Overview
- The end-Cretaceous mass extinction 66 million years ago eliminated the non-avian dinosaurs and triggered the most spectacular adaptive radiation in mammalian history, with surviving lineages diversifying explosively to fill vacated ecological niches within roughly 10 million years.
- Molecular clock analyses and the fossil record offer complementary but sometimes conflicting timelines for placental diversification, with most evidence now supporting a model in which interordinal stem lineages diverged in the Late Cretaceous but the ecological and morphological radiation of crown groups occurred primarily after the K-Pg boundary.
- Continental separation, climate upheavals such as the Paleocene-Eocene Thermal Maximum, and the repeated opening and closing of land bridges shaped mammalian biogeography, producing striking patterns of convergent evolution between marsupials in Australia and placentals on other continents.
The mammalian adaptive radiation is the explosive diversification of mammals that followed the end-Cretaceous mass extinction approximately 66 million years ago. For more than 160 million years, mammals had coexisted with dinosaurs, largely confined to small body sizes and limited ecological roles. The asteroid impact that extinguished the non-avian dinosaurs removed the dominant large-bodied terrestrial vertebrates virtually overnight, and the ecological vacuum that resulted set the stage for the most dramatic diversification event in mammalian history.1, 7 Within roughly 10 million years of the extinction, mammals had radiated into an astonishing array of forms — from tiny insectivores to massive herbivores, from ocean-going whales to echolocating bats — and every major living order of placental mammals had appeared in the fossil record.3, 4
Understanding this radiation requires integrating evidence from paleontology, molecular phylogenetics, biogeography, and climate science. The timing and tempo of the diversification, the ecological dynamics that drove it, and the roles of continental drift and climate change in shaping the global distribution of mammalian lineages remain among the most actively investigated questions in evolutionary biology.2, 22
The Mesozoic mammalian bottleneck
Mammals first appeared in the Late Triassic, approximately 225 million years ago, as small-bodied descendants of the synapsid lineage.1 Throughout the Jurassic and Cretaceous periods, mammals diversified considerably in terms of dental morphology and locomotor adaptation — recent fossil discoveries have revealed gliding, swimming, burrowing, and even predatory Mesozoic mammals — but the vast majority remained small, rarely exceeding a few kilograms in body mass.1, 24 The traditional characterization of Mesozoic mammals as uniformly tiny, nocturnal, and insectivorous has been substantially revised by discoveries from Chinese Jurassic and Cretaceous deposits, which document far greater ecological diversity than previously appreciated. Nevertheless, the large-body-size niche was effectively monopolized by dinosaurs throughout the Mesozoic, constraining mammalian diversification within a relatively narrow morphospace.1, 16
Several major mammalian groups were already well established before the end-Cretaceous extinction. Multituberculates, an ancient and successful lineage of rodent-like herbivores, had diversified extensively during the Late Cretaceous and would survive the extinction to persist well into the Eocene.24 Metatherians (the group including modern marsupials) had achieved moderate diversity in the Late Cretaceous, particularly in North America, though they suffered disproportionately severe losses at the boundary.11, 13 Eutherians, the group that includes all modern placental mammals, were present but relatively inconspicuous, represented by small-bodied forms such as Maelestes gobiensis from the Late Cretaceous of Mongolia.19 Phylogenetic analysis of Late Cretaceous eutherians suggests that the common ancestor of placental mammals was a small, generalized insectivore weighing less than 500 grams.2, 19
The K-Pg boundary as ecological release
The Chicxulub asteroid impact 66 million years ago caused the extinction of all non-avian dinosaurs, most marine reptiles, ammonites, and many other groups, eliminating approximately 76 percent of all species on Earth. For mammals, the extinction was itself severe: studies of the best-sampled boundary sections in northeastern Montana suggest that mammalian species-level extinction may have reached 93 percent locally, with 86 percent of genera disappearing.8 The survivors were overwhelmingly small-bodied generalists, a pattern consistent with ecological selectivity against larger, more specialized species.8, 13
The ecological consequences of the extinction were transformative. The removal of dinosaurs as the dominant large-bodied terrestrial vertebrates freed an enormous range of ecological niches — large herbivore, apex predator, arboreal browser, aquatic forager — that had been inaccessible to mammals for more than 160 million years. This ecological release is the central mechanism invoked to explain the rapidity and breadth of the subsequent mammalian radiation.7, 12 Analysis of dental morphology and body size across the K-Pg boundary reveals that new mammalian lineages rapidly invaded dietary niches that had been unoccupied before the extinction, including dedicated herbivory and large-prey carnivory, within the first few hundred thousand years of the Paleocene.13
Remarkably detailed evidence for the tempo of early recovery comes from the Corral Bluffs site in the Denver Basin of Colorado, which preserves an exceptionally continuous fossil record spanning the first million years after the extinction. Within 100,000 years, mammalian body mass had recovered to near pre-extinction levels, with raccoon-sized species appearing in the fauna. By 300,000 years post-extinction, some mammals had reached beaver-sized proportions (~25 kg), coincident with a significant increase in plant species richness. By 700,000 years, the herbivore Ectoconus ditrigonus exceeded 45 kilograms, and the mammalian fauna had achieved a diversity and ecological breadth far exceeding anything documented in the Cretaceous.7
The Paleocene radiation and archaic orders
The Paleocene epoch (66 to 56 million years ago) witnessed the initial explosion of mammalian diversity following the extinction. The mammals that dominated Paleocene ecosystems were, however, largely members of archaic orders that bear little resemblance to modern mammalian families.

