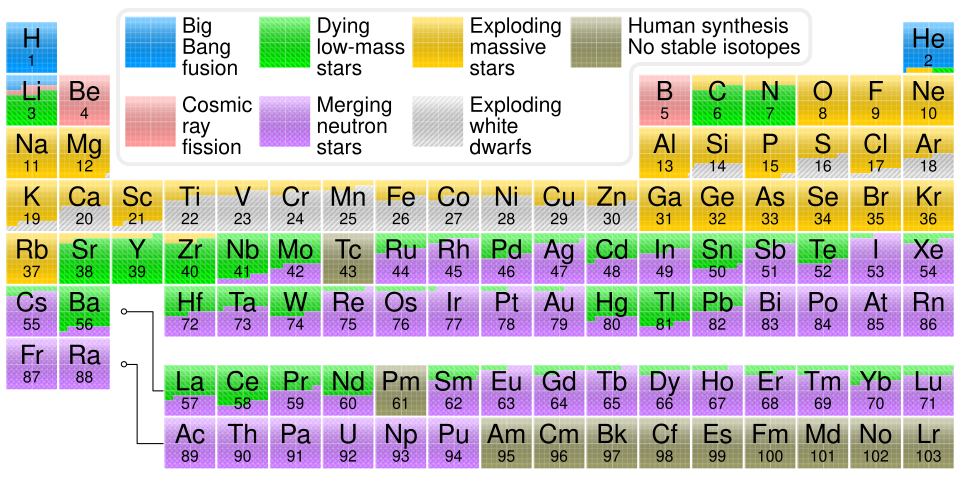
Overview
- All naturally occurring elements were forged by nuclear reactions in three broad settings: the hot, dense universe during the first twenty minutes after the Big Bang (producing hydrogen, helium, and trace lithium), the interiors of successive generations of stars (building elements up to iron through fusion), and explosive or neutron-rich environments such as supernovae and neutron star mergers (synthesizing the heaviest elements through rapid neutron capture).
- The iron peak at atomic number 26 marks the transition from energy-releasing fusion to energy-consuming fusion, meaning elements heavier than iron cannot be produced by ordinary stellar burning and instead require neutron capture processes — the slow s-process in asymptotic giant branch stars and the rapid r-process in neutron star mergers and core-collapse supernovae.
- Cosmic chemical evolution has progressively enriched the universe from a nearly pure hydrogen-helium plasma to one containing the full periodic table, with each generation of stars manufacturing and dispersing heavier elements that become the raw material for subsequent stars, planets, and ultimately the carbon, nitrogen, oxygen, and iron essential for life.
Nucleosynthesis is the set of nuclear processes responsible for the creation of all naturally occurring elements and their isotopes. The hydrogen and most of the helium in the universe were produced in the first twenty minutes after the Big Bang, when the cosmos was a rapidly cooling plasma of protons, neutrons, and radiation.1, 2 Every element heavier than lithium, from the carbon in organic molecules to the uranium in Earth's mantle, was subsequently forged in the interiors of stars, in the violent explosions that end their lives, or in the collisions of compact stellar remnants.5 Understanding nucleosynthesis is therefore understanding the origin of the matter from which planets, oceans, atmospheres, and living organisms are composed.
The foundations of the field were laid in the mid-twentieth century. In 1948, Ralph Alpher, Hans Bethe, and George Gamow proposed that the light elements were synthesized in the early universe.1 In 1957, the landmark paper by Margaret Burbidge, Geoffrey Burbidge, William Fowler, and Fred Hoyle — known as B2FH — systematically demonstrated that stars are the sites where heavier elements are built, identifying eight distinct nuclear processes that together account for the observed cosmic abundances.5 These two pillars, cosmological and stellar nucleosynthesis, remain the framework within which all subsequent discoveries have been placed.
