Overview
- Radiometric dating measures the predictable decay of radioactive isotopes to determine the age of rocks and minerals, with independent methods consistently converging on the same dates.
- Major systems include uranium-lead, potassium-argon, rubidium-strontium, samarium-neodymium, and radiocarbon, each calibrated against laboratory-measured decay constants and spanning timescales from centuries to billions of years.
- Cross-checking between independent systems, isochron plots, and concordia diagrams provide internal consistency tests that confirm the reliability of radiometric ages and rule out systematic errors.
Radiometric dating is the principal scientific method by which geologists and geochronologists determine the absolute ages of rocks, minerals, and other geological materials. The technique rests on the observation, first made systematic by Ernest Rutherford and Frederick Soddy in 1902, that certain unstable atomic nuclei transform spontaneously into different nuclei at rates that are fixed, predictable, and entirely independent of temperature, pressure, chemical state, or any other environmental variable.1 Because these transformation rates are constant, the ratio of original atoms to product atoms in a mineral serves as a natural clock, recording the time elapsed since the mineral crystallized and its internal isotopic system was effectively sealed. Over a century of laboratory measurement and geological application has established radiometric dating as one of the most precisely calibrated and internally consistent tools in all of the natural sciences.3, 4
Principles of radioactive decay
Every atom of a chemical element has a nucleus composed of protons and neutrons. While the number of protons defines the element, the number of neutrons can vary, producing different isotopes of the same element. Some combinations of protons and neutrons are energetically unstable, and the nucleus spontaneously rearranges itself by emitting radiation in a process called radioactive decay. The original, unstable isotope is called the parent, and the stable product is called the daughter. When uranium-238 decays, for instance, it eventually produces lead-206; when potassium-40 decays, it produces argon-40 and calcium-40; when carbon-14 decays, it produces nitrogen-14.1, 3
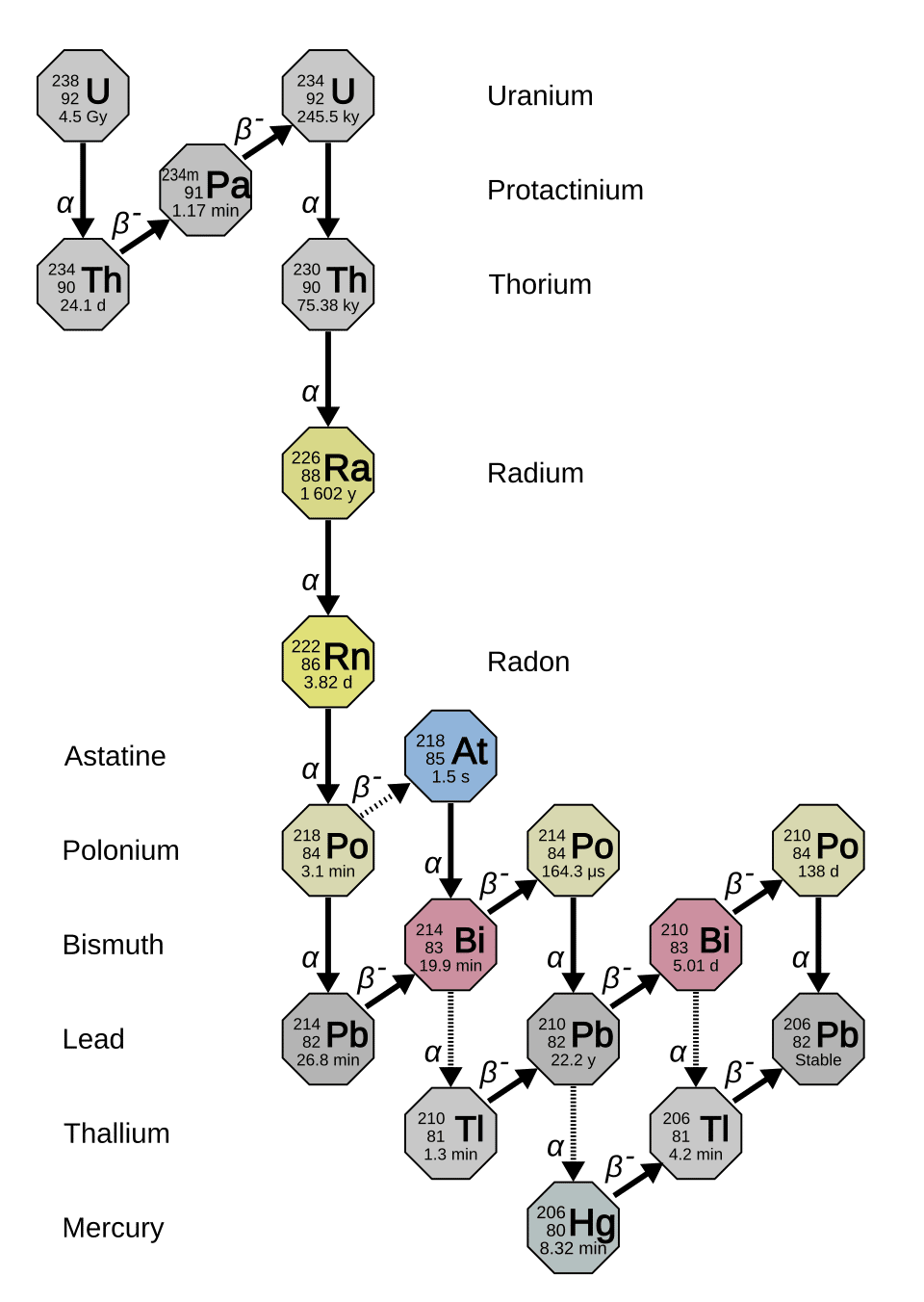