These include the condylarths, a paraphyletic assemblage of generalized ungulate-like herbivores and omnivores that occupied the large-herbivore niche vacated by dinosaurs; the creodonts, archaic carnivorous mammals that filled predatory roles before being replaced by members of the modern order Carnivora; and the multituberculates, which persisted from the Mesozoic and remained ecologically abundant as small- to medium-sized herbivores throughout much of the Paleocene.12, 10
Other distinctive Paleocene groups included the pantodonts, among the first mammals to attain body masses exceeding 100 kilograms, and the taeniodonts, bizarre digging mammals with enormous claws and ever-growing teeth. In North America, the earliest Paleocene faunas were dominated by immigrant lineages — periptychids, arctocyonids, and triisodontids — that appear abruptly in the fossil record above the iridium anomaly marking the K-Pg boundary, suggesting rapid opportunistic colonization of emptied niches.13 The phylogenetic relationships among these Paleocene groups have proved exceptionally difficult to resolve because many represent short-lived evolutionary experiments that left no modern descendants, and their morphological characters are often highly convergent.10
Halliday, Upchurch, and Goswami constructed the largest morphological phylogeny of Paleocene placental mammals to date, incorporating 177 taxa and 680 morphological characters. Their analysis confirmed that evolutionary rates were dramatically elevated in the immediate aftermath of the K-Pg extinction, with eutherians experiencing a burst of morphological and taxonomic diversification that greatly exceeded background rates.9, 10 This burst was not sustained indefinitely; by the middle Paleocene, rates of origination had begun to slow as available niche space became increasingly occupied.
Molecular clocks versus the fossil record
One of the most contentious debates in mammalian evolution concerns the timing of placental diversification relative to the K-Pg boundary. Three competing models, formalized by Archibald and Deutschman in 2001, frame the discussion.25 The Explosive Model holds that both interordinal divergences (the splits between major placental orders) and intraordinal diversification occurred after the K-Pg extinction, consistent with the fossil record's abrupt appearance of modern orders in the early Cenozoic. The Short Fuse Model proposes that both interordinal and intraordinal divergences occurred well before the extinction, deep in the Cretaceous. The Long Fuse Model posits that interordinal stem lineages diverged in the Late Cretaceous, but that the ecological and morphological radiation of crown groups occurred primarily after the K-Pg boundary.5, 25
Molecular clock analyses have consistently placed the divergences between placental superorders (Afrotheria, Xenarthra, Laurasiatheria, and Euarchontoglires) in the Late Cretaceous, approximately 85 to 100 million years ago, and the divergences between many constituent orders at 75 to 90 million years ago.3, 5 Bininda-Emonds and colleagues, using a supertree of nearly all mammalian species, found that net diversification rates barely changed across the K-Pg boundary, with the major pulse of speciation occurring instead in the Eocene.3 These results initially appeared to support the Short Fuse Model, in which the extinction played a minimal role in mammalian diversification.