Big Bang nucleosynthesis
In the first fraction of a second after the Big Bang, the universe was far too hot for atomic nuclei to survive; any nucleus that formed was immediately destroyed by high-energy photons. As the universe expanded and cooled, a brief window of opportunity opened roughly one to twenty minutes after the initial singularity, during which temperatures and densities were sufficient for nuclear fusion but not so extreme as to photodisintegrate the products.2, 3 During this interval, virtually all of the free neutrons were incorporated into helium-4 nuclei, along with smaller amounts of deuterium, helium-3, and lithium-7.
The outcome of Big Bang nucleosynthesis (BBN) was determined primarily by the neutron-to-proton ratio at the time nuclear reactions began (approximately one neutron for every seven protons — a ratio established by the weak-interaction freeze-out at about one second after the Big Bang and then reduced further by neutron decay during the intervening minutes) and by the baryon-to-photon ratio, which controls the density of baryonic matter available for reactions.3, 4 The predicted primordial mass fractions are approximately 75 percent hydrogen and 25 percent helium-4, with deuterium at roughly 0.003 percent and lithium-7 at approximately 10−10 relative to hydrogen by number.4 These predictions agree remarkably well with observations of primordial abundances measured in metal-poor galaxies and quasar absorption systems, constituting one of the strongest quantitative confirmations of the hot Big Bang model.2, 17
The baryon density inferred from BBN abundance measurements is independently confirmed by observations of the cosmic microwave background by the Planck satellite, which yields a baryon density parameter of Ωbh2 = 0.0224 ± 0.0001, in excellent agreement with the BBN-derived value.17 A persistent discrepancy remains, however, for lithium-7: the predicted primordial abundance exceeds the value observed in the oldest, most metal-poor halo stars by a factor of roughly three, a tension known as the cosmological lithium problem that remains unresolved after four decades of investigation.3, 4
Hydrogen burning in stars
Once the first stars ignited several hundred million years after the Big Bang, a new era of nucleosynthesis began. The most fundamental stellar nuclear process is hydrogen burning, in which four hydrogen nuclei (protons) are fused into one helium-4 nucleus, releasing 26.7 MeV of energy per reaction and powering stars for the vast majority of their lifetimes.6, 20 Hydrogen burning proceeds through two principal pathways, each dominant in different stellar mass regimes.
In stars with masses up to approximately 1.3 solar masses, including the Sun, the proton-proton (pp) chain dominates. The chain begins with the fusion of two protons to form a deuterium nucleus, a positron, and a neutrino — a reaction mediated by the weak nuclear force and therefore extremely slow, setting the overall pace of solar energy generation. The deuterium subsequently captures another proton to form helium-3, and two helium-3 nuclei fuse to produce helium-4 and two protons. Alternative branches of the pp chain (pp II and pp III) proceed through beryllium-7 and boron-8 intermediates and become increasingly important at higher temperatures.6
In more massive stars, where core temperatures exceed approximately 17 million kelvin, the CNO cycle (carbon-nitrogen-oxygen cycle) becomes the dominant hydrogen-burning mechanism. First elucidated by Hans Bethe in 1939 and independently by Carl Friedrich von Weizsäcker, the CNO cycle uses pre-existing carbon-12 nuclei as catalysts: protons are successively added to carbon-12, producing nitrogen-13, carbon-13, nitrogen-14, oxygen-15, and nitrogen-15 before the cycle closes with the ejection of a helium-4 nucleus and the regeneration of carbon-12.20 The net result is identical to the pp chain — four protons become one helium-4 nucleus — but the CNO cycle's rate depends on the sixth power of temperature, making it overwhelmingly dominant in massive stars. The CNO cycle also converts most of the initial carbon and oxygen catalyst into nitrogen-14, the slowest step in the cycle, which is why nitrogen is relatively abundant in the universe.6, 9
Helium burning and the triple-alpha process
When a star exhausts the hydrogen fuel in its core, gravitational contraction raises the central temperature and density until helium can begin to fuse. The synthesis of carbon from helium presented a theoretical puzzle, because there is no stable nucleus with mass number 5 or 8, meaning that collisions between two helium-4 nuclei produce only beryllium-8, which decays in approximately 10−16 seconds. Fred Hoyle recognized in 1953 that carbon-12 could nevertheless be produced if a third helium-4 nucleus collided with the fleeting beryllium-8 before it decayed, but only if carbon-12 possessed a resonant excited state near 7.65 MeV that would greatly enhance the reaction rate.7 This state, subsequently confirmed experimentally, is known as the Hoyle state, and the overall reaction is called the triple-alpha process.
