Overview
- Multicellularity has evolved independently at least 25 times across the tree of life, including in animals, land plants, fungi, and multiple algal lineages, making it one of the most recurrent major evolutionary transitions in the history of life.
- Two fundamental routes to multicellularity exist: clonal development, in which cells remain attached after division and produce genetically identical collectives, and aggregative development, in which previously independent cells come together, producing potentially chimeric groups — with clonal development associated with all lineages that have achieved complex tissue-level organization.
- Experimental evolution studies have demonstrated that simple multicellularity can arise de novo in laboratory populations within hundreds of generations, confirming that the initial transition from unicellular to multicellular life may be far less difficult than achieving the complex cell differentiation and division of labor that characterize animals, plants, and fungi.
Multicellularity — the organization of an organism's body from cooperating cells rather than a single autonomous cell — is one of the major evolutionary transitions in the history of life. In their influential 1995 framework, John Maynard Smith and Eörs Szathmáry identified the transition from unicellular to multicellular life as one of a small number of events in which previously independent replicating units became integrated into a higher-level entity with a new, collective capacity for reproduction and selection.1 Unlike some other major transitions that appear to have occurred only once — such as the origin of the genetic code or the endosymbiotic acquisition of mitochondria — multicellularity has evolved independently on at least 25 separate occasions across the tree of life, in bacteria, archaea, and eukaryotes alike.2, 3 This remarkable recurrence suggests that the initial steps toward multicellular organization are comparatively accessible from unicellular ancestors, even as the subsequent evolution of complex tissue-level differentiation has proven far more restricted.
The organisms that have achieved complex multicellularity — defined as three-dimensional bodies with differentiated cell types and specialized tissues for bulk transport of nutrients and signals — belong to only a handful of lineages: animals, embryophytic land plants, florideophyte red algae, laminarialean brown algae, and two groups of fungi (ascomycetes and basidiomycetes).3 Understanding why multicellularity arose so often, yet complex multicellularity so rarely, is one of the central questions in evolutionary biology and has driven research spanning comparative genomics, experimental evolution, developmental biology, and paleontology.
Defining multicellularity
The concept of multicellularity encompasses a wide spectrum of biological organization, from loose colonies of essentially identical cells to the highly integrated bodies of animals and plants with hundreds of distinct cell types. Distinguishing these grades is essential for understanding how the transition has occurred and what it has produced. Simple multicellularity refers to organisms in which cells are attached to one another but remain in direct contact with the external environment, lacking the bulk transport systems that would permit interior cells to receive nutrients and oxygen independently. Filamentous cyanobacteria, many fungi, and colonial algae exemplify this grade.3 Complex multicellularity, by contrast, involves three-dimensional organization with cellular differentiation and intercellular communication systems that allow cells deep within the body to be provisioned by specialized transport tissues. Only the six lineages noted above have achieved this grade.3, 4
This distinction matters because the barriers to simple and complex multicellularity are very different. Simple multicellularity may require nothing more than incomplete cell separation after division or the expression of adhesion molecules that cause cells to stick together. Complex multicellularity demands a far more elaborate genetic and developmental toolkit: mechanisms for cell-cell signaling, coordinated gene regulation across differentiated cell types, programmed cell death for sculpting tissues, and channels or vascular systems for bulk transport.3, 10 The relative ease of the first step and the difficulty of the second help explain the paradox that multicellularity has evolved dozens of times while complex multicellularity has evolved only a few times.
Routes to multicellularity
All multicellular organisms originate through one of two fundamental developmental routes. In clonal development (sometimes called the "staying together" route), a single founding cell divides and the daughter cells remain physically attached, producing a genetically uniform multicellular body. Because every cell in the resulting organism is a clone of the founder, genetic conflict between cells is minimal, and kin selection strongly favors cooperation: any gene that promotes the collective good automatically benefits copies of itself in every other cell of the organism.2, 11 Animals, land plants, and most complex multicellular organisms develop clonally, beginning life as a single cell (a zygote or spore) and building the adult body through successive rounds of mitotic division.