The rate of decay is governed by a fundamental physical constant, the decay constant (λ), which is unique to each radioactive isotope. The decay constant expresses the probability per unit time that any given nucleus will decay. This leads directly to the law of radioactive decay: the number of parent atoms decreases exponentially over time according to the equation N(t) = N0 e-λt, where N(t) is the number of parent atoms at time t, N0 is the initial number, and e is the base of the natural logarithm.2, 3 A closely related concept is the half-life (t1/2), which is the time required for exactly half of any given quantity of parent isotope to decay. Half-life and decay constant are mathematically linked by the simple relationship t1/2 = ln(2) / λ.19 Half-lives span an enormous range, from fractions of a second for some isotopes to billions of years for others, making different decay systems suitable for dating processes on very different timescales.
Because the number of daughter atoms produced equals the number of parent atoms lost, the age of a sample can be calculated by measuring the present-day ratio of daughter to parent atoms. The fundamental geochronological equation is D = D0 + N(eλt − 1), where D is the measured number of daughter atoms, D0 is the number of daughter atoms initially present when the mineral formed, N is the current number of parent atoms, and t is the age to be determined.3, 17 The central challenge in applying this equation is determining D0, the initial daughter isotope abundance, which is where different dating systems take different approaches—most notably through the isochron method described below.
Closure temperature and what a date means
A radiometric date does not measure the moment a rock first formed in some general sense; it measures the moment a specific mineral cooled below its closure temperature—the temperature at which diffusion of the daughter isotope out of the crystal lattice became negligibly slow and the isotopic clock effectively started ticking.21 The concept of closure temperature was rigorously formalized by geologist Martin Dodson in 1973. Different minerals have different closure temperatures for different isotopic systems, a property that geologists exploit deliberately. Zircon, for example, retains uranium and lead so tenaciously that its U-Pb closure temperature exceeds 900 degrees Celsius, meaning a zircon U-Pb date records the time of crystallization with great fidelity. Hornblende has an Ar-Ar closure temperature of roughly 500 degrees Celsius, while muscovite closes at around 350 degrees Celsius, and biotite at around 300 degrees Celsius.16, 21 By dating multiple minerals from the same rock with different closure temperatures, geologists can reconstruct the entire thermal history of a rock body, tracing how rapidly it cooled after being buried or intruded by magma—a field known as thermochronology.
