Overview
- The evolutionary transition from synapsids to mammals spanned roughly 100 million years, from the Carboniferous pelycosaurs through the Permian therapsids to the Late Triassic mammaliaforms, and is documented by hundreds of fossils preserving gradual, mosaic changes in skull, jaw, ear, and dental anatomy.
- The transformation of the jaw joint from the ancestral articular-quadrate articulation to the mammalian dentary-squamosal joint, with the former jaw bones repurposed as middle-ear ossicles, is one of the most completely documented major anatomical transitions in the vertebrate fossil record.
- Mesozoic mammals were far more ecologically diverse than traditionally assumed, with multituberculates, docodonts, and eutriconodonts occupying climbing, burrowing, swimming, and herbivorous niches tens of millions of years before the end-Cretaceous extinction opened new ecological opportunities.
The origin of mammals from their synapsid ancestors is one of the most thoroughly documented evolutionary transitions in the vertebrate fossil record. Spanning roughly 100 million years from the Carboniferous Period to the Late Triassic, the transition encompasses hundreds of fossil species that preserve gradual, mosaic changes in skull architecture, jaw mechanics, dental morphology, limb posture, and sensory anatomy.1, 8 The transformation is not a single event but a protracted series of overlapping innovations: the progressive enlargement of the dentary bone and reduction of the postdentary elements, the development of a secondary bony palate, the differentiation of teeth into incisors, canines, and complex cheek teeth, and the repurposing of bones that once formed the jaw joint into the tiny ossicles of the mammalian middle ear. Each of these changes is preserved in intermediate states across a dense succession of fossil forms, making the synapsid-to-mammal lineage one of the most powerful case studies in macroevolutionary biology.2, 3, 6
It is important to note that the phrase "reptile-to-mammal transition," though widely used in older literature, is a misnomer. Synapsids are not reptiles and never were. The synapsid and reptile lineages diverged from a common amniote ancestor in the Carboniferous, more than 310 million years ago, and have been separate evolutionary lines ever since. Dimetrodon, the iconic sail-backed predator of the early Permian, is more closely related to humans than to any dinosaur or living reptile.23 The term "mammal-like reptile," once standard in textbooks, has been abandoned by specialists in favour of "non-mammalian synapsid," which accurately reflects the phylogenetic position of these animals as members of the mammalian stem lineage rather than aberrant reptiles.1, 11
Basal synapsids and the pelycosaur grade
Synapsids are distinguished from all other amniotes by the possession of a single temporal fenestra, an opening in the skull behind each eye socket that provided expanded attachment area for jaw muscles. The earliest synapsids appear in the fossil record of the late Carboniferous, approximately 310 to 315 million years ago, and by the early Permian they had diversified into the dominant terrestrial vertebrate fauna of Pangaea.1, 11 These early forms, traditionally grouped as "pelycosaurs," are not a natural (monophyletic) group but rather a paraphyletic grade of basal synapsids from which the more derived therapsids evolved. Nevertheless, the pelycosaur grade is useful shorthand for the array of synapsid body plans that preceded the therapsid radiation.11

The best-known pelycosaur is Dimetrodon, a large apex predator of the early Permian (approximately 295 to 272 million years ago) that bore a dramatic dorsal sail formed by elongated neural spines of the vertebrae. Dimetrodon reached body lengths of more than three metres and body masses estimated at 100 to 150 kilograms. Despite its ubiquitous appearance in children's dinosaur toy sets, Dimetrodon predated the earliest dinosaurs by approximately 40 million years and belongs to the Sphenacodontidae, the family most closely related to the therapsid clade that ultimately produced mammals.23 The function of the sail has been debated for over a century; hypotheses include thermoregulation (the sail providing a large surface area for absorbing or radiating heat), species recognition, and sexual display. The independent evolution of a similar dorsal sail in the herbivorous pelycosaur Edaphosaurus, an only distantly related lineage, suggests that similar selective pressures acted on multiple synapsid clades.1, 23
Beyond the sphenacodontids, the pelycosaur grade included a range of ecological types: the herbivorous caseids, some of which reached body masses exceeding 500 kilograms and were the largest terrestrial vertebrates of the early Permian; the insectivorous varanopids; and the small, lizard-like eothyridids. Pelycosaurs dominated terrestrial ecosystems through the early to middle Permian, but their diversity declined sharply after approximately 270 million years ago as the therapsids, a more derived synapsid clade, radiated and replaced them.1, 7, 11
The therapsid radiation
Therapsids first appeared in the middle Permian, approximately 270 million years ago, and rapidly diversified into the dominant large-bodied terrestrial vertebrates of the late Permian. They are distinguished from pelycosaur-grade synapsids by a suite of derived features: an enlarged temporal fenestra with a reduced temporal arch above it, a more upright limb posture with the legs drawn partially beneath the body rather than sprawling laterally, and an expanded dentary bone in the lower jaw at the expense of the postdentary elements.1, 7 These changes reflect a shift toward more energetically efficient locomotion and more powerful, precisely controlled jaw mechanics.