In aggregative development (the "coming together" route), previously free-living cells aggregate into a multicellular collective. The cellular slime mold Dictyostelium discoideum is the paradigmatic example: when starved, individual amoebae stream together to form a multicellular slug that differentiates into a fruiting body with dead stalk cells and viable spores.2 Aggregative multicellularity also occurs in the social amoeba Copromyxa, the filasterean Capsaspora owczarzaki, and certain bacteria such as Myxococcus xanthus. Because aggregation can produce genetically heterogeneous (chimeric) groups, it opens the door to social conflict: a mutant genotype that avoids the costly stalk-cell fate and disproportionately enters the spore population gains a short-term advantage at the expense of the group.11, 18
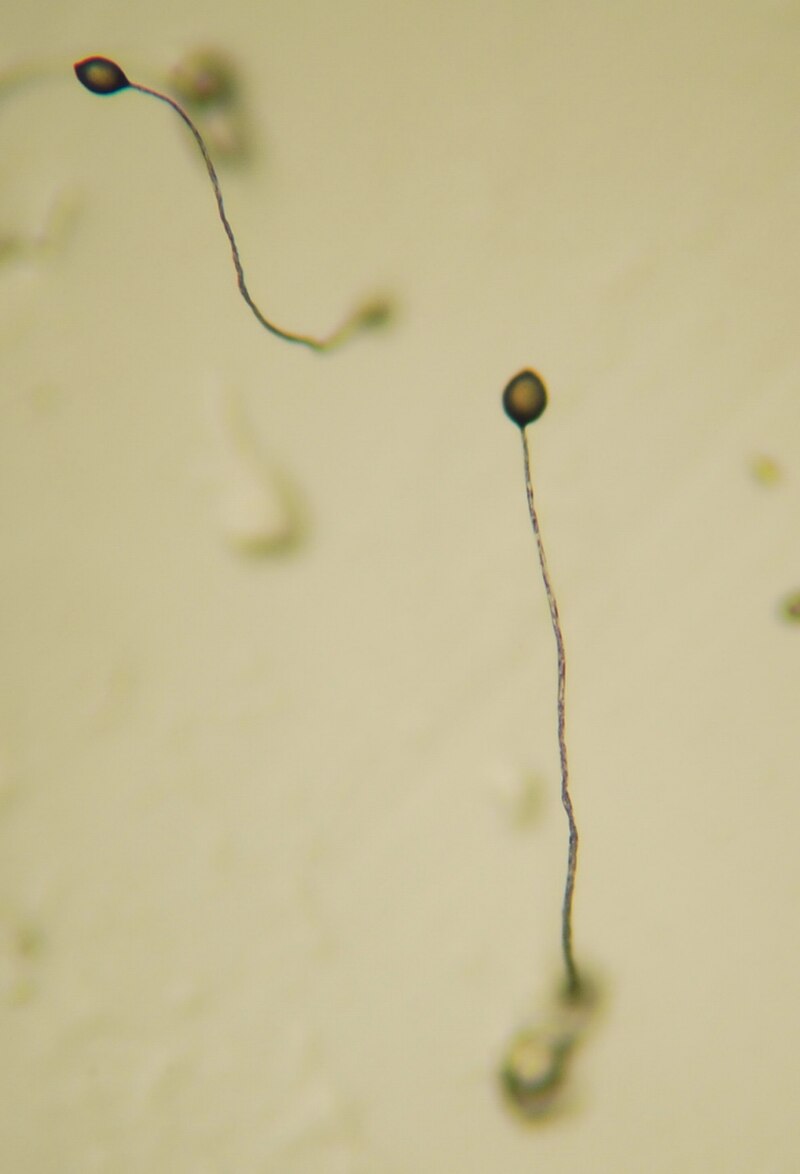
Experimental comparisons between clonal and aggregative development in isogenic yeast have demonstrated that the mechanism of group formation has profound evolutionary consequences. Clonal "snowflake yeast" groups, which arise through incomplete cell separation, evolve larger and more complex morphologies under selection for increased size, while aggregative "floc yeast" groups, which form through reversible cell-surface binding, remain simple because genetic cheaters readily exploit the group-formation mechanism.18 This finding provides direct experimental support for the longstanding theoretical prediction that high genetic relatedness among cells is a prerequisite for the evolution of complex cooperative traits, including cellular differentiation and self-sacrifice through programmed cell death.11, 16
How often has multicellularity evolved?