It is important to recognize, then, that a "date" returned by a radiometric system is always the answer to a specific question: when did this particular mineral, in this particular isotopic system, cool below its closure temperature? For a volcanic lava that erupts and solidifies at the surface, cooling is rapid and the date closely approximates the eruption age. For a deep crustal rock that cools slowly over tens of millions of years, different minerals record different episodes of that cooling history. Understanding which question has been answered is fundamental to correct interpretation of radiometric data.3, 16
Major dating systems
Several radioactive decay chains are routinely used in geochronology. Each is suited to a particular range of ages and geological context, and their independent agreement on the ages of the same rocks constitutes one of the most powerful validations of the method.4, 14
Principal radiometric dating systems used in geochronology2, 3, 17
| System | Parent isotope | Daughter isotope | Half-life | Useful age range | Common materials |
|---|---|---|---|---|---|
| Uranium-Lead (238U-206Pb) | 238U | 206Pb | 4.468 billion yr | 1 million–4.6 billion yr | Zircon, monazite, baddeleyite |
| Uranium-Lead (235U-207Pb) | 235U | 207Pb | 703.8 million yr | 1 million–4.6 billion yr | Zircon, monazite, baddeleyite |
| Potassium-Argon (K-Ar / Ar-Ar) | 40K | 40Ar | 1.25 billion yr | 100,000 yr–4.6 billion yr | Feldspars, hornblende, biotite, whole rock |
| Rubidium-Strontium (Rb-Sr) | 87Rb | 87Sr | 49.6 billion yr | 10 million–4.6 billion yr | Muscovite, biotite, K-feldspar, whole rock |
| Samarium-Neodymium (Sm-Nd) | 147Sm | 143Nd | 106 billion yr | 100 million–4.6 billion yr | Garnet, clinopyroxene, whole rock |
| Radiocarbon (14C) | 14C | 14N | 5,730 yr | 200–50,000 yr | Wood, bone, charcoal, shell, organic sediment |
The uranium-lead system is widely regarded as the most precise geochronological tool available, and for the oldest rocks and minerals it is nearly indispensable. Uranium has two naturally occurring radioactive isotopes, 238U and 235U, that decay in parallel to 206Pb and 207Pb respectively, via different multi-step decay chains. Because both chains operate simultaneously in the same mineral, the two resulting ages can be compared with each other as an internal consistency check. The mineral zircon (ZrSiO4) is particularly favored for U-Pb dating because it readily incorporates uranium into its crystal lattice during formation but excludes lead almost entirely, meaning essentially all measured lead in a zircon is radiogenic daughter product rather than initial contamination. Zircons are also physically and chemically robust, surviving weathering, metamorphism, and sedimentary transport for billions of years. The oldest known terrestrial mineral, a detrital zircon from the Jack Hills of Western Australia, has yielded a U-Pb age of 4.404 billion years, confirmed by atom-probe tomography.7, 8
The potassium-argon (K-Ar) and its more precise variant, argon-argon (40Ar/39Ar), dating exploit the decay of 40K to 40Ar. Potassium is one of the most abundant elements in the Earth's crust, present in nearly all common minerals, making this system broadly applicable. In the conventional K-Ar method, potassium and argon are measured separately on different aliquots of the sample, which introduces uncertainty from sample heterogeneity. The 40Ar/39Ar method, developed in the 1960s, improves on this by irradiating the sample in a nuclear reactor to convert a known fraction of stable 39K into 39Ar; both argon isotopes are then measured simultaneously on the same aliquot, greatly reducing analytical uncertainty. By incrementally heating the sample and measuring the 40Ar/39Ar ratio released at each temperature step, analysts obtain a "plateau age" that confirms the isotopic system remained closed since crystallization, or reveals partial resetting if the system was disturbed.16, 3
The rubidium-strontium (Rb-Sr) system was among the first widely applied in geochronology following the development of mass spectrometry in the mid-20th century.25 Rubidium-87 decays to strontium-87 with a half-life of approximately 49.6 billion years, making it ideal for dating ancient rocks. Because strontium has a non-radiogenic isotope, 86Sr, the ratio 87Sr/86Sr can be measured precisely and used as a reference. The Rb-Sr system is most powerfully applied through the isochron method, which eliminates the need to know the initial strontium isotope ratio directly.3, 17
The samarium-neodymium (Sm-Nd) system is particularly valuable for dating mafic and ultramafic rocks (those rich in magnesium and iron), which tend to have low rubidium and potassium abundances but contain samarium and neodymium in garnet and pyroxene. Because neodymium is relatively immobile in fluids, the Sm-Nd system is also more resistant to disturbance during metamorphism than some other systems, making it useful for dating ancient metamorphic terrains and mantle-derived rocks.3, 17 Its extremely long half-life of approximately 106 billion years makes it sensitive primarily to rocks older than about 100 million years.