Tom Kemp has proposed that the origin of therapsids resulted from a correlated progression of character evolution, in which increased metabolic activity, more efficient locomotion, and improved food processing mutually reinforced one another in a positive feedback loop.20 This hypothesis is consistent with the mosaic pattern of the fossil record, in which different therapsid lineages acquired different subsets of mammalian features at different rates, rather than all features evolving in lockstep along a single lineage.8
The major therapsid clades of the Permian include the dinocephalians, massive herbivores and carnivores that were the largest land animals of the middle Permian; the anomodonts, particularly the dicynodonts, which were the dominant herbivores of the late Permian and survived into the Triassic; the gorgonopsians, apex predators with large saber-like canine teeth; the therocephalians, a diverse group that included both carnivores and insectivores; and the cynodonts, the lineage that would ultimately give rise to mammals.7, 11 The end-Permian mass extinction approximately 252 million years ago devastated all therapsid groups, but several lineages survived and re-diversified in the Triassic, with the dicynodonts and cynodonts becoming particularly prominent components of early Triassic ecosystems.7
Cynodonts and the approach to mammalian anatomy
The cynodonts, whose name means "dog-tooth" in reference to their differentiated dentition, are the therapsid clade that includes the direct ancestors of mammals. First appearing in the late Permian approximately 260 million years ago, cynodonts radiated extensively in the Triassic and acquired, in a stepwise fashion, many of the anatomical features that define living mammals.1, 7
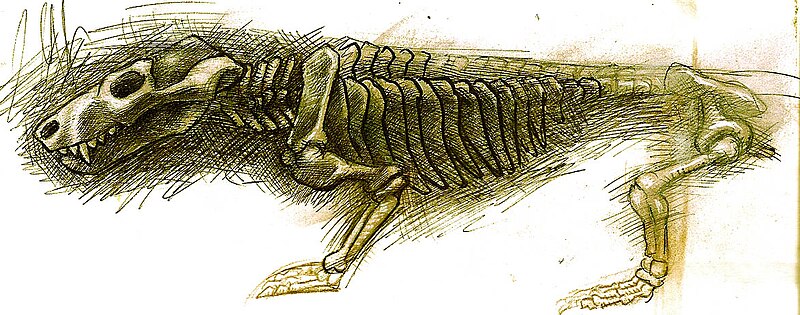
One of the most significant cynodont innovations is the secondary bony palate, a shelf of bone in the roof of the mouth that separates the nasal passage from the oral cavity. In animals without a secondary palate, the internal nostrils (choanae) open directly into the mouth, making it difficult to breathe and chew simultaneously. The secondary palate reroutes airflow from the nostrils to the back of the throat, allowing continuous breathing during prolonged chewing. This adaptation appears in progressively more complete form across the cynodont family tree, from the partial palate of the early cynodont Procynosuchus to the fully enclosed bony palate seen in advanced eucynodonts and all living mammals.1, 24 The secondary palate was not merely a convenience; it was an essential prerequisite for the sustained metabolic demands of endothermy, because an endothermic animal requires continuous, high-volume oxygen intake even while processing food.14, 20
Cynodont teeth became increasingly differentiated into distinct functional regions: incisors at the front for grasping and cutting, enlarged canines for piercing, and postcanine teeth with multiple cusps for shearing and crushing. In advanced cynodonts, the upper and lower postcanine teeth occlude precisely against one another, enabling the kind of efficient oral food processing that characterises mammals and that is absent in reptiles, which typically swallow food in large, unchewed pieces.2, 13 The development of precise dental occlusion is closely linked to the evolution of a more powerful and more precisely controlled jaw musculature, reflected in the progressive enlargement of the dentary bone and the expansion of the attachment area for the masseter and temporalis muscles.2
Evidence for endothermy in cynodonts comes from multiple independent lines of evidence. Oxygen isotope analysis of therapsid bone and tooth apatites, compared to co-existing ectothermic tetrapods from the same geological formations, demonstrates that eucynodonts maintained body temperatures significantly elevated above ambient environmental temperatures, consistent with endothermic metabolism. A 2017 study by Rey and colleagues concluded that mammalian endothermy originated in the Epicynodontia during the middle to late Permian, suggesting that elevated thermometabolism was acquired well before the origin of mammals themselves.14 Additional anatomical evidence includes the presence of infraorbital foramina in the snout bones of advanced cynodonts, consistent with a rich supply of blood vessels and nerves to the face, implying the presence of whiskers (vibrissae) and by extension fur or hair, which would have served as insulation for an endothermic animal.24