The number of independent origins of multicellularity depends on the criterion used to define it. If simple colonial aggregates and undifferentiated filaments are counted, multicellularity has arisen on the order of 25 or more times among eukaryotes alone, plus at least once in bacteria (cyanobacteria and myxobacteria) and possibly in archaea.2, 3 Among eukaryotes, independent origins include the animals (Metazoa), the land plants (Embryophyta), the brown algae (Phaeophyceae), the red algae (Rhodophyta), multiple fungal lineages, the volvocine green algae, the ulvophyte green algae, the charophyte green algae, the diatoms, the oomycetes, and several groups of slime molds and social amoebae.2
Complex multicellularity, however, has evolved far fewer times. Andrew Knoll's 2011 analysis identified only six eukaryotic clades in which complex, three-dimensional bodies with differentiated tissues and bulk transport have arisen: animals, embryophytic land plants, florideophyte red algae, laminarialean brown algae, and two fungal lineages (the ascomycetes and basidiomycetes forming complex fruiting bodies).3 Each of these transitions appears to have been independent, as the last common ancestor of any two of these groups was unicellular. The sheer disparity between the number of origins of simple multicellularity (many) and complex multicellularity (few) underscores that the initial transition to group living is comparatively easy, while the subsequent elaboration of differentiated tissues and integrated physiology is a much rarer achievement.3, 16
Independent origins of multicellularity across eukaryotic lineages2, 3
| Lineage | Type | Development | Complex? |
|---|---|---|---|
| Animals (Metazoa) | Heterotroph | Clonal | Yes |
| Land plants (Embryophyta) | Phototroph | Clonal | Yes |
| Red algae (Florideophyceae) | Phototroph | Clonal | Yes |
| Brown algae (Laminariales) | Phototroph | Clonal | Yes |
| Fungi (Ascomycota, Basidiomycota) | Heterotroph | Clonal | Yes |
| Volvocine green algae | Phototroph | Clonal | No |
| Cellular slime molds (Dictyostelium) | Heterotroph | Aggregative | No |
| Myxobacteria | Heterotroph | Aggregative | No |
| Filamentous cyanobacteria | Phototroph | Clonal | No |
Selective advantages of multicellular life
For multicellularity to evolve, group living must confer a fitness advantage sufficient to offset the costs of reduced individual autonomy. Several selective pressures have been proposed and, in some cases, experimentally demonstrated. Predation resistance is among the best supported. Larger organisms are more difficult for size-selective predators to capture, and simple multicellular clusters can exceed the maximum prey size of filter-feeding or engulfing predators. In a landmark experiment, Herron and colleagues subjected populations of the unicellular green alga Chlamydomonas reinhardtii to predation by the ciliate Paramecium tetraurelia. Within approximately 750 asexual generations, two of five replicate populations evolved heritable multicellular clusters that were not observed in predator-free control populations, directly demonstrating that predation can select for de novo multicellularity.12
Enhanced motility and feeding represent another class of advantages. A multicellular body can generate stronger locomotory currents and sweep larger volumes of water for food particles than a single cell, and coordination among cells can produce directed swimming that individual cells cannot achieve. The volvocine algae illustrate this gradient: the unicellular Chlamydomonas swims with two flagella, the 16-cell Gonium colony coordinates its flagella to achieve directional swimming, and the 500- to 50,000-cell Volvox executes phototactic turns by differential beating of flagella on its light-exposed and shaded hemispheres.7, 15
Metabolic cooperation is a third advantage. Cells within a multicellular group can share metabolic products, buffer each other against environmental fluctuations, and exploit resources unavailable to single cells. Filamentous cyanobacteria such as Anabaena differentiate a fraction of their cells into heterocysts, specialized nitrogen-fixing cells that supply combined nitrogen to the rest of the filament in exchange for carbon fixed by the vegetative cells. This division of metabolic labor allows the filament to simultaneously carry out two biochemically incompatible processes — oxygenic photosynthesis and oxygen-sensitive nitrogen fixation — that no single cell can perform at once.2, 3
Finally, the evolution of a reproductive division of labor — the separation of somatic (non-reproductive) and germline (reproductive) cell fates — represents the deepest form of multicellular cooperation. In organisms with germ-soma differentiation, somatic cells sacrifice their own reproductive potential to support the reproduction of the germ cells, a form of altruism that is stable only when the cells share a recent common ancestor and therefore have high genetic relatedness.10, 11 Richard Michod has modeled this process as a "fitness trade-off" at the cellular level: as cells specialize in either viability (soma) or fecundity (germ), the fitness of the individual cells declines, but the fitness of the collective organism increases because specialization improves the efficiency of both functions.10