However, subsequent studies using larger genomic datasets and more sophisticated dating methods have produced results more consistent with the Long Fuse Model. Dos Reis and colleagues, analysing 36 nuclear and 274 mitochondrial genomes with Bayesian relaxed-clock methods and robust fossil calibrations, estimated that Placentalia diverged approximately 88 to 90 million years ago, but that most present-day orders originated within a 20-million-year window from 65 to 45 million years ago — squarely after the K-Pg boundary.6 Meredith and colleagues similarly found that interordinal cladogenesis predated the extinction, but that intraordinal splitting and ecological diversification exploded afterward in response to newly available niche space.4
The most comprehensive morphological analysis, by O'Leary and colleagues, scored more than 4,500 phenomic characters for 86 fossil and living species and concluded that crown Placentalia itself originated after the K-Pg boundary, a result consistent with the Explosive Model and the observation that no unambiguous crown placental has been identified in Cretaceous rocks.2 The resolution of these competing timelines remains an active frontier, but the emerging consensus favours a model in which deep molecular divergences among stem lineages occurred in the Cretaceous while the morphological, ecological, and species-level radiation that produced recognizable modern orders was concentrated in the Paleocene and Eocene.4, 6, 22
Three models for the timing of placental mammal diversification5, 25
| Model | Interordinal divergence | Intraordinal radiation | Primary evidence |
|---|---|---|---|
| Explosive | After K-Pg (66–56 Ma) | After K-Pg (66–56 Ma) | Fossil record; phenomic analysis |
| Long Fuse | Late Cretaceous (100–85 Ma) | After K-Pg (66–45 Ma) | Molecular clocks + fossil calibration |
| Short Fuse | Late Cretaceous (100–85 Ma) | Late Cretaceous (85–66 Ma) | Early molecular supertrees |
Emergence of modern orders
By the late Paleocene and early Eocene, the archaic mammalian orders were being progressively replaced by members of modern placental orders that are still extant today. The transition was not instantaneous; in many cases, archaic and modern lineages coexisted for millions of years before the archaic groups declined to extinction. The following paragraphs summarize the origins of several key modern orders as documented by the fossil and molecular evidence.
Primates first appear in the fossil record at or near the Paleocene-Eocene boundary, approximately 55.5 million years ago. The earliest known primates include the omomyid Teilhardina, a tiny primate that appeared on three northern continents nearly simultaneously during the Paleocene-Eocene Thermal Maximum (PETM), suggesting remarkably rapid intercontinental dispersal facilitated by high-latitude land bridges.18 Earlier Paleocene forms sometimes referred to as stem primates (plesiadapiforms) lack the diagnostic features of crown Primates and their phylogenetic position remains debated.12
Rodentia, the most species-rich mammalian order today with more than 2,500 living species, appeared by the late Paleocene and diversified rapidly in the Eocene. Their gnawing dentition, with continuously growing incisors, proved extraordinarily successful across virtually every terrestrial habitat.22 Carnivora arose in the Paleocene as small, marten-like miacids and viverravids, gradually replacing the archaic creodonts and oxyaenids as the dominant mammalian predators by the middle Eocene.12
Perissodactyla (odd-toed ungulates, including horses, rhinoceroses, and tapirs) and Artiodactyla (even-toed ungulates, now classified within Cetartiodactyla to include whales) both appeared near the Paleocene-Eocene boundary, coincident with the PETM.17, 18 These two orders rapidly diversified to dominate the large-herbivore niche, replacing the archaic condylarths. The molecular evidence places the divergence of Cetacea (whales) from within artiodactyls, with the earliest fossil whale, Pakicetus, dating to approximately 50 million years ago.22
Chiroptera (bats) represent one of the most remarkable outcomes of the mammalian adaptive radiation. The earliest known bat with a complete skeleton, Onychonycteris finneyi from the Early Eocene of Wyoming (~52.5 million years ago), could fly but apparently lacked echolocation, suggesting that flight evolved before sonar in the chiropteran lineage.20 Molecular divergence estimates place the origin of crown Chiroptera somewhat earlier, in the late Paleocene, but the fossil record of the group's earliest evolution remains sparse.22
The Paleocene-Eocene Thermal Maximum and mammalian dispersal
The Paleocene-Eocene Thermal Maximum (PETM), a dramatic global warming event approximately 55.5 million years ago, profoundly influenced mammalian evolution and biogeography. During the PETM, global temperatures rose by 5 to 8 degrees Celsius over a geologically brief interval of approximately 20,000 years, driven by a massive release of carbon into the atmosphere.17 The warming eliminated thermal barriers at high latitudes, allowing tropical and subtropical forests to extend to polar regions and creating continuous corridors of warm-climate habitat across the northern continents.