Radiocarbon dating
Radiocarbon dating occupies a special place among radiometric methods because of its extraordinary reach into human prehistory and its applicability to organic materials directly relevant to archaeology and Quaternary geology. Carbon-14 is a radioactive isotope of carbon with a half-life of 5,730 years, produced continuously in the upper atmosphere when cosmic-ray-produced neutrons bombard nitrogen-14 atoms.13 This cosmogenic 14C is oxidized to 14CO2 and enters the global carbon cycle, so that living organisms continuously incorporate 14C at a ratio to stable 12C that reflects the atmospheric concentration. When an organism dies, it ceases to exchange carbon with the environment, and its 14C decays without replenishment. The age of the organism's remains can therefore be calculated from the remaining fraction of 14C, assuming knowledge of the original ratio.
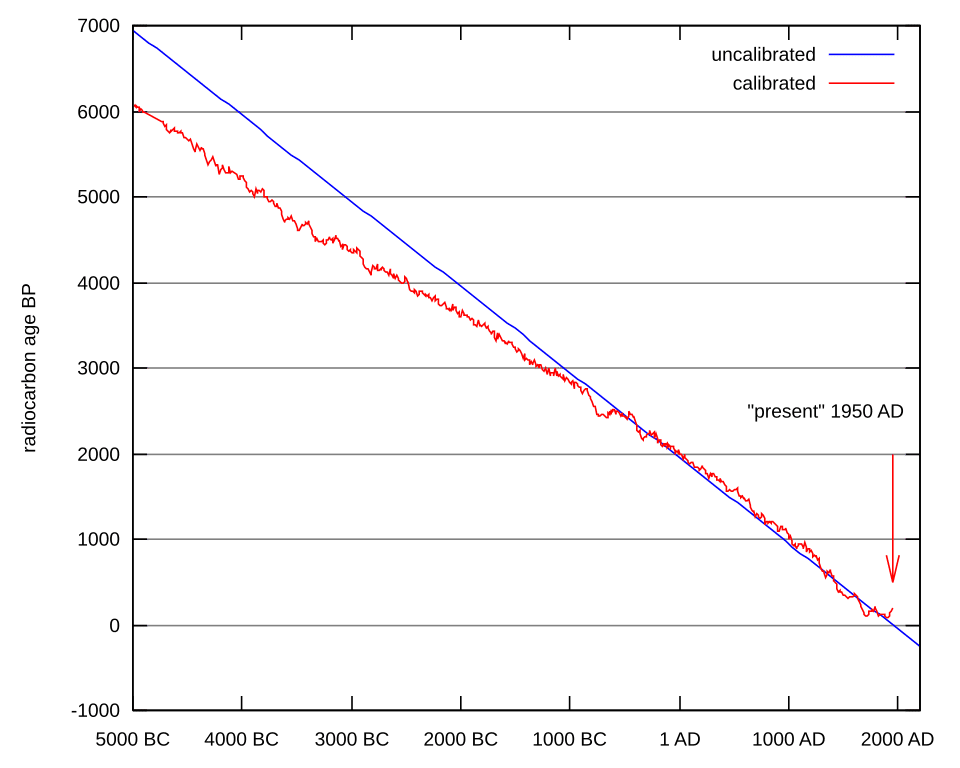
Willard Libby, who received the Nobel Prize in Chemistry in 1960 for the invention of radiocarbon dating, demonstrated in 1949–1951 that the method could reliably date organic materials across thousands of years.13 The practical upper limit of conventional radiocarbon dating is approximately 50,000 years, beyond which the residual 14C activity falls below detectable thresholds. Because the half-life of carbon-14 is short relative to the age of the Earth, radiocarbon is entirely inapplicable to geological questions about billion-year timescales; it is used exclusively for dating organic materials from the Quaternary period and is entirely distinct from the long-lived decay systems used in geochronology.