Genetic prerequisites
Comparative genomics has revealed that the genetic toolkit required for multicellularity was not assembled de novo at the moment of each transition but was largely present in the unicellular ancestors of multicellular lineages. Across independent origins of multicellularity, three categories of genes are consistently implicated: those involved in cell adhesion, cell-cell signaling, and transcriptional regulation of cell differentiation.3, 4
Cell adhesion molecules allow cells to bind to one another and to an extracellular matrix, creating the physical cohesion necessary for a multicellular body. In animals, the cadherin and integrin protein families serve these roles and were long thought to be metazoan innovations. However, the sequencing of the Monosiga brevicollis genome — a choanoflagellate, the closest unicellular relative of animals — revealed homologues of cadherin domains, C-type lectins, and tyrosine kinase signaling components, demonstrating that key elements of the animal adhesion and signaling toolkit were present before the origin of animal multicellularity.6 Similarly, unicellular relatives of plants and fungi possess homologues of the cell-wall synthesis enzymes and polysaccharide-based adhesion molecules used by their multicellular descendants.3, 4
Cell-cell communication allows cells within a multicellular body to coordinate their behavior, and all complex multicellular organisms possess molecular channels that facilitate the transfer of nutrients and signaling molecules between adjacent cells. In animals, gap junctions serve this role; in plants, plasmodesmata; in fungi, septal pores. Knoll has argued that the evolution of such intercellular channels is a critical bottleneck for complex multicellularity, because only lineages possessing them have been able to build three-dimensional bodies with interior cells that depend on neighbors for nutrient supply.3
Transcription factor diversification is the third pillar. In each independently evolved complex multicellular lineage, the transition to multicellularity is associated with the expansion of particular transcription factor families that regulate cell-type-specific gene expression. Animals have expanded the homeobox, bHLH, and zinc-finger families; land plants have expanded the MADS-box and MYB families; fungi have expanded C2H2 zinc-finger transcription factors.3, 4 The independent expansion of different transcription factor families in different lineages indicates that the underlying principle — the need for regulatory complexity to control cell differentiation — is universal, even though the specific molecular solutions are convergent rather than homologous.
The volvocine algae as a model system
The volvocine green algae form a lineage of closely related organisms spanning the full spectrum from unicellularity to complex multicellularity with a complete division of reproductive labor, making them one of the most informative model systems for studying the evolution of multicellularity. The lineage includes the unicellular Chlamydomonas reinhardtii, which possesses a single chloroplast, two flagella, and a well-characterized genome; the small colonial genus Gonium, a flat sheet of 8 to 16 cells held together by an extracellular matrix; the intermediate genus Pleodorina, in which a fraction of cells are specialized for somatic functions; and the large spheroidal genus Volvox, which contains up to 50,000 cells of which the vast majority are terminally differentiated somatic cells that will never reproduce, with only a small number of interior germ cells (gonidia) giving rise to the next generation.7
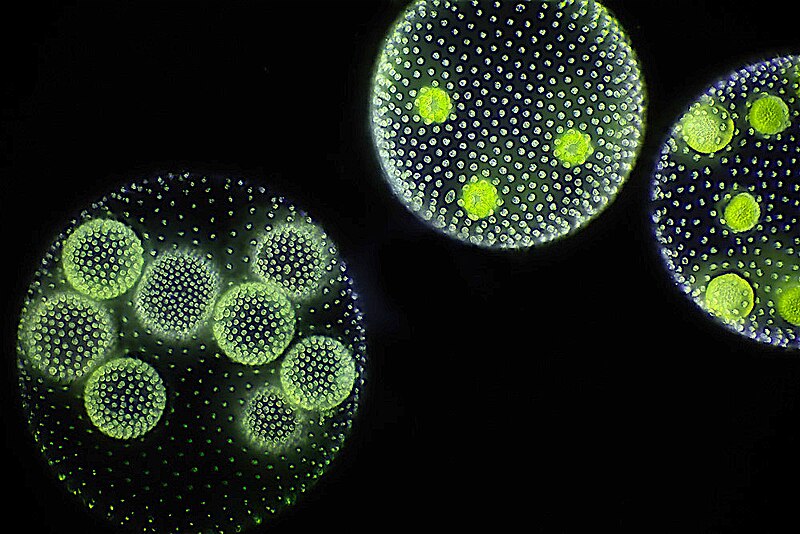
David Kirk proposed a "twelve-step program" for the evolution of Volvox from a Chlamydomonas-like ancestor. Each step corresponds to a specific developmental modification: incomplete cytokinesis (cells remaining attached after division), inversion of the embryo so that flagella face outward, rotation of basal bodies for coordinated swimming, evolution of an expanded extracellular matrix, genetic control of cell number, evolution of enlarged reproductive cells (gonidia), asymmetric cell division producing cells of different sizes, bifurcation of the cell fate program into somatic and germline tracks, terminal differentiation of somatic cells, and the loss of somatic cell reproductive capacity through regA-mediated gene regulation.7 The sequence is not merely hypothetical: extant volvocine species appear to occupy intermediate positions along this trajectory, and molecular phylogenetic analyses have confirmed that the steps occurred roughly in the order Kirk proposed, though with some lineage-specific variations.
