolation follows. The heavy-metal-tolerant ecotypes of bent-grass (Agrostis tenuis) that evolved in the vicinity of copper-mine tailings in Wales, which show partial reproductive isolation from adjacent non-tolerant populations, are a frequently cited example of parapatric divergence.3
Sympatric speciation—the formation of new species within a single, continuously distributed, freely interbreeding population, without any geographical barrier to gene flow—was long regarded with skepticism. The concern was theoretical: if a population mates randomly, any genetic variant associated with incipient reproductive isolation would be diluted by recombination before it could spread. However, both theoretical models and empirical evidence have established that sympatric speciation can occur when disruptive selection is strong, when a mating preference is genetically correlated with the trait under selection, and when gene flow between ecological forms is sufficiently reduced by assortative mating.9 The cichlid radiations of crater lakes in Africa and the host-race formation in apple maggot flies (discussed in later sections) represent the best-supported contemporary evidence for this mode.9, 21
Reproductive isolation mechanisms
Reproductive isolation—any barrier that prevents or reduces gene flow between two populations—is the essence of speciation under the biological species concept.
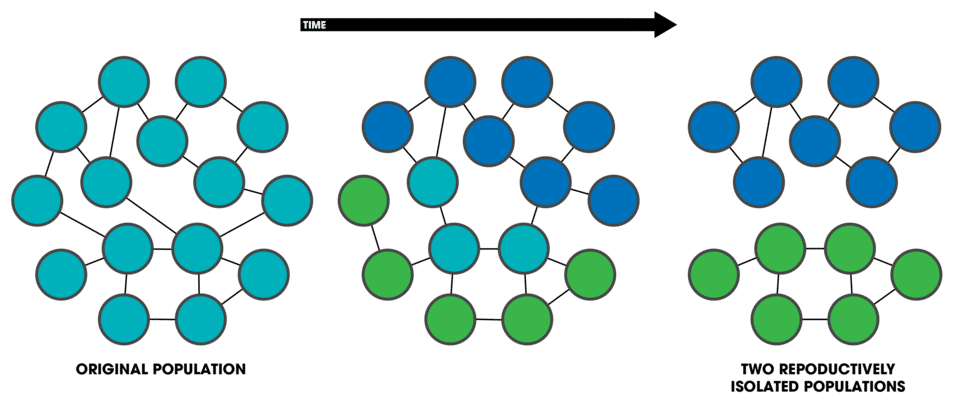
Biologists divide these barriers into two broad classes based on when they act relative to fertilization. Prezygotic barriers prevent the formation of hybrid zygotes, while postzygotic barriers reduce the viability or fertility of hybrids that do form. In most well-studied species pairs, multiple prezygotic and postzygotic barriers operate simultaneously, and their combined effect is nearly complete reproductive isolation.3
Prezygotic barriers include temporal isolation (breeding at different times of year or day), habitat isolation (occupying different microenvironments within the same area), behavioral isolation (mate choice differences that prevent cross-species pairings), mechanical isolation (incompatible genital morphology or floral structures), and gametic isolation (biochemical incompatibilities between sperm and egg that prevent fertilization even after mating). Behavioral isolation is particularly strong in animals with elaborate courtship rituals or species-specific mating calls: female frogs, for instance, distinguish conspecific males by call frequency with high precision, rarely responding to the calls of closely related species.3
Postzygotic barriers include hybrid inviability (hybrids that die before reaching reproductive age), hybrid sterility (hybrids that survive but are infertile), and hybrid breakdown (first-generation hybrids are fertile, but their offspring show reduced fitness). The mule—the sterile offspring of a horse and a donkey—is a familiar example of hybrid sterility. In plants, crosses between species frequently yield seeds with abnormal endosperms that fail to germinate, a form of hybrid inviability rooted in dosage imbalances in genomic imprinting systems.3
A generalization known as Haldane's rule describes a recurrent pattern in postzygotic isolation: when only one sex of hybrid offspring is absent, sterile, or inviable, it is almost always the heterogametic sex—that is, the sex carrying two different sex chromosomes (XY in mammals and many insects, ZW in birds and Lepidoptera).12 J. B. S. Haldane described this pattern in 1922, and it holds across an extraordinarily diverse range of animal taxa. Jerry Coyne's review of Haldane's rule, published in 1992, documented it in over 100 species pairs spanning insects, amphibians, birds, and mammals, and proposed that it results from the exposure of X-linked (or Z-linked) recessive incompatibilities in the hemizygous sex.12
The genetics of speciation
