Overview
- Natural selection is the differential survival and reproduction of organisms due to heritable variation in traits that affect fitness, and it is the only known mechanism capable of producing complex adaptations in living systems.
- The mathematical foundations of natural selection were established by Fisher, Haldane, and Wright in the 1920s and 1930s, uniting Darwinian selection with Mendelian genetics, and modern quantitative methods allow the strength and form of selection to be measured directly in wild populations.
- Natural selection has been observed operating in real time in Darwin's finches, Trinidadian guppies, peppered moths, and bacterial populations, and it acts in multiple modes—directional, stabilizing, and disruptive—at multiple levels of biological organization from genes to groups.
Natural selection is the process by which organisms with heritable traits that improve their survival and reproduction in a given environment leave more offspring than organisms lacking those traits, thereby increasing the frequency of advantageous traits in the population over successive generations. First articulated jointly by Charles Darwin and Alfred Russel Wallace in 1858 and developed comprehensively in Darwin's On the Origin of Species the following year, natural selection remains the central explanatory mechanism of evolutionary biology—the only known process capable of producing the functional complexity observed in living systems.1, 2 Unlike genetic drift, which changes allele frequencies by chance, natural selection is directional: it systematically increases the frequency of variants that enhance fitness and decreases the frequency of those that reduce it.
The concept is deceptively simple. Any population in which individuals vary, in which that variation affects reproductive success, and in which variation is heritable will undergo natural selection. From these three conditions—variation, differential fitness, and heredity—adaptive evolution follows as a logical necessity, not a contingent event. Yet the consequences of this simple algorithm, operating across billions of years and millions of lineages, account for the entire diversity of life on Earth, from the metabolic pathways of thermophilic archaea to the cognitive architecture of the human brain.1, 7
Historical development
The idea that organisms change over time was not new to Darwin and Wallace. By the mid-nineteenth century, the fossil record had already revealed that past life differed strikingly from present life, and Jean-Baptiste Lamarck had proposed a mechanism of evolutionary change based on the inheritance of acquired characteristics. What Darwin and Wallace provided was a mechanism—natural selection—that could explain adaptive change without invoking purpose, foresight, or the inheritance of traits acquired during an organism's lifetime.1
Wallace articulated the basic principle in an 1855 paper on the geographical distribution of species and, more explicitly, in a manuscript sent to Darwin from the Malay Archipelago in 1858. Darwin, who had been developing the idea independently since the late 1830s, recognized the convergence immediately. The two men's papers were read together before the Linnean Society of London on 1 July 1858, constituting the first public presentation of the theory of evolution by natural selection.2 Darwin's extended treatment appeared the following year in On the Origin of Species, where he marshalled evidence from biogeography, embryology, morphology, and the practices of animal and plant breeders to argue that all species descended from common ancestors and that natural selection was the primary agent of their modification.1
Darwin recognized the analogy between natural selection and artificial selection—the deliberate breeding of domesticated plants and animals for desired traits. If human breeders could produce dramatic changes in pigeons, dogs, and crops within a few generations by selecting which individuals to breed, then the relentless pressure of the natural environment, acting on every organism in every generation over vast spans of time, could produce changes of far greater magnitude. This analogy occupies the first chapter of the Origin and was central to Darwin's rhetorical strategy for making the concept of natural selection intuitively accessible.1
Despite the persuasiveness of Darwin's argument, natural selection faced a serious conceptual obstacle: in Darwin's time, the mechanism of heredity was unknown. Without understanding how traits were transmitted from parent to offspring, it was difficult to explain how favourable variations could accumulate rather than being diluted or blended away. The resolution came with the rediscovery of Gregor Mendel's work on particulate inheritance at the turn of the twentieth century. Mendelian genetics showed that hereditary factors (genes) are discrete units that do not blend in offspring, meaning that a beneficial allele introduced into a population retains its identity across generations and can be systematically enriched by selection.6, 7