Molecular clock analyses using multiple fossil calibration points have estimated that Volvox diverged from a unicellular ancestor approximately 200 million years ago, during the Triassic period, substantially earlier than the 50 to 75 million years previously assumed.15 This dating implies that the evolutionary elaboration from unicellular Chlamydomonas-like organisms to the fully differentiated body plan of Volvox occurred over a timescale of roughly 200 million years, placing meaningful constraints on the rate at which the twelve developmental steps accumulated. The volvocine lineage demonstrates that the genetic distance between unicellularity and a fully differentiated germ-soma division of labor need not be vast: the Volvox carteri genome contains approximately the same number of protein-coding genes as the Chlamydomonas reinhardtii genome, and the key differences involve the co-option and regulatory rewiring of pre-existing genes rather than the invention of fundamentally new ones.7
Choanoflagellates and the origin of animals
The choanoflagellates — a clade of aquatic, bacterivorous protists characterized by a single flagellum surrounded by a collar of microvilli — are the closest known unicellular relatives of animals. Their phylogenetic position, as the sister group to the Metazoa, makes them indispensable for reconstructing the cellular and genomic features of the last unicellular ancestor of animals.5, 6 Several choanoflagellate species form simple multicellular colonies: the marine species Salpingoeca rosetta can exist as a single cell, a chain colony, or a spherical rosette colony in which cells are physically connected and coordinate their flagellar beating to generate feeding currents.5
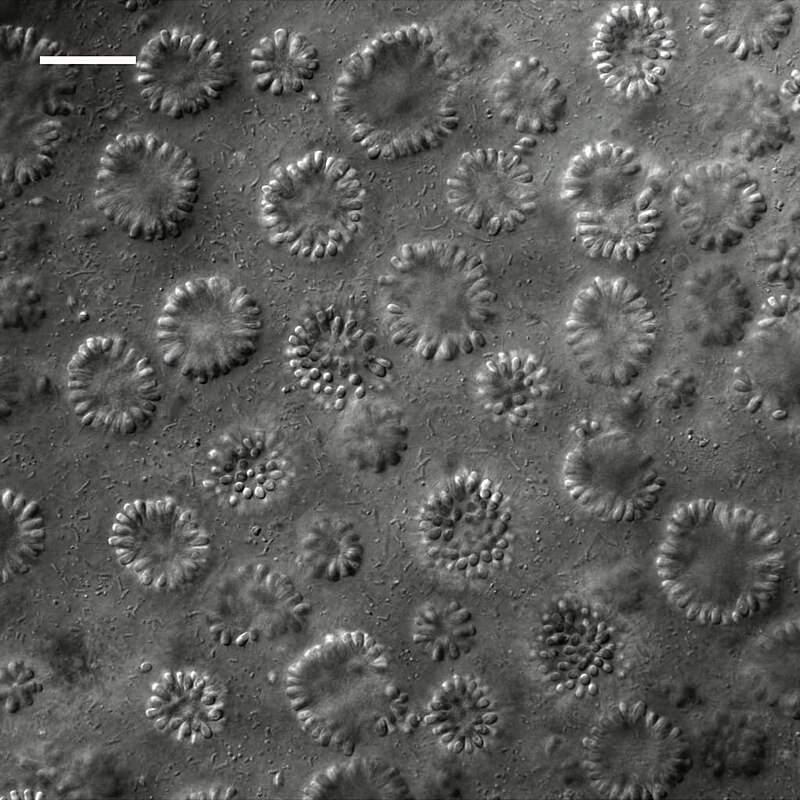
The 2008 sequencing of the Monosiga brevicollis genome revealed a gene repertoire far richer in "animal" signaling and adhesion domains than anyone had anticipated. The genome encodes tyrosine kinase signaling proteins, cadherin-domain proteins, C-type lectins, and other molecules previously considered exclusive to metazoans.6 Subsequent genomic and transcriptomic analyses of additional choanoflagellate species and other unicellular holozoans — including the filasterean Capsaspora owczarzaki and the ichthyosporean Creolimax fragrantissima — have expanded this picture. These organisms possess homologues of numerous metazoan transcription factors, components of the Wnt and Notch signaling pathways, and elements of the extracellular matrix toolkit, indicating that a substantial fraction of the genetic infrastructure for animal multicellularity was assembled before the multicellular transition itself.17
Brunet and King have proposed that the origin of animal multicellularity occurred through the modification of pre-existing mechanisms for extracellular matrix synthesis, regulation of cytokinesis, and temporal cell-type differentiation. In Salpingoeca rosetta, rosette colony formation is induced by a specific bacterial lipid, suggesting that the earliest multicellular animal ancestors may have formed colonies in response to environmental cues rather than as an obligate developmental program.5 The reconstruction of the last common ancestor of animals and choanoflagellates points to a cell that was already capable of regulated cell adhesion, receptor-mediated signal transduction, and the expression of at least several differentiated cell states. The metazoan innovation was not the invention of these capacities from scratch but their integration into a stable, heritable multicellular body plan with a dedicated germ-soma axis.5, 17