The molecular basis of reproductive isolation has been one of the most intensively studied questions in evolutionary genetics over the past three decades. The dominant theoretical framework is the Dobzhansky–Muller (DM) model of incompatibilities, named for Theodosius Dobzhansky and Hermann Muller, who independently proposed it in the 1930s and 1940s. The model explains how postzygotic isolation can evolve without requiring either population to pass through a fitness valley. Consider two gene loci, A and B, that interact. In the ancestral population, the combination A1B1 works normally. When one population is isolated, its A locus evolves to a new state, A2, while the B locus stays as B1; the combination A2B1 functions fine in the derived population because natural selection tested it. In the second isolated population, the B locus evolves to B2 while A stays as A1; the combination A1B2 also works fine. But when the two populations meet, hybrids carrying A2B2 may be inviable or infertile because this combination was never tested by selection in either lineage and the proteins or regulatory elements involved may be incompatible.11
The first DM incompatibility to be characterized at the molecular level was the Lhr–Hmr system in Drosophila melanogaster–D. simulans hybrids, where specific mutations in heterochromatin-binding proteins on different chromosomes cause lethality when combined in hybrid genomes. Subsequent research identified numerous additional DM incompatibilities in Drosophila and in other organisms.11 A meta-analysis by Presgraves (2009) found that DM incompatibilities accumulate faster than linearly with divergence time—a phenomenon called the "snowball effect"—consistent with the prediction that each new substitution in one lineage can potentially create a new incompatibility with any previously evolved substitution in the other lineage.11
Chromosomal rearrangements—inversions, translocations, and fusions—also contribute to reproductive isolation. When chromosomal segments are inverted, heterozygotes for the inversion produce unbalanced gametes and thus have reduced fertility. This can reduce gene flow between populations carrying different arrangements even in secondary contact, allowing other genetic differences to accumulate in the inverted regions without being erased by recombination.3 The role of chromosomal rearrangements in speciation has been documented in house mice (Mus musculus), in sunflowers (Helianthus), and in numerous other taxa.3 Studies of Heliconius butterflies have further revealed that "genomic islands of divergence"—regions of elevated genetic differentiation between incipient species—cluster around chromosomal inversions, which suppress recombination and thus allow divergence to persist despite ongoing gene flow.10
Polyploidy and instantaneous speciation
In most animal groups, speciation is a gradual process playing out over thousands to millions of generations. Plants, however, have a speciation mechanism available to them that can produce complete reproductive isolation within a single generation: polyploidy, or the multiplication of entire chromosome sets. When a polyploid individual arises in a population, it cannot easily interbreed with the diploid parent population because crosses produce aneuploid offspring with odd numbers of chromosomes, which are typically sterile. If two polyploid individuals can find each other and mate, or if a single polyploid self-fertilizes, a new reproductively isolated lineage can be established almost instantly.13
Autopolyploidy involves the doubling of a single species' own genome, producing a tetraploid from a diploid. Allopolyploidy results from hybridization between two different species followed by chromosome doubling, producing an organism with two complete diploid genomes—one from each parent species. Allopolyploidy is particularly common and has been a major force in the evolution of flowering plants: estimates suggest that between 70 and 80 percent of all flowering plant species have experienced one or more polyploidization events in their evolutionary history, and many economically important crops—wheat, cotton, tobacco, and oilseed rape—are allopolyploids.13, 14
The clearest natural experiment in allopolyploid speciation has been observed in cord grass (Spartina). The species Spartina anglica arose in the estuaries of southern England in the late nineteenth century through hybridization between the native S. maritima (2n = 60) and the introduced North American S. alterniflora (2n = 62), followed by chromosome doubling in the sterile F1 hybrid to produce a fertile allopolyploid (2n = 122 or 124). Spartina anglica was first documented around 1892 and is reproductively isolated from both parent species. Because its origin was witnessed in historical time and can be reconstructed in detail, it constitutes one of the few cases where the entire process of speciation has been directly observed.15