The jaw-to-ear transition
The transformation of the jaw joint is the single most celebrated anatomical change in the synapsid-to-mammal transition, and one of the most thoroughly documented evolutionary transitions in all of vertebrate paleontology. In non-mammalian amniotes, the lower jaw is composed of multiple bones: the dentary, which carries the teeth, the articular, the surangular, the angular, and several smaller elements. The jaw joint is formed by the articulation of the articular bone of the lower jaw with the quadrate bone of the skull. In living mammals, by contrast, the lower jaw consists of a single bone, the dentary, and the jaw joint is formed by the articulation of the dentary with the squamosal bone of the skull. The articular and quadrate bones have not been lost; instead, they have been repurposed as two of the three tiny ossicles of the mammalian middle ear, the malleus and incus respectively. The homology between these jaw bones and ear bones was recognised by comparative anatomists such as Karl Reichert in the 1830s and has been confirmed by embryological, genetic, and paleontological evidence.2, 3

The fossil record documents this transition in extraordinary anatomical detail across a succession of intermediate forms. In basal cynodonts, the dentary is large but still accompanied by several postdentary bones, and the jaw joint is formed exclusively by the articular-quadrate contact, as in the standard non-mammalian configuration. In more derived cynodonts such as Probainognathus and Diarthrognathus, the dentary has enlarged dramatically and a new contact has developed between the dentary and the squamosal, creating a condition in which two jaw joints exist simultaneously: the ancestral articular-quadrate joint and the novel dentary-squamosal joint.19 Diarthrognathus, a small tritheledontid cynodont from the Early Jurassic of South Africa whose name means "two-joint jaw," preserves this dual articulation with particular clarity and stands as one of the most striking transitional fossils in the vertebrate record.19, 22
In the earliest mammaliaforms, represented by Morganucodon from the Late Triassic to Early Jurassic (approximately 205 million years ago), the dentary-squamosal joint has become the primary load-bearing articulation, while the articular-quadrate joint persists but is reduced in size and no longer carries major chewing forces. The postdentary bones are present but tiny, forming what is termed a mandibular middle ear still connected to the jaw by a slender ossified rod of Meckel's cartilage.4, 12 In more derived Mesozoic mammals, the postdentary bones have separated entirely from the jaw and migrated into the middle ear, functioning solely as auditory ossicles. The Early Cretaceous eutriconodont Yanoconodon allini, described by Luo and colleagues in 2007, preserves the intermediate condition with exceptional clarity: its middle-ear bones are still connected to the lower jaw by Meckel's cartilage, documenting the precise anatomical stage at which the ear ossicles were in the process of detaching from the mandible.5
The fully detached three-boned middle ear of mammals is significantly more sensitive to high-frequency airborne sounds than the single-bone ear of non-mammalian amniotes. The impedance-matching function of the malleus-incus-stapes chain, amplifying sound pressure as vibrations are transmitted from the large tympanic membrane to the small oval window of the inner ear, was a major sensory innovation that likely enabled early mammals to detect insect prey and communicate at frequencies inaudible to their reptilian and dinosaurian contemporaries.3, 9 Recent fossil discoveries have revealed that this transition occurred independently in multiple mammalian lineages, with the detachment of the middle ear from the mandible evolving at least three times in parallel among monotremes, therians, and other mammaliaform groups, representing a remarkable instance of homoplasy driven by convergent functional demands.9, 22
Key stages in the synapsid jaw-to-ear transition2, 6, 9
| Taxon | Age | Jaw joint | Postdentary bones |
|---|---|---|---|
| Dimetrodon (pelycosaur) | ~295–272 Ma | Articular-quadrate only | Large, load-bearing |
| Thrinaxodon (basal cynodont) | ~252–247 Ma | Articular-quadrate only | Reduced but functional |