The Modern Synthesis and mathematical foundations
The integration of Darwinian natural selection with Mendelian genetics—an achievement known as the Modern Synthesis—was accomplished primarily through the mathematical work of three population geneticists in the 1920s and 1930s: Ronald Fisher, J. B. S. Haldane, and Sewall Wright. Each developed mathematical models demonstrating that natural selection acting on Mendelian variation could account for the patterns of adaptation and diversification observed in nature.3, 4, 5
Fisher's 1930 book The Genetical Theory of Natural Selection provided the first rigorous mathematical treatment of selection in large populations. Fisher demonstrated that even very small selective advantages—fitness differentials of one percent or less—could drive the fixation of beneficial alleles in populations over hundreds to thousands of generations. His fundamental theorem of natural selection stated that the rate of increase in the mean fitness of a population at any time is equal to its additive genetic variance in fitness at that time, linking the raw material of evolution (heritable variation) directly to its rate of progress.3
Haldane, in a series of ten papers titled "A Mathematical Theory of Natural and Artificial Selection" published between 1924 and 1934, worked out the expected rates of change in allele frequency under various conditions: dominant, recessive, and codominant alleles; selection of varying intensity; selection with migration; and selection in sex-linked and polyploid systems. His calculations showed that natural selection was quantitatively sufficient to explain the rates of evolutionary change inferred from the fossil record and from observed cases of adaptation such as industrial melanism.4
Wright contributed the concept of the adaptive landscape—a metaphorical topography in which peaks represent combinations of allele frequencies that confer high fitness and valleys represent low-fitness combinations. Wright emphasized the interaction between natural selection, which pushes populations uphill toward local fitness peaks, and random genetic drift, which allows small populations to cross fitness valleys and potentially reach higher peaks. His shifting balance theory proposed that the interplay of drift and selection among semi-isolated subpopulations could produce adaptive evolution faster than selection alone in a single large population.5
Theodosius Dobzhansky's 1937 book Genetics and the Origin of Species translated the mathematical framework of Fisher, Haldane, and Wright into language and evidence accessible to field biologists and systematists, catalysing the broader Modern Synthesis. Dobzhansky documented extensive genetic variation in natural populations of Drosophila pseudoobscura—variation maintained by balancing selection—thereby demonstrating that natural populations contained the raw material on which selection could act.6
Conditions for natural selection
Natural selection operates whenever three conditions are simultaneously met in a population. First, there must be variation among individuals in some trait. Second, that variation must be correlated with differences in fitness—that is, with differences in survival, mating success, fecundity, or any other component of reproductive output. Third, the variation must be at least partially heritable, meaning that offspring tend to resemble their parents more than they resemble randomly chosen members of the population. When all three conditions hold, the trait distribution in the next generation will differ from that in the current generation in the direction of higher fitness. This is natural selection.7, 8
Richard Lewontin formalized these conditions in a 1970 paper that became a foundational reference for the logic of selection. Lewontin emphasized that natural selection is an algorithmic process: it does not require any particular substrate. Any population of entities that exhibits heritable variation in fitness will evolve by natural selection, whether the entities are organisms, genes, cells, or even cultural practices. This abstraction clarified debates about the units of selection—the question of whether natural selection acts primarily on genes, individuals, kin groups, or species—by showing that selection can in principle operate at any level at which the three conditions are satisfied.8