An equally remarkable case of repeated allopolyploid speciation was documented in goatsbeards (Tragopogon) in North America. Three diploid species—T. dubius, T. pratensis, and T. porrifolius—were introduced from Europe in the early twentieth century. Within roughly 80 to 100 years, two new tetraploid species, T. mirus and T. miscellus, arose through hybridization between different parental combinations followed by genome doubling. Remarkably, these allopolyploid species formed not once but repeatedly and independently: at least 21 separate origins of T. mirus and 11 of T. miscellus have been documented from different localities. First described by Marion Ownbey in 1950, the Tragopogon system demonstrates that allopolyploid speciation is not a one-off accident but a repeatable, predictable process that occurs whenever the ecological and genetic conditions are met.25, 13
Examples of allopolyploid crop species and their parent species13, 14
| Allopolyploid species | Common name | Parent species | Ploidy level |
|---|---|---|---|
| Triticum aestivum | Bread wheat | T. urartu × Aegilops speltoides × Ae. tauschii | Hexaploid (6x) |
| Gossypium hirsutum | Upland cotton | G. arboreum × G. raimondii | Tetraploid (4x) |
| Nicotiana tabacum | Common tobacco | N. sylvestris × N. tomentosiformis | Tetraploid (4x) |
| Brassica napus | Oilseed rape / canola | B. oleracea × B. rapa | Tetraploid (4x) |
| Spartina anglica | Common cord grass | S. maritima × S. alterniflora | Dodecaploid (12x) |
Ring species and hybrid zones
Ring species occupy a special conceptual role in speciation biology because they make visible, in geographical space, what normally unfolds across geological time. A ring species consists of a chain of intergrading populations that wraps around a geographical barrier. At the two ends of the chain, populations have diverged sufficiently that they cannot interbreed when they meet, even though each adjacent link in the chain can interbreed with its neighbors. The ring species thus presents, simultaneously and geographically arranged, a complete continuum from interbreeding populations to reproductively isolated "species"—demonstrating that the boundary between intraspecific and interspecific divergence is not a sharp discontinuity but a gradient.16
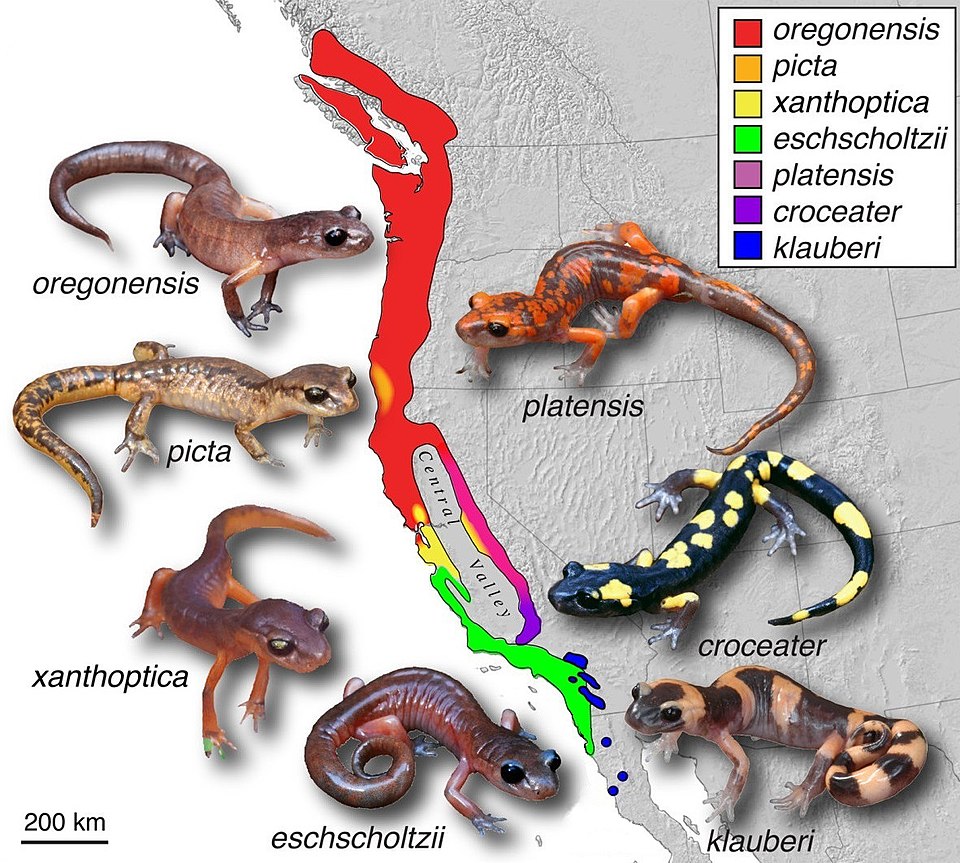
The Ensatina salamander complex in California is frequently cited as a ring species example. Populations of Ensatina eschscholtzii form a ring around the Central Valley, with populations along the Sierra Nevada to the east and the Coast Ranges to the west that meet in southern California. Where the two end-populations come into contact, they hybridize rarely if at all and are treated by some taxonomists as distinct species, while all adjacent populations along the ring hybridize freely.16 More recently, the greenish warbler (Phylloscopus trochiloides) has been studied as a ring species expanding around the Tibetan Plateau. Darren Irwin and colleagues documented that adjacent populations within the ring interbreed freely, but the two terminal populations that meet in Siberia are behaviorally isolated—their songs have diverged so substantially that males do not respond to the songs of the terminal-population neighbors, and hybrids are rare in the contact zone.17
Hybrid zones—regions where two partially isolated populations meet and exchange genes—are also highly informative about the speciation process. Because hybrid zones function as natural experiments in which reproductive isolation is tested against gene flow, they reveal which genetic differences between populations are most important in maintaining separation. Studies of hybrid zones in European crows (Corvus corone and C. cornix), house mice, Drosophila, and numerous plant taxa have shown that hybrid zones are often narrow relative to the dispersal distance of the organisms involved, indicating that selection against hybrids is strong enough to maintain sharp boundaries despite ongoing dispersal across them.3
Observed and documented speciation events
Speciation is sometimes portrayed as a process too slow to observe directly.