The triple-alpha process begins in earnest at core temperatures of approximately 100 million kelvin. In low- and intermediate-mass stars (roughly 0.8 to 8 solar masses), helium ignition occurs under degenerate conditions in a thermonuclear runaway called the helium flash, after which the star settles onto the horizontal branch burning helium steadily. In massive stars, helium burning proceeds non-degenerately in the core following hydrogen exhaustion.8 A competing reaction, the capture of an additional helium-4 nucleus by carbon-12 to form oxygen-16, occurs simultaneously. The ratio of carbon-12 to oxygen-16 produced during helium burning depends sensitively on the nuclear reaction rates involved and determines the subsequent chemical evolution of the star, making it one of the most consequential quantities in all of nuclear astrophysics.7, 9
Edwin Salpeter first described the basic physics of helium burning in stars in 1952, establishing the foundation upon which Hoyle's resonance prediction and the subsequent experimental confirmation were built.19
Advanced burning stages and the iron peak
In stars more massive than approximately 8 solar masses, the core temperatures and densities reached after helium exhaustion are sufficient to ignite a succession of increasingly brief nuclear burning stages, each consuming the ash of the previous stage as fuel. These advanced burning stages proceed in an onion-shell structure, with each successive fuel burning in a thinner, hotter shell closer to the centre of the star.8, 16
Carbon burning ignites at roughly 600 million kelvin, fusing two carbon-12 nuclei to produce sodium-23, neon-20, and magnesium-24 through various channels. Neon burning follows at approximately 1.2 billion kelvin, proceeding through the photodisintegration of neon-20 by high-energy gamma rays, which liberates helium-4 nuclei that are captured by remaining neon-20 to form magnesium-24. Oxygen burning commences at roughly 1.5 billion kelvin, fusing two oxygen-16 nuclei to yield silicon-28, phosphorus-31, and sulfur-32. Finally, silicon burning occurs at temperatures exceeding 2.7 billion kelvin, but rather than proceeding through direct silicon-silicon fusion, it operates through a complex network of photodisintegration and alpha-capture reactions in which silicon-28 is progressively converted into iron-group elements, predominantly nickel-56 (which subsequently decays to iron-56).8, 16
Each successive burning stage is dramatically shorter than the last. A 25-solar-mass star burns hydrogen for roughly 7 million years, helium for about 500,000 years, carbon for approximately 600 years, neon for about one year, oxygen for roughly six months, and silicon for only about one day before the core collapses.8 This acceleration reflects both the declining energy yield per reaction and the increasing energy losses to neutrino emission at high temperatures.