| Probainognathus (derived cynodont) | ~235 Ma | Dual: articular-quadrate + incipient dentary-squamosal | Small, partially load-bearing |
| Diarthrognathus (tritheledontid) | ~200 Ma | Dual: articular-quadrate + dentary-squamosal | Small, transitional |
| Morganucodon (mammaliaform) | ~205–190 Ma | Dentary-squamosal primary; articular-quadrate vestigial | Tiny, forming mandibular middle ear |
| Yanoconodon (eutriconodont) | ~122 Ma | Dentary-squamosal only | Detaching via Meckel's cartilage |
| Modern mammals (Theria) | Present | Dentary-squamosal only | Fully detached as middle-ear ossicles |
Morganucodon and other early mammaliaforms
Morganucodon, first described from fissure-fill deposits in Glamorgan, Wales, and subsequently found in Yunnan Province, China, and other sites across Europe and North America, is the most extensively studied early mammaliaform and one of the most important transitional fossils in vertebrate paleontology. It was a small animal, approximately 10 centimetres in body length, with a body plan broadly resembling that of a modern shrew. Morganucodon possessed differentiated teeth with precise occlusion, a dentary-squamosal jaw joint functioning as the primary articulation, and a braincase proportionally larger than that of its cynodont predecessors, but it retained a vestigial articular-quadrate joint and other features absent in crown-group mammals.4, 13, 15
The classification of Morganucodon illustrates the difficulty of drawing a sharp boundary between "non-mammal" and "mammal" in what is fundamentally a continuous evolutionary gradient. Timothy Rowe defined Mammalia in 1988 as the clade originating with the most recent common ancestor of living monotremes (platypus and echidnas) and therians (marsupials and placentals), a crown-group definition that excludes Morganucodon and places it among the mammaliaforms, the broader group of animals more closely related to living mammals than to any other living group but outside the crown.10 Under this definition, Morganucodon is not technically a mammal, though it possesses many features traditionally considered mammalian. Other workers have preferred a more inclusive definition based on the possession of the dentary-squamosal jaw joint, which would include Morganucodon within Mammalia. The debate is not about the fossils themselves but about where to draw an inherently arbitrary taxonomic line across a continuous morphological spectrum.6, 10
Another key early mammaliaform is Hadrocodium wui, a tiny animal from the Early Jurassic Lufeng Formation of Yunnan, China, approximately 195 million years old. Hadrocodium is remarkable for possessing an expanded braincase and, critically, no postdentary trough on the mandible, indicating that the middle-ear bones had already separated from the jaw. This extends the earliest evidence for the fully detached mammalian middle ear to approximately 45 million years earlier than previously documented and suggests that the brain expansion and middle-ear separation may have been functionally correlated.4
Mosaic evolution and the gradual acquisition of mammalian characters
A central finding of synapsid paleontology is that the features defining living mammals did not evolve as a coordinated package but accumulated gradually and in different combinations across different lineages, a pattern termed mosaic evolution. Sidor and Hopson demonstrated in a 1998 quantitative analysis that the temporal pattern of character acquisition in the synapsid lineage leading to mammals shows a statistically significant positive correlation between the number of derived features acquired and the elapsed geological time, consistent with gradual, sustained character evolution rather than punctuated bursts of rapid change.8
This mosaic pattern is visible at every level of the transition. Some cynodonts evolved a secondary palate but retained a primitive jaw joint. Others developed precise dental occlusion while still possessing a sprawling limb posture. The detachment of the middle-ear bones from the mandible occurred independently in at least three separate mammaliaform lineages.9, 22 Zhe-Xi Luo characterised early mammalian evolution as "a complex branching network with a number of dead ends where mammalian features evolved repeatedly in separate lineages and were sometimes lost," rather than a simple linear progression from reptile to mammal.6