Experimental evolution of multicellularity
One of the most transformative developments in the study of multicellularity has been the application of experimental evolution, in which researchers subject unicellular organisms to selective pressures that favor group formation and then observe the resulting evolutionary trajectories in real time. These experiments have demonstrated that the initial transition to multicellularity can occur rapidly and repeatedly under appropriate selection.
In 2012, William Ratcliff and colleagues at the University of Minnesota applied a simple selective protocol to the unicellular yeast Saccharomyces cerevisiae: they subjected populations to daily settling selection, allowing clusters of cells to sediment through liquid medium more rapidly than single cells and preferentially transferring the fast-settling fraction to fresh medium. Within 60 days (approximately 350 generations), all ten replicate populations had independently evolved multicellular "snowflake" clusters that arose through incomplete cell separation after budding. These clusters exhibited a novel multicellular life cycle characterized by juvenile growth, determinate adult size, and reproduction through the release of multicellular propagules when internal stress caused branches to fracture.8
A 2023 follow-up study by Ratcliff and collaborators extended this selection experiment to 3,000 generations, subjecting populations to progressively stronger selection for increased group size. After 600 rounds of selection, snowflake yeast in the anaerobic treatment evolved to macroscopic dimensions, becoming approximately 20,000 times larger than their ancestors and reaching the millimeter scale visible to the unaided eye. The evolved organisms accomplished this through biophysical adaptation: cells became elongated, reducing the strain from cellular packing and facilitating branch entanglements that held groups together even after many cellular bonds fractured. The groups also evolved increased toughness, withstanding approximately 10,000 times more physical stress than their ancestors.9 This result was significant because it demonstrated that the evolution of macroscopic multicellular body size — a hallmark of complex multicellularity in animals, plants, and fungi — can arise de novo under sustained directional selection in a simple model organism.
Increase in group size during experimental evolution of snowflake yeast8, 9
In parallel, experiments with the unicellular green alga Chlamydomonas reinhardtii have demonstrated that predation, rather than settling selection, can also drive de novo multicellularity. When Chlamydomonas populations were exposed to the ciliate predator Paramecium tetraurelia, two of five experimental populations evolved heritable multicellular clusters within approximately 750 asexual generations. The multicellular phenotype persisted in the absence of predators, indicating that it was genetically encoded rather than a transient plastic response.12 Together, these experiments establish that the unicellular-to-multicellular transition is evolutionarily accessible from multiple starting points and under multiple selective regimes, consistent with its repeated independent occurrence in nature.