The PETM is associated with the nearly simultaneous first appearance of three modern mammalian orders — Primates, Perissodactyla, and Artiodactyla — on all three northern continents (North America, Europe, and Asia). The earliest Eocene primate Teilhardina dispersed across Asia, Europe, and North America within 13,000 to 22,000 years of the onset of the PETM, a rate of intercontinental dispersal that implies the existence of continuous land connections (likely via Beringia and the De Geer and Thulean routes across the North Atlantic) and the absence of climatic barriers to movement.18 Simultaneously, the archaic mammalian faunas that had dominated the Paleocene — including many condylarths and plesiadapiforms — declined sharply, replaced by the modern orders that would come to dominate Cenozoic ecosystems.17
The PETM thus functioned as both an accelerant of dispersal and a catalyst of faunal turnover, concentrating a disproportionate amount of evolutionary change into a narrow temporal window. The event demonstrated the sensitivity of mammalian communities to rapid climate change and established biogeographic connections that shaped the distribution of mammalian lineages for millions of years afterward.17, 18
Body size evolution and the filling of the large-body-size vacuum
One of the most conspicuous consequences of the mammalian adaptive radiation was the rapid increase in body size. Mesozoic mammals were overwhelmingly small; the largest known Cretaceous mammal weighed an estimated 10 to 15 kilograms, roughly the size of a badger.1 Within the first million years of the Paleocene, mammalian maximum body mass had already increased substantially, with the Corral Bluffs record documenting species exceeding 45 kilograms by 700,000 years after the extinction.7 By the late Paleocene, pantodonts and other archaic herbivores had surpassed 100 kilograms, and by the middle Eocene, the largest terrestrial mammals — including the brontotheres and uintatheres — weighed several hundred kilograms.12, 15
Smith and colleagues compiled a global dataset of maximum body size at the ordinal level across the Cenozoic and found a near-exponential increase in maximum mammalian body mass after the K-Pg boundary, continuing for approximately 25 million years on each continent before plateauing at asymptotic values. The maximum size achieved on any given continent was correlated with the land area available, consistent with the hypothesis that body size evolution is constrained by resource availability and home-range scaling.15

This pattern is often discussed in the context of Cope's rule, the empirical generalization that body size tends to increase within evolutionary lineages over time. Alroy, analysing body mass estimates for 1,534 North American fossil mammal species, confirmed that new species are on average 9.1 percent larger than their congeners, a within-lineage effect that persists throughout the Cenozoic and accounts for the gradual overall increase in mean body mass.14 Slater demonstrated that the K-Pg boundary coincided with a fundamental shift in the mode of body size evolution: during the Mesozoic, mammalian body size was constrained by an Ornstein-Uhlenbeck process (a model implying attraction toward a stationary optimum), whereas after the extinction, body size evolution shifted to an unconstrained Brownian motion process, reflecting release from the ecological constraints imposed by dinosaur dominance.16
Maximum mammalian body mass through the early Cenozoic7, 15
Convergent evolution across continents
The breakup of Gondwana and the progressive isolation of the southern continents during the Late Cretaceous and Cenozoic created a series of natural experiments in mammalian evolution. As placental mammals diversified across the northern continents and Africa, marsupial mammals underwent their own independent radiation in South America and Australia, producing one of the most striking examples of convergent evolution in the history of life.21, 23
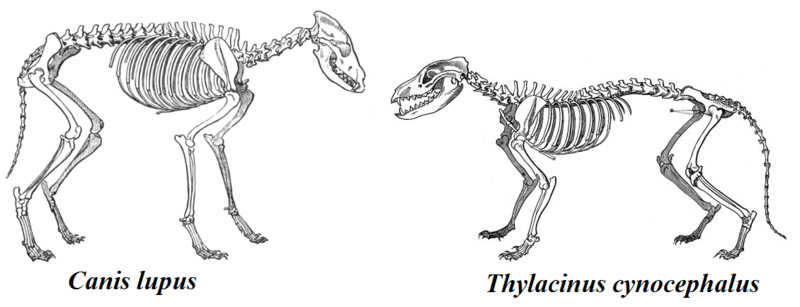
In Australia, which became isolated from Antarctica by the opening of the Tasmanian Seaway approximately 35 to 30 million years ago, marsupials radiated to fill virtually every terrestrial ecological niche. The result was a fauna that paralleled placental mammals on other continents to a remarkable degree: marsupial moles converged on the body plan of placental moles; wombats occupied the large-herbivore niche filled by rodents and lagomorphs elsewhere; the sugar glider evolved a patagium functionally identical to that of placental flying squirrels; and the thylacine, or Tasmanian wolf, converged so closely on the morphology of placental canids that its skull is nearly indistinguishable from that of a gray wolf.21 Newton and colleagues demonstrated that this cranial convergence between the thylacine and the wolf has a deep developmental basis, with the two species developing along nearly parallel growth trajectories despite 160 million years of independent evolution, accompanied by homoplasy in gene regulatory networks associated with neural crest cells.21