George Price provided an even more general mathematical formalization. The Price equation, published in Nature in 1970, expresses the change in any trait across one generation as a function of the covariance between the trait and fitness, plus a term for transmission bias. In its simplest form, the equation states that the change in the mean value of a trait equals the covariance of the trait with relative fitness. The Price equation is entirely general: it makes no assumptions about the genetic system, mating pattern, population structure, or number of loci involved, and it applies to any form of selection at any level of biological organization.10
Fitness and the targets of selection
Fitness, in the context of natural selection, is a measure of an organism's expected contribution of offspring to the next generation. It is not strength, speed, or size per se, but the net reproductive output that results from the totality of an organism's traits and their interaction with the environment. A moth that evades predators but fails to find a mate has zero fitness; a slow-growing tree that produces seeds for centuries may have higher lifetime fitness than a fast-growing competitor that dies in a decade. Fitness is always relative to a specific environment and a specific population: a trait that is advantageous in one context may be neutral or deleterious in another.7, 21
Population geneticists distinguish several measures of fitness. Absolute fitness is the total number of surviving offspring produced by a genotype. Relative fitness is the ratio of a genotype's absolute fitness to the absolute fitness of the most successful genotype (or the population mean), and it is relative fitness that determines the direction and rate of allele frequency change under selection. Marginal fitness refers to the average fitness of an allele across all genetic backgrounds in which it appears, and it is this quantity that governs the fate of the allele when selection acts on multiple loci simultaneously.21
An important distinction in understanding what selection acts upon is the difference between selection of traits and selection for traits. Natural selection acts directly on phenotypes—the observable characteristics of organisms—and only indirectly on the genes that influence those phenotypes. A mutation that improves camouflage is selected because it reduces predation, not because it alters a nucleotide sequence. But because genes are the units of heredity that are faithfully transmitted across generations, the genetic basis of the selected phenotype increases in frequency. Lewontin's formulation clarified this: phenotypes are the targets of selection, but genes are the units of inheritance that carry the response to selection into future generations.8
Connecting genotype to phenotype to fitness in specific natural populations has become a central goal of modern evolutionary genetics. Barrett and Hoekstra, in a 2011 review, argued that demonstrating an allele is truly adaptive requires three lines of evidence: identification of the gene and its molecular variants, demonstration that those variants cause differences in a phenotypic trait relevant to ecological performance, and evidence that the phenotypic differences translate into fitness differences in natural populations. Many studies satisfy one or two of these criteria, but the full causal chain—from molecular variant to phenotype to fitness—has been completed in only a handful of cases.22
Modes of selection
Natural selection takes different forms depending on how fitness relates to the distribution of a trait in the population. The three principal modes—directional, stabilizing, and disruptive selection—produce distinct evolutionary outcomes and leave different signatures in the trait distribution across generations.7, 9
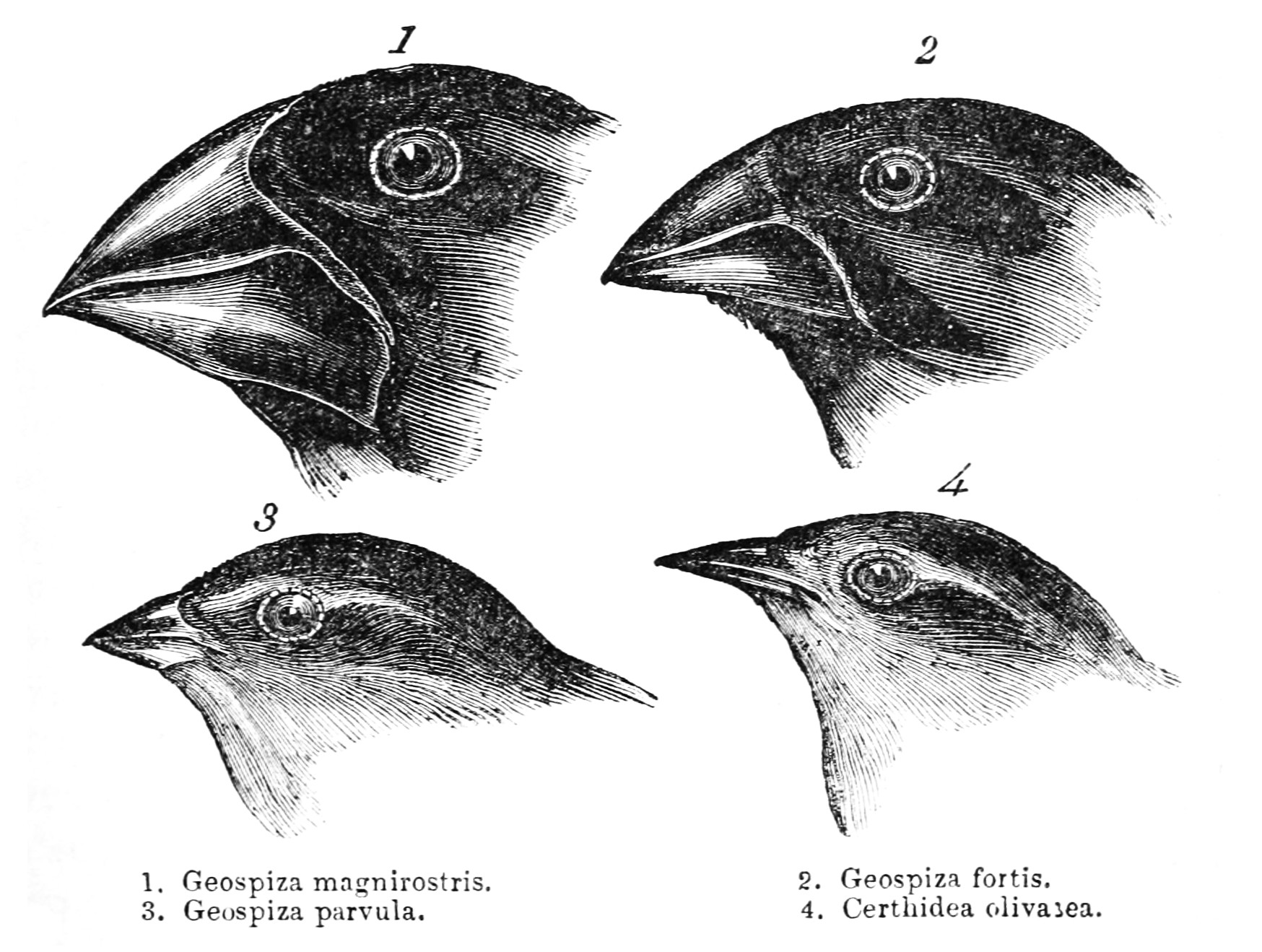
Directional selection occurs when individuals at one extreme of a trait distribution have higher fitness than those at the other extreme or at the mean. The result is a shift in the population mean toward the favoured extreme. Directional selection is the mode most people associate with natural selection, and it is the mode responsible for adaptive trends in the fossil record, such as the progressive increase in brain size in the human lineage or the elongation of horse limbs over the Cenozoic. The 1977 drought on Daphne Major in the Galápagos, which selectively killed smaller-beaked Darwin's finches and shifted the population mean toward larger, deeper beaks within a single generation, is one of the most precisely documented episodes of directional selection in a wild vertebrate population.14