This pattern has implications for how the synapsid-to-mammal transition is understood. There is no single moment at which "reptiles became mammals." Instead, the features that collectively define Mammalia were assembled over roughly 100 million years, with different components appearing at different times, in different lineages, and sometimes being lost and re-evolved. The fossil record preserves not a ladder but a bush, with numerous side branches experimenting with different combinations of mammalian and non-mammalian features, most of which went extinct without leaving modern descendants.6, 8
Molecular clocks and the fossil record
The timing of key events in the synapsid-to-mammal transition has been addressed by both the fossil record and molecular clock analyses, and the two approaches have not always agreed. Molecular divergence estimates, which use the rate of DNA sequence change calibrated against securely dated fossils to extrapolate the timing of lineage splits, have generally suggested that the major mammalian lineages diverged earlier than the oldest known fossils of those lineages would indicate. Bininda-Emonds and colleagues estimated in 2007 that the divergence of modern placental mammalian orders began deep in the Cretaceous, approximately 100 to 85 million years ago, tens of millions of years before the oldest fossil representatives of most orders appear in the Paleocene.21
The discrepancy between molecular and fossil-based divergence estimates is a general feature of mammalian phylogenetics and is not unique to the synapsid-to-mammal transition specifically. It reflects, in part, the inevitable incompleteness of the fossil record: the earliest members of a lineage are the least likely to be preserved and discovered, meaning that the oldest known fossil of any group will almost always postdate the true divergence of that group from its closest relative. Molecular clocks, for their part, are sensitive to assumptions about rate constancy, calibration points, and the models used to account for rate variation across lineages, and can produce systematically older or younger estimates depending on methodology.6, 21
For the earlier portions of the transition, the fossil record is remarkably dense. The synapsid lineage from pelycosaurs through therapsids to mammaliaforms is sampled at numerous points across the Permian and Triassic, and the morphological changes are documented in sufficient detail that quantitative analyses of character acquisition rates are possible. However, even here, ghost lineages, the intervals of time during which a lineage must have existed based on the ages of its relatives but for which no fossils are known, indicate that substantial portions of the record remain undiscovered.8 The ongoing pace of new fossil discoveries from China, South America, and Africa continues to fill gaps and refine the timeline of the transition.6, 22
Mesozoic mammalian diversity
The traditional view of Mesozoic mammals as a uniform assemblage of small, nocturnal, insect-eating animals has been overturned by discoveries made primarily since the 1990s. Far from being marginal players in dinosaur-dominated ecosystems, Mesozoic mammals achieved a remarkable range of body sizes, dietary specialisations, and locomotor adaptations. Close and colleagues demonstrated in 2015 that mammalian morphological evolution was significantly elevated during the Early to Middle Jurassic, constituting a major adaptive radiation that coincided with a stepwise increase in long-term diversity and the attainment of maximum morphological disparity.17
The multituberculates are the most diverse order of Mesozoic mammals, with more than 200 described species ranging from the Middle Jurassic to the Eocene, a temporal span of roughly 130 million years that makes them the longest-lived mammalian order in history. Multituberculates are named for their distinctive cheek teeth, which bear rows of multiple cusps (tubercles) suited to an omnivorous or herbivorous diet. They occupied ecological roles analogous to those of modern rodents and were both numerically abundant and taxonomically diverse in Late Cretaceous ecosystems. Wilson and colleagues showed in 2012 that multituberculate adaptive radiation accelerated at least 20 million years before the end-Cretaceous extinction, driven by dietary expansion toward herbivory during the ecological rise of flowering plants (angiosperms).15, 16