South America's mammalian history is equally instructive. Isolated for most of the Cenozoic following its separation from Antarctica and Africa, South America developed an endemic fauna of marsupials (including the sparassodont predators), notoungulates, litopterns, and xenarthrans (sloths, armadillos, anteaters) that evolved independently of placental mammals on other continents.23 The formation of the Isthmus of Panama approximately 3 million years ago during the Great American Biotic Interchange allowed the exchange of faunas between North and South America, with dramatic consequences: many South American endemic lineages, including the sparassodonts and most notoungulates, were replaced by invading placental carnivores and ungulates from the north, while certain South American groups, notably armadillos, opossums, and porcupines, successfully colonized North America.12
Plate tectonics and mammalian biogeography
The global distribution of mammalian lineages is inseparable from the history of plate tectonics. The configuration of continents during the Late Cretaceous and early Cenozoic determined which mammalian groups had access to which landmasses, and the subsequent opening and closing of seaways and land bridges controlled the timing and direction of dispersal events.12, 23
During the Late Cretaceous, the northern continents (Laurasia) were intermittently connected, allowing faunal exchange between North America and Asia via Beringia and between North America and Europe via North Atlantic land bridges. These connections facilitated the spread of eutherian mammals across the northern hemisphere and are reflected in the cosmopolitan distribution of many early Paleocene lineages.19 The southern continents were fragmenting from the former supercontinent Gondwana: Africa had separated from South America by approximately 100 million years ago, though some workers have proposed intermittent island-hopping connections persisting into the Eocene; Antarctica, Australia, and South America remained connected until the late Eocene and Oligocene, providing a route for marsupial dispersal from South America through Antarctica to Australia.23
The molecular phylogeny of marsupials confirms the South American origin of the Australasian marsupial radiation: the oldest divergence within crown Marsupialia separates the American opossums (Didelphimorphia) from the remaining marsupials, and the Australasian clade (Australidelphia) is nested within a broader South American-origin group. Molecular dating places the dispersal of marsupials from South America to Australia via Antarctica at approximately 55 to 50 million years ago, when Antarctica was still warm enough to support forested habitats.23
Afrotheria, the clade containing elephants, hyraxes, aardvarks, tenrecs, golden moles, and manatees, provides a dramatic example of biogeographic signal in mammalian phylogeny. Despite the morphological dissimilarity of its members, Afrotheria is strongly supported by molecular data and reflects the long isolation of Africa as an island continent during much of the early Cenozoic. The in situ radiation of these morphologically disparate lineages from a common African ancestor illustrates how geographic isolation can drive extraordinary evolutionary diversification within a single clade.4, 22
The collision of India with Asia, the closure of the Tethys Sea, the opening of the Drake Passage between South America and Antarctica, and the formation of the Isthmus of Panama each triggered profound reorganizations of mammalian biogeography. The opening of the Drake Passage approximately 34 million years ago initiated the Antarctic Circumpolar Current, which thermally isolated Antarctica, led to the formation of the Antarctic ice sheet, and permanently severed the warm-climate corridor that had allowed marsupial dispersal between South America and Australia. The resulting climatic cooling contributed to a major faunal turnover at the Eocene-Oligocene boundary, the Grande Coupure in Europe and the Mongolian Remodeling in Asia, in which archaic mammalian lineages were replaced by more modern forms.12, 22
References
Impacts of the Cretaceous Terrestrial Revolution and KPg extinction on mammal diversification
Phylogenomic datasets provide both precision and accuracy in estimating the timescale of placental mammal phylogeny
Exceptional continental record of biotic recovery after the Cretaceous–Paleogene mass extinction
Severe extinction and rapid recovery of mammals across the Cretaceous–Palaeogene boundary, and the effects of rarity on patterns of extinction and recovery
Eutherians experienced elevated evolutionary rates in the immediate aftermath of the Cretaceous–Palaeogene mass extinction
Therian mammals experience an ecomorphological radiation during the Late Cretaceous and selective extinction at the K–Pg boundary
Mammals across the K/Pg boundary in northeastern Montana, U.S.A.: dental morphology and body-size patterns reveal extinction selectivity and immigrant-fueled ecospace filling
Phylogenetic evidence for a shift in the mode of mammalian body size evolution at the Cretaceous–Palaeogene boundary
Rapid Asia–Europe–North America geographic dispersal of earliest Eocene primate Teilhardina during the Paleocene–Eocene Thermal Maximum
Cretaceous eutherians and Laurasian origin for placental mammals near the K/T boundary
Ontogenetic origins of cranial convergence between the extinct marsupial thylacine and placental gray wolf
Inferring the mammal tree: species-level sets of phylogenies for questions in ecology, evolution, and conservation
Quantitative analysis of the timing of the origin and diversification of extant placental orders