Stabilizing selection favours intermediate phenotypes and acts against both extremes. When organisms near the population mean have the highest fitness, the variance of the trait is reduced over time even though the mean remains approximately constant. Stabilizing selection is thought to be the most common mode of selection operating on most traits in most populations at most times, because most organisms are already reasonably well adapted to their environments and extreme variants tend to perform poorly. The classic example is human birth weight: infants of intermediate weight have the highest survival rates, while very small and very large babies experience elevated mortality.7, 9
Disruptive selection (also called diversifying selection) favours individuals at both extremes of a trait distribution at the expense of intermediates. Disruptive selection can maintain polymorphism in a population and, if combined with assortative mating, can initiate the divergence of subpopulations that may eventually lead to speciation. The African seedcracker finch (Pyrenestes ostrinus), in which both large-billed and small-billed morphs coexist while intermediate-billed birds have reduced fitness, provides one of the best-documented cases of disruptive selection in a natural population.7
Sexual selection, which Darwin treated as a distinct process from natural selection in The Descent of Man (1871), is now generally considered a subset of natural selection that acts specifically on traits affecting mating success. Sexual selection encompasses both intrasexual selection (competition between members of the same sex for access to mates) and intersexual selection (mate choice, typically by females). Sexual selection can produce traits—such as the peacock's tail or the elk's antlers—that reduce survival but increase mating success, and it can act in opposition to natural selection on viability when the ornaments or weapons favoured by mates or rivals are costly to bear.7
Measuring selection in nature
For much of the twentieth century, natural selection was primarily a theoretical construct supported by indirect evidence. The development of quantitative methods for measuring selection directly in wild populations transformed the field from one of inference to one of direct observation. Two methodological advances were particularly important: the selection gradient approach of Lande and Arnold (1983) and the meta-analytic syntheses that followed.11
Lande and Arnold introduced a regression-based framework for estimating the strength and form of natural selection operating on quantitative traits in natural populations. By regressing individual fitness (survival, mating success, or fecundity) on standardized trait values, researchers can estimate selection gradients—the partial regression coefficients that measure the direct effect of each trait on fitness while statistically controlling for correlated traits. Linear selection gradients (β) measure the strength of directional selection, while quadratic selection gradients (γ) measure stabilizing or disruptive selection. This framework gave field biologists a standardized, widely applicable method for quantifying selection, and it sparked an explosion of empirical studies across taxa.11
Kingsolver and colleagues compiled the first large-scale synthesis of these studies in 2001, assembling more than 2,500 estimates of selection from 63 studies of 62 species. Their analysis revealed that the median absolute value of linear selection gradients was 0.16—a moderate level of directional selection—but that the distribution was strongly right-skewed, with most estimates being weak to moderate and a few being very strong. They also found that selection on morphological traits tended to be weaker than selection on life-history traits, and that stabilizing selection, while frequently expected on theoretical grounds, was less commonly detected than directional selection.12
Distribution of directional selection strength across species12
Siepielski, DiBattista, and Carlson extended this work in 2009 by assembling a database of 5,519 estimates of selection from 89 studies that included temporal replicates—repeated measurements of selection on the same trait in the same population across multiple years. Their analysis revealed that the strength and even the direction of selection frequently varied from year to year. Selection that was strongly directional in one year could be absent or reversed in the next, a pattern consistent with fluctuating environmental conditions and suggesting that the long-term outcome of selection in many populations is a dynamic balance rather than a steady march in one direction.13
Observed examples in the wild
Natural selection has been documented operating in real time in numerous natural populations. Three case studies—Darwin's finches, Trinidadian guppies, and peppered moths—illustrate the range of ecological contexts in which selection has been directly measured.