A critical refinement of the method came from the recognition that atmospheric 14C concentrations have not been perfectly constant over time. Variations in solar activity and the geomagnetic field alter cosmic ray flux, shifting the production rate of 14C. To account for this, radiocarbon ages must be calibrated against an independent chronology of known age. The IntCal calibration curve, updated most recently in 2020 as IntCal20, extends back approximately 55,000 years and is constructed from tree rings, speleothems (cave formations), corals, and annually laminated lake sediments called varves.11 Particularly important is the sediment record from Lake Suigetsu in Japan, whose annually laminated layers provide an independent, continuous record extending back beyond 52,000 years and anchoring the calibration curve for its oldest segments.12
Modern radiocarbon dating is predominantly performed using accelerator mass spectrometry (AMS), a technique that counts individual 14C atoms directly using a particle accelerator rather than measuring their radioactive decay events. AMS requires sample sizes orders of magnitude smaller than conventional decay-counting methods—typically one milligram of carbon rather than several grams—and achieves greater precision and sensitivity, pushing the practical age limit to around 50,000 years with exceptional sample preparation.23
Isochron methods and concordia diagrams
One of the most elegant and rigorous approaches in geochronology is the isochron method, which eliminates the need to assume an initial daughter isotope composition and simultaneously tests whether the sample suite behaved as a closed isotopic system. In the Rb-Sr isochron method, for example, a suite of cogenetic rocks or minerals that formed at the same time from a homogeneous source will have had the same initial 87Sr/86Sr ratio but different 87Rb/86Sr ratios due to different rubidium and strontium concentrations. As time passes, each sample accumulates radiogenic 87Sr in proportion to its Rb/Sr ratio. When the current 87Sr/86Sr is plotted against 87Rb/86Sr for all samples on a graph, the data fall on a straight line called an isochron. The slope of this line is directly proportional to the age of the suite, and the y-intercept gives the initial strontium isotope ratio at the time of formation. A tight linear array of points on an isochron diagram is powerful evidence that the system has remained closed and that the age is meaningful.3, 17 Scatter from the isochron, by contrast, signals open-system behavior or heterogeneous initial conditions, alerting geochronologists that simple age interpretation is not warranted.
For uranium-lead dating, George Wetherill devised an analogous graphical tool in 1956 known as the concordia diagram.15 Because uranium has two decay chains operating simultaneously—238U to 206Pb and 235U to 207Pb—any closed system will produce two independent age estimates that must agree with each other. On a concordia diagram, the 206Pb/238U ratio is plotted against the 207Pb/235U ratio. The theoretical curve traced by samples of any closed system of any age is called the concordia curve. A sample that has remained perfectly closed since crystallization will plot exactly on the concordia, and its position on the curve gives the age. If a rock was partially reset by a later thermal event—if lead was lost from zircons, for instance, during a metamorphic episode—the data points fall off the concordia in a systematic way, lying on a chord called a discordia. The two endpoints of this discordia chord define the age of original crystallization and the age of the later disturbance, allowing both events to be dated simultaneously. This ability to recover meaningful age information even from isotopically disturbed rocks is a distinctive strength of the U-Pb system.15, 24
Cross-checking between independent methods
The most compelling evidence for the reliability of radiometric dating is the systematic convergence of completely independent decay systems on the same ages for the same rocks. If the fundamental physical assumption underlying radiometric dating—that decay rates are constant—were incorrect, or if the methods were plagued by systematic errors, there would be no reason to expect K-Ar, Rb-Sr, Sm-Nd, and U-Pb dates from the same rock to agree with each other. They do agree, repeatedly and precisely, across the entire history of the solar system.4, 14, 18
The Acasta Gneiss of northwestern Canada, the oldest known intact crustal rock, provides a well-documented example. Multiple independent studies using U-Pb zircon geochronology, Sm-Nd whole-rock isochrons, and Rb-Sr systematics on different mineral fractions have all converged on an age of approximately 4.0 billion years for the protolith.20 The internal consistency of these independent measurements, conducted in different laboratories using different instrumentation and different mineral fractions, provides strong corroboration that the dates reflect the actual age of the rock rather than an artifact of any particular method.
A similar cross-validation is provided by meteorites. The Canyon Diablo iron meteorite, used by Clair Patterson in his landmark 1956 study, was dated by Pb-Pb isochron methods to 4.55 billion years.5 Subsequent studies of calcium-aluminum-rich inclusions (CAIs)—the oldest known solids in the solar system—have refined this to 4.5682 billion years using high-precision U-Pb dating.9, 10 These ages are in excellent agreement with Sm-Nd and Rb-Sr isochron ages from chondrites, and with the combined lead isotope systematics of Earth, Moon, and meteorite samples. The convergence of five independent isotopic systems on the same age for solar system formation is powerful evidence that the methods are recording real chronological information.6, 18
Cross-checking extends to the human timescale as well. Radiocarbon dates from samples of known historical age—objects from ancient Egypt of confirmed archaeological date, wood from trees whose rings have been independently counted—consistently agree with the dates those objects are known to have.11, 13 The calibrated radiocarbon timescale furthermore overlaps seamlessly with the dendrochronological (tree-ring) record for recent millennia, and with the varve record and speleothem uranium-series dates for the older Quaternary, providing a multi-method chain of verification extending continuously from the present back more than 50,000 years.