Duration of nuclear burning stages in a 25 M⊙ star8
The endpoint of this sequence is the iron peak, the group of elements centred on iron (atomic number 26), cobalt, and nickel that possess the highest nuclear binding energy per nucleon of any elements in the periodic table. Because iron-group nuclei are the most tightly bound, fusing them into heavier elements requires a net input of energy rather than releasing it. The star has reached a thermodynamic dead end: no further exothermic fusion is possible, and the inert iron core grows until it exceeds the Chandrasekhar mass (approximately 1.4 solar masses) and collapses catastrophically, triggering a core-collapse supernova.5, 16
Neutron capture processes
Because fusion cannot build elements beyond the iron peak through exothermic reactions, the synthesis of heavier elements requires a fundamentally different mechanism: the capture of free neutrons by existing seed nuclei. A nucleus that captures a neutron increases its mass number by one; if the resulting isotope is unstable, it undergoes beta decay, converting a neutron into a proton and thereby moving one step up the periodic table. The two principal neutron capture processes — the slow (s) process and the rapid (r) process — differ in the timescale of neutron capture relative to beta decay and operate in very different astrophysical environments.5, 11, 12
The s-process (slow neutron capture) occurs in asymptotic giant branch (AGB) stars, evolved low- and intermediate-mass stars (roughly 1 to 8 solar masses) in which thermal pulses in a helium-burning shell periodically inject fresh helium into a hydrogen-rich layer, generating free neutrons through reactions such as 13C(α, n)16O and 22Ne(α, n)25Mg.10, 11 The neutron densities in AGB interiors are relatively modest (approximately 106 to 1011 neutrons per cubic centimetre), so a nucleus typically waits years to centuries between successive neutron captures. This gives unstable isotopes ample time to beta-decay before capturing another neutron, causing the s-process path to follow the valley of beta stability closely. The s-process is responsible for roughly half of all isotopes heavier than iron and produces characteristic abundance peaks at nuclei with magic neutron numbers (N = 50, 82, and 126), where neutron capture cross sections are unusually small and nuclei accumulate — corresponding to strontium, barium, and lead.11
The r-process (rapid neutron capture) requires environments with extraordinarily high neutron densities (exceeding 1020 neutrons per cubic centimetre) and temperatures above one billion kelvin, conditions under which a seed nucleus can capture tens to hundreds of neutrons in seconds, far faster than beta decay can occur. The nucleus is driven deep into the neutron-rich side of the nuclear chart before the neutron flux subsides and the accumulated neutrons beta-decay back toward stability.12 The r-process produces roughly the other half of all isotopes above iron, including most of the heaviest naturally occurring elements such as gold, platinum, thorium, and uranium.
The astrophysical site of the r-process was debated for decades. Core-collapse supernovae were long considered the primary candidate, but detailed simulations struggled to reproduce the necessary neutron-rich conditions in the neutrino-driven wind above the proto-neutron star.12, 21 The question was substantially resolved in August 2017, when the LIGO and Virgo gravitational-wave detectors observed the binary neutron star merger GW170817, and electromagnetic follow-up observations across the spectrum detected a kilonova — a transient powered by the radioactive decay of freshly synthesized r-process elements.13, 14 Spectroscopic analysis of the kilonova identified strontium among the newly formed elements, providing the first direct identification of a specific r-process element in a neutron star merger ejecta.24 While neutron star mergers are now confirmed as a major r-process site, core-collapse supernovae and collapsars (the engines of long gamma-ray bursts) may also contribute, and the relative importance of these channels remains an active area of research.12, 21
Supernovae and explosive nucleosynthesis
Supernovae contribute to the chemical enrichment of the universe in two distinct ways: through the explosive processing of material during the detonation itself and through the dispersal of elements that were synthesized during the star's prior hydrostatic burning stages.
In a core-collapse supernova (Types II, Ib, and Ic), the shock wave that rebounds off the collapsed core heats the overlying shells to temperatures exceeding several billion kelvin In a core-collapse supernova (Types II, Ib, and Ic), the shock wave that rebounds off the collapsed core heats the overlying shells to temperatures exceeding several billion kelvin, driving explosive nucleosynthesis that modifies the composition of the silicon, oxygen, neon, and carbon shells. Explosive silicon burning produces iron-group elements (predominantly nickel-56), explosive oxygen burning yields silicon and sulfur, and explosive neon and carbon burning contribute additional magnesium, sodium, and aluminium.9, 16
Type Ia supernovae, which result from the thermonuclear detonation of a carbon-oxygen white dwarf that has accreted sufficient mass from a companion star or merged with another white dwarf, are the dominant producers of iron-peak elements in the universe. A single Type Ia event synthesizes approximately 0.6 solar masses of nickel-56, which decays through cobalt-56 to iron-56, contributing roughly two-thirds of all the iron in the cosmos.23 Type Ia supernovae also produce significant quantities of silicon, sulfur, calcium, and other intermediate-mass elements. Because Type Ia supernovae occur on a delayed timescale relative to core-collapse supernovae (their white dwarf progenitors require time to accrete mass), they enrich the interstellar medium with iron-peak elements on a characteristically longer timescale, producing a distinctive signature in the chemical evolution of galaxies.22, 23
Cosmic chemical evolution
The cumulative effect of billions of years of stellar nucleosynthesis has been the progressive chemical enrichment of the universe, a process called cosmic chemical evolution or galactic chemical evolution.