The docodonts, a clade of mammaliaforms known primarily from the Jurassic, further illustrate the ecological breadth of early mammals. In 2015, Meng and colleagues described an arboreal docodont from the Middle Jurassic of China with skeletal adaptations for climbing and dental features indicating an omnivorous diet that included plant sap, revealing that tree-dwelling lifestyles had been adopted by mammaliaforms far earlier than previously suspected. Other docodonts show adaptations for burrowing and swimming, demonstrating that a single mammaliaform clade could encompass a diversity of locomotor and dietary strategies comparable to that seen in modern mammalian families.18
The competitive displacement debate has centred on whether the coexistence of mammals with non-avian dinosaurs suppressed mammalian diversification. The evidence is mixed. Mesozoic mammals were overwhelmingly small, with body masses rarely exceeding a few kilograms, and the absence of large-bodied mammals during the Mesozoic may reflect ecological constraint imposed by dinosaurian dominance of large-bodied niches. However, within the small-body-size range, Mesozoic mammals achieved levels of morphological disparity and ecological specialisation that rival those of comparable modern clades.6, 15, 17 The end-Cretaceous extinction 66 million years ago removed non-avian dinosaurs and opened large-body-size niches to mammals, but the raw material for the subsequent Cenozoic radiation, including dental complexity, locomotor diversity, and sensory sophistication, had been accumulating for over 100 million years during the Mesozoic.6, 21
Significance of the transition
The synapsid-to-mammal transition holds a special place in evolutionary biology for several reasons. First, the sheer density and quality of the fossil record make it possible to trace the acquisition of mammalian characters with a precision rarely achieved for any other major evolutionary transition. The number of intermediate forms is so large, and the morphological gaps between them so small, that the transition has been described as one of the most complete gradational series in the vertebrate fossil record.1, 8
Second, the transition illustrates fundamental evolutionary principles. The jaw-to-ear transformation demonstrates how natural selection can repurpose existing structures for entirely new functions, a process termed exaptation. The mosaic pattern of character acquisition shows that major evolutionary transitions do not occur as sudden leaps but as the gradual, overlapping, and sometimes convergent accumulation of individual innovations across multiple lineages. The independent evolution of the detached mammalian middle ear in at least three separate lineages demonstrates that convergent evolution can produce remarkably similar anatomical outcomes when similar functional demands are at work.6, 9, 22
Third, the transition spans a sufficiently long interval, roughly 100 million years, that it provides a natural laboratory for studying the tempo and mode of macroevolution. Quantitative analyses of character acquisition rates across the synapsid phylogeny have established that evolution proceeded gradually and relatively uniformly, with no evidence for a sudden burst of mammalian character evolution at any single point in the sequence.8 This conclusion is robust across multiple analytical methods and data sets, and it provides empirical support for the view that major evolutionary innovations are typically the product of sustained, incremental change rather than saltational leaps. The continuous spectrum from pelycosaur through therapsid through cynodont through mammaliaform to crown mammal is, in this respect, a model case study for understanding how the accumulated small changes of microevolution produce the large-scale transformations of macroevolution.1, 6, 8
References
Ghost lineages and ‘mammalness’: assessing the temporal pattern of character acquisition in the Synapsida
Oxygen isotopes suggest elevated thermometabolism within multiple Permo-Triassic therapsid clades
The origin and early radiation of the therapsid mammal-like reptiles: a palaeobiological hypothesis
Explosive radiation of Meso-Cenozoic mammals inferred by molecular phylogenetics and the fossil record
Dimetrodon is not a dinosaur: using tree thinking to understand the ancient relatives of mammals and their evolution
Craniodental anatomy in Permian–Jurassic Cynodontia and Mammaliaformes as a gateway to defining mammalian soft tissue and behavioural traits