Reliability of the method
Some objections to radiometric dating focus on the assumption that decay rates are constant. Physicists have tested this assumption in multiple ways. Decay rates have been measured over decades in laboratory settings without detectable variation.2 Nuclear physics theory provides a mechanistic explanation for why decay constants should be insensitive to environmental conditions: radioactive decay is governed by the weak and strong nuclear forces, whose strengths are set by fundamental constants of nature that are not affected by temperature, pressure, or chemistry at geological magnitudes. Experimental searches for variation in decay rates under extreme conditions have found no meaningful effect on the timescales relevant to geochronology.4, 14
A related objection is that the initial isotopic compositions of rocks cannot be known. The isochron method directly addresses this by measuring initial compositions from the intercept of the isochron line, requiring no prior assumption about what the initial ratio was. The concordia diagram for U-Pb dating provides a further check by requiring two independent decay chains to agree. These internal consistency tests mean that even if an investigator did not know the initial isotopic ratios, the methods would detect inconsistencies and flag the age as unreliable if the assumption of a closed system were violated.3, 15
Another common concern is the possibility of daughter product contamination or loss. Geochronologists are acutely aware of these possibilities and have developed extensive analytical protocols to identify and exclude affected samples. In U-Pb zircon dating, isotopic discordance—the failure of the two U-Pb decay chains to give the same age—immediately signals lead loss or gain. In Ar-Ar dating, the step-heating experiment directly tests whether argon has been added or lost, because disturbed systems produce irregular or sloping age spectra rather than flat plateau ages. In Rb-Sr and Sm-Nd, scatter from the isochron flags open-system behavior. The self-consistency tests built into every modern geochronological approach mean that the methods are not simply taken on faith; they carry internal diagnostic tools that distinguish reliable ages from compromised ones.3, 16, 17
Applications and significance
Radiometric dating is foundational to every field that operates on geological timescales.

In geology, it provides the absolute calibration for the geologic time scale—converting the relative sequence of stratigraphic layers, established by fossil succession in the 19th century, into a precisely dated chronology.22 Mass extinction boundaries, the duration of geological periods, the timing of major tectonic events, and the rates of mountain building and erosion are all quantified using radiometric dates. Without this chronological foundation, geology would remain a purely descriptive science of before-and-after sequences without any measure of pace or rate.
In planetary science, radiometric dating established the age of the solar system with a precision that no astronomical observation alone could match. Patterson's 1956 determination of the Earth's age at approximately 4.55 billion years from lead isotope ratios in meteorites, later refined by Allegre and colleagues to 4.54 ± 0.01 billion years, remains one of the landmark measurements in the history of science.5, 6 The U-Pb system applied to CAIs in primitive meteorites has since refined the age of the solar system's oldest solids to 4.5682 ± 0.0002 billion years.9, 10 These dates set the timescale within which all models of solar system formation, planetary differentiation, and the origin of life must be framed.
In human prehistory, calibrated radiocarbon dating has transformed archaeology by providing absolute ages for organic materials across the last 50,000 years, spanning the dispersal of anatomically modern humans, the Neanderthal extinction, the origin of agriculture, and the rise of the first cities. The development of AMS radiocarbon dating in the 1980s allowed the direct dating of individual seeds, animal bones, and even individual amino acids within bones, with milligram-sized samples, enabling precision impossible with earlier decay-counting techniques.23
Together, the suite of radiometric dating methods provides a coherent, precisely calibrated, multi-independent chronological framework for the entire 4.6-billion-year history of the Earth and solar system. The consistent agreement among independent methods across this entire range—from the formation of the earliest solar system solids to the death of the last woolly mammoth—represents one of the most robust empirical achievements of modern science.4, 18
References
Subcommission on geochronology: convention on the use of decay constants in geo- and cosmochronology
Evidence from detrital zircons for the existence of continental crust and oceans on the Earth 4.4 Gyr ago
The age of the Solar System redefined by the oldest Pb–Pb age of a meteoritic inclusion
Radiometric dating, geologic time, and the age of the Earth: a reply to 'scientific' creationism
A short history of the discovery of radioactivity and the origin of the term 'half-life'
U-Pb geochronology: from alpha counting to isotope ratio mass spectrometry using multiple Faraday collectors