The first generation of stars (Population III) formed from the nearly pure hydrogen and helium left by Big Bang nucleosynthesis, containing no elements heavier than lithium. These stars synthesized the first carbon, oxygen, and iron and dispersed them into the surrounding gas through stellar winds and supernovae. Subsequent generations of stars formed from gas that was progressively enriched in heavy elements, or metals in the astronomical usage of the term (which encompasses all elements heavier than helium).15, 22
Astronomers quantify a star's heavy-element content by its metallicity, conventionally expressed as [Fe/H], the logarithmic ratio of iron to hydrogen relative to the solar value. The oldest stars in the Milky Way's halo have metallicities as low as [Fe/H] ≈ −5 (one hundred-thousandth of the solar iron abundance), while the youngest stars in the disk approach or exceed solar metallicity.15, 22 The systematic increase in metallicity from old to young stellar populations provides a direct record of the integrated nucleosynthetic output of all prior stellar generations.
Different elements are produced on different timescales and by different sources, creating diagnostic abundance ratios that encode the star formation history of a galaxy. The alpha elements (oxygen, magnesium, silicon, calcium, titanium) are produced predominantly by core-collapse supernovae, which occur promptly after a burst of star formation because their massive progenitors live only a few million years. Iron, by contrast, is produced in large part by Type Ia supernovae, which have a delay time of roughly 0.1 to several billion years.22, 23 As a result, old, metal-poor stars in the Milky Way show enhanced [α/Fe] ratios relative to the Sun, reflecting an era when core-collapse supernovae dominated the enrichment. At later times, the onset of Type Ia supernovae adds iron without proportionally increasing alpha elements, causing the [α/Fe] ratio to decline toward the solar value — a transition known as the alpha-element knee that is observed at [Fe/H] ≈ −1 in the Milky Way disk.9, 15
The origin of the elements essential for life
The elements most critical for biochemistry — carbon, nitrogen, oxygen, and iron — each trace their origins to specific nucleosynthetic processes and stellar populations. Carbon is produced primarily by the triple-alpha process in helium-burning cores and shells, with significant contributions from both intermediate-mass AGB stars (which dredge carbon to their surfaces and expel it through stellar winds) and massive stars (which release it in supernovae).7, 10 Nitrogen is synthesized largely through the CNO cycle, in which pre-existing carbon and oxygen are converted to nitrogen-14; both intermediate-mass AGB stars and massive rotating stars are major nitrogen producers.9, 15 Oxygen, the most abundant element by mass in Earth's crust, is produced almost exclusively by massive stars through helium burning (yielding oxygen-16 via the 12C(α, γ)16O reaction) and dispersed by core-collapse supernovae.9, 16
Iron is critical for life as the oxygen-carrying atom in haemoglobin and a cofactor in numerous enzymes. Approximately one-third of cosmic iron comes from core-collapse supernovae and two-thirds from Type Ia supernovae.23 The solar abundance of iron, approximately 0.14 percent by mass, reflects the cumulative output of roughly 4.6 billion years of galactic chemical evolution before the formation of the Sun.18
The solar photosphere provides the most precisely determined set of elemental abundances for any astronomical object, serving as the reference standard against which all other stellar compositions are compared. Current best estimates place the solar mass fractions at X = 0.7381 (hydrogen), Y = 0.2485 (helium), and Z = 0.0134 (all heavier elements combined), with oxygen, carbon, neon, and iron as the four most abundant metals in that order.18 Every atom of these elements heavier than lithium was forged in a star that lived and died before the solar nebula condensed, making the solar system a direct inheritor of billions of years of cosmic nucleosynthesis.5, 22
References
The r-process of stellar nucleosynthesis: astrophysics and nuclear physics achievements and mysteries
Origin of the heavy elements in binary neutron-star mergers from a gravitational-wave event