In reality, a growing body of evidence documents speciation occurring within historical time or within the span of a scientific study. The apple maggot fly (Rhagoletis pomonella) provides one of the most intensively studied examples of incipient sympatric speciation in an animal. R. pomonella is native to North America, where it historically fed and mated on hawthorn fruits (Crataegus spp.). Domestic apples (Malus domestica) were introduced to North America from Europe in the seventeenth century, and by the mid-nineteenth century, R. pomonella had begun infesting apple orchards. Research by Guy Bush, Jeffrey Feder, and colleagues demonstrated that hawthorn-reared and apple-reared flies differ significantly in their host preferences for mating and oviposition, in the timing of their adult emergence (which correlates with the fruiting phenology of their respective hosts), and in the frequencies of alleles at several enzyme loci that show evidence of host-associated selection.8, 22 The two host races have not achieved complete reproductive isolation and are therefore not yet separate species under the BSC, but the degree of assortative mating and ecological differentiation observed in fewer than 200 years represents an early stage of the speciation process unfolding in observable time.22
African cichlid fishes represent perhaps the most spectacular natural experiment in rapid speciation known to science. The cichlids of the East African Great Lakes—Victoria, Tanganyika, and Malawi—have undergone explosive adaptive radiations, producing hundreds of morphologically, ecologically, and behaviorally distinct species in geologically brief intervals. Lake Victoria, which may have dried completely as recently as 12,000–15,000 years ago, is now home to an estimated 500 species of hcichlid, all descended from a small ancestral population that re-colonized the lake after refilling.9, 21 Lake Malawi harbors roughly 600 species, and Lake Tanganyika, the oldest of the three, contains approximately 250 species, many of which belong to lineages not found elsewhere.23
The cichlid radiations have been driven by a combination of ecological opportunity (the vast, previously unoccupied resource space of the lakes), morphological versatility rooted in the cichlid jaw anatomy (their decoupled oral and pharyngeal jaw systems permit fine-scale dietary specialization), and strong sexual selection mediated by male coloration and female color preference. Ole Seehausen and colleagues demonstrated in 1997 that the turbidity of Lake Victoria water, which has increased dramatically due to agricultural runoff, is reducing the effectiveness of female color discrimination—a prezygotic barrier—and thereby causing hybridization between formerly isolated species, providing a direct observation of reproductive isolation breaking down.9
The Heliconius butterflies of Central and South America illustrate both sympatric ecological speciation and the role of mimicry as a reproductive isolating mechanism. Different species and races of Heliconius display striking warning coloration patterns that signal their unpalatability to predators, and closely related species frequently share or exchange pattern elements through hybridization and introgression. Studies by Jiggins and colleagues showed that color pattern differences serve as mate recognition cues: female Heliconius preferentially mate with males sharing their own wing pattern, creating assortative mating that is both ecologically maintained (predator selection keeps convergent mimicry patterns) and reproductively isolating (females prefer conspecific patterns).24 Genomic analyses of Heliconius populations have revealed that divergence is highly heterogeneous across the genome, with islands of high differentiation concentrated in regions controlling color pattern even while gene flow continues in the rest of the genome—a pattern consistent with speciation with ongoing gene flow driven by strong disruptive selection at specific loci.10
Reinforcement and the completion of speciation
When two partially isolated populations come back into contact in a secondary contact zone, the evolutionary outcome depends on the degree of reproductive isolation already accumulated. If reproductive isolation is nearly complete, the populations will persist as distinct species. If it is weak, gene flow may erode the genetic differences between them, and the two incipient species will merge back into one. A third possibility, reinforcement, occurs when natural selection in the contact zone strengthens prezygotic isolation: if hybrids have reduced fitness relative to parental types (due to postzygotic incompatibilities), then individuals that mate preferentially with their own type produce more surviving offspring and thus leave more descendants. Over generations, this selection pressure causes mating preferences to become more discriminating precisely in the contact zone, reinforcing the reproductive barrier and pushing the two populations further toward full species status.18