The fossil record of early multicellular life
The fossil record provides direct, if incomplete, evidence for the antiquity and pattern of multicellular evolution. The oldest widely accepted fossils of multicellular eukaryotes are filamentous and colonial forms preserved in Proterozoic sedimentary rocks. Bangiomorpha pubescens, a filamentous fossil from the approximately 1.05-billion-year-old Hunting Formation of arctic Canada, has been identified as a bangiacean red alga on the basis of its morphology, including the presence of spore-like structures interpreted as evidence of sexual reproduction. It represents the oldest taxonomically resolved complex multicellular eukaryote in the fossil record and the earliest direct evidence of sexual reproduction.13
Considerably older but far more controversial are the macroscopic fossils from the approximately 2.1-billion-year-old Francevillian Formation of Gabon, West Africa. In 2010, Abderrazak El Albani and colleagues reported centimeter-scale pyritized structures with coordinated morphologies from the Francevillian B Formation. The specimens, which grew to over 17 centimeters in length and displayed radial fabric and scalloped margins, were interpreted as colonial organisms that exhibited cell-to-cell signaling and coordinated growth in an oxygenated shallow-marine environment.14 If biogenic, these fossils would push the record of macroscopic multicellular life back by approximately one billion years relative to the Ediacaran biota. However, the interpretation remains debated: some researchers have argued that the structures could be abiogenic products of diagenetic pyritization, and the absence of preserved cellular detail makes it difficult to confirm that they represent true multicellular organisms.14
The Proterozoic fossil record includes numerous additional examples of simple multicellularity: filamentous microfossils in the approximately 1.9-billion-year-old Gunflint Formation, colonial cyanobacteria in stromatolites dating back more than 2.5 billion years, and eukaryotic acritarchs of uncertain affinity from Mesoproterozoic and Neoproterozoic sediments. The Ediacaran period (approximately 635 to 539 million years ago) marks the first appearance of large, architecturally complex multicellular organisms in the fossil record, including rangeomorphs, dickinsoniids, and possible early animals, preceding the Cambrian explosion of animal body plans by tens of millions of years.3 The fossil record thus suggests a long "fuse" in which simple multicellularity appeared early in Earth's history, while complex multicellularity with differentiated tissues emerged much later, during the Neoproterozoic and Phanerozoic.
The major transitions framework
The evolution of multicellularity is central to the broader theoretical framework of major evolutionary transitions, first articulated by Maynard Smith and Szathmáry in 1995 and subsequently refined by a generation of evolutionary theorists. A major transition occurs when previously independent replicating entities become integrated into a higher-level unit that itself becomes the target of natural selection. In the case of multicellularity, individual cells — each capable of independent reproduction — become subordinated to the collective organism, which reproduces as a unit while its constituent cells lose the ability to replicate independently outside the organism's body.1, 16
Szathmáry has distinguished between two types of major transition: fraternal transitions, in which the cooperating units are genetically similar or identical (as in the cells of a clonally developing multicellular organism), and egalitarian transitions, in which genetically dissimilar entities merge (as in the endosymbiotic origin of eukaryotes). The transition to multicellularity is the paradigmatic fraternal transition, because high relatedness among cells minimizes the potential for genetic conflict and provides the conditions under which altruistic behaviors, such as terminal somatic differentiation, can evolve through kin selection.11, 16
Michod has formalized this process in terms of fitness decoupling: during the transition from unicellularity to multicellularity, the fitness of the group (measured in terms of group-level reproduction and survival) becomes decoupled from the fitness of individual cells. In a unicellular organism, cell fitness and organism fitness are identical; in a mature multicellular organism with differentiated soma and germ, many cells have zero individual fitness (somatic cells that never reproduce) but contribute to high group fitness through their specialized roles in nutrient acquisition, defense, or structural support.10 The evolution of this fitness decoupling is, in Michod's framework, the defining feature of the transition to multicellular individuality.
The major transitions framework highlights an important general pattern: the evolution of cooperation and integration at one level of biological organization creates a new level at which selection can act, and the new higher-level individual can then evolve traits that would be impossible for its component parts. The complex morphology, physiology, and behavior of animals, plants, and fungi — the products of hundreds of millions of years of evolution after the multicellular transition — are consequences of this shift in the level of selection from the individual cell to the multicellular organism.1, 16
References
The origins of multicellularity and the early history of the genetic toolkit for animal development
Evolution of individuality during the transition from unicellular to multicellular life
Bangiomorpha pubescens n. gen., n. sp.: implications for the evolution of sex, multicellularity, and the Mesoproterozoic/Neoproterozoic radiation of eukaryotes
Large colonial organisms with coordinated growth in oxygenated environments 2.1 Gyr ago
Ecological advantages and evolutionary limitations of aggregative multicellular development