Reinforcement was long controversial because early theoretical models showed it to be easily swamped by recombination, which breaks up the association between preference genes and performance genes. More recent models and empirical studies, reviewed by Servedio and Noor in 2003, have rehabilitated reinforcement as a genuine evolutionary force, particularly when hybridization is costly and when mating preferences and viability are genetically linked in the relevant populations.18 Drosophila populations provide the strongest empirical support: species pairs that come into secondary contact show greater behavioral isolation in sympatry than allopatric populations of the same species pairs, the pattern predicted by reinforcement but not by simple allopatric divergence alone.18
The tempo of speciation
How fast does speciation occur? The question is empirically tractable through two independent approaches: calibrating the divergence between known species against geological or molecular clocks, and examining the fossil record for evidence of rapid versus gradual change. The answers reveal that speciation rates vary enormously across taxa and circumstances, spanning at least six orders of magnitude from the nearly instantaneous speciation of allopolyploids to the slow, million-year divergence of morphologically conservative lineages.3
The prevailing view through much of the twentieth century, associated with the Modern Synthesis, was that speciation is a gradual process of slow, continuous anagenetic change. Charles Darwin, in On the Origin of Species, emphasized the imperceptible gradation of change and was skeptical of saltational models. However, in 1972, Niles Eldredge and Stephen Jay Gould proposed a different pattern based on the fossil record: punctuated equilibrium. Examining the stratigraphic record of numerous lineages, Eldredge and Gould argued that most species show long intervals of morphological stasis punctuated by geologically rapid episodes of change associated with speciation events, rather than the slow, directional anagenesis predicted by phyletic gradualism.19
The punctuated equilibrium hypothesis generated intense debate, some of it acrimonious, but has since been partially vindicated by quantitative analyses. Studies by Erwin and Anstey and others have confirmed that morphological change in many fossil lineages is indeed concentrated near cladogenetic events (speciation) rather than distributed evenly through anagenesis.20 This pattern is broadly consistent with the idea that allopatric speciation in peripheral isolates, where populations are small and both drift and selection are strong, produces rapid morphological change, while large central populations are held near an adaptive optimum by stabilizing selection for most of their history. The debate between gradualism and punctuated equilibrium is thus not about whether evolution occurs, but about its tempo and its relationship to the speciation process.19, 20
Molecular clock analyses have provided independent estimates of speciation rates. The cichlid radiations of the African Great Lakes, for instance, represent among the fastest known vertebrate radiations: roughly 500 species in Lake Victoria in perhaps 15,000 years corresponds to a speciation rate orders of magnitude faster than average for vertebrates.21, 23 By contrast, the living coelacanth (Latimeria chalumnae) and its relatives show among the slowest morphological evolution known, with forms essentially unchanged over hundreds of millions of years. These extremes reflect not a contradiction in evolutionary theory but the dependence of speciation rate on ecological opportunity, population structure, generation time, and the intensity of disruptive selection.3
Speciation as ongoing process
Speciation is not a historical curiosity confined to the remote past. It is an active, ongoing process documented in populations studied by living scientists. From the host-race formation of apple maggot flies in North American orchards, to the explosive diversification of cichlids in African rift lakes, to the witnessed birth of Spartina anglica in British estuaries, and to the ring-species continua of salamanders and warblers that make visible the spectrum from population to species, the evidence is consistent and cumulative: new species arise through the gradual or rapid accumulation of reproductive isolation driven by geography, ecology, genetics, and the chance events of mutation and drift.3, 7, 15
The genetic mechanisms underlying this process are increasingly understood at the molecular level. Dobzhansky–Muller incompatibilities accumulate as a predictable by-product of independent evolution in isolated populations, and their rate of accumulation increases nonlinearly with divergence time.11 Chromosomal rearrangements create local barriers to gene flow that allow divergence to persist in genomic islands even when flanking regions continue to exchange alleles.10 Polyploidy provides an instantaneous route to reproductive isolation in plants, contributing to the extraordinary diversity of flowering plant lineages.13 Taken together, these mechanisms show that the origin of species is not a mysterious or improbable event but a natural consequence of the processes of mutation, recombination, selection, and drift operating within and between populations across time.
References
Spartina anglica C. E. Hubb., a natural model system for analysing early evolutionary changes that affect allopolyploid genomes
Testing the punctuated equilibrium model by comparing schedules of anagenesis and cladogenesis
Ecological speciation and the roles of sexual selection and secondary contact in the radiation of Heliconius butterflies