The medium ground finch (Geospiza fortis) on the Galápagos island of Daphne Major has been the subject of continuous study by Peter and Rosemary Grant since 1973. During the severe drought of 1977, the supply of small, soft seeds collapsed, leaving only large, hard seeds. The population crashed from approximately 1,200 individuals to just 180 survivors. Finches with larger, deeper beaks were better able to crack the remaining hard seeds and survived at higher rates; finches with smaller beaks died disproportionately. The survivors had beaks that were 3 to 4 percent larger than the pre-drought population average, a measurable shift in beak depth within a single generation representing a selection differential of 0.74 standard deviations.14 Over the subsequent three decades, the Grants documented repeated episodes of selection in both directions, driven by fluctuating rainfall and seed availability. Their 2002 synthesis showed that natural selection in this population was frequent, strong, and unpredictable in direction, confirming that selection is not a rare or weak force but a routine feature of population dynamics in the wild.15

David Reznick and colleagues exploited a natural experimental system in the rivers of Trinidad, where guppy populations in upstream sites above barrier waterfalls coexist with only minor predators, while populations in downstream sites below the waterfalls face intense predation from the pike cichlid (Crenicichla alta). Guppies from high-predation sites mature earlier, reproduce at a smaller size, produce more numerous but smaller offspring, and allocate a greater fraction of their body mass to reproduction—all predictions of life-history theory for organisms under heavy predation pressure. When Reznick's team transplanted guppies from high-predation sites to predator-free upstream sites in 1981, the transplanted populations evolved toward the low-predation life-history phenotype within just four to eleven years (approximately seven to eighteen generations), at rates comparable to those seen under artificial selection in the laboratory—and up to seven orders of magnitude faster than rates of change inferred from the fossil record.16
The peppered moth (Biston betularia) in industrial Britain remains one of the most extensively documented cases of natural selection by differential predation. Before the Industrial Revolution, the pale, speckled typical form of the moth was well camouflaged against lichen-covered tree bark, and a dark (melanic) form known as f. carbonaria was extremely rare. As industrial pollution killed the lichens and darkened the bark with soot, the melanic form gained a survival advantage against visually hunting birds and increased dramatically in frequency, reaching over 95 percent in some industrial regions by the mid-twentieth century. In 1924, J. B. S. Haldane used the rapid rise of the melanic form to calculate an approximate 50 percent survival advantage for melanic moths over typical moths in polluted environments—one of the first quantitative estimates of the strength of natural selection in the wild.4 H. B. D. Kettlewell's mark-recapture experiments in the 1950s demonstrated that melanic moths survived at higher rates in polluted woodlands and typical moths survived at higher rates in unpolluted woodlands, providing direct experimental evidence that differential predation was the selective agent.17, 23 Following the passage of the Clean Air Acts in the 1950s and 1960s, pollution declined, lichens recolonised the trees, and the melanic form decreased steadily in frequency—a reversal of the direction of selection documented over decades of moth surveys. Michael Majerus's long-running study near Cambridge, involving approximately 5,000 moths over seven years, was published posthumously in 2012 and decisively confirmed through direct observation of bird predation events that differential avian predation drives the frequency changes and that the recovery of the typical form closely tracked the improvement in air quality.18
Experimental evolution in the laboratory
While field studies document natural selection as it operates in uncontrolled natural environments, experimental evolution in the laboratory allows researchers to impose known selection pressures, control environmental variables, replicate populations, and trace the molecular basis of adaptive change in real time. The most famous such experiment is Richard Lenski's Long-Term Evolution Experiment (LTEE), which has followed twelve initially identical populations of Escherichia coli since 24 February 1988.19
Each population in the LTEE is propagated daily by transferring one percent of the culture into fresh glucose-limited medium, imposing selection for growth rate in a simple, constant environment. By the 10,000-generation mark, all twelve populations had evolved higher fitness relative to the ancestor, with an average fitness increase of approximately 50 percent. The pattern of adaptation followed a characteristic trajectory: rapid improvement in the first 2,000 generations, followed by a gradual deceleration as diminishing returns set in. This pattern is consistent with theoretical expectations for adaptation to a fixed environment, where the most beneficial mutations are fixed first and the remaining opportunities for improvement become progressively smaller.19

The LTEE's most celebrated result came after 31,500 generations, when one of the twelve populations evolved the ability to metabolize citrate under aerobic conditions—a trait that E. coli had never been observed to possess. Zachary Blount, Christina Borland, and Lenski showed that this innovation required the prior accumulation of at least two mutations: one that was selectively neutral when it first arose but that potentiated the citrate-using phenotype, and a second that actualized it. By replaying evolution from frozen ancestral stocks taken at different time points, the researchers demonstrated that the probability of evolving citrate utilization depended on the genetic background of the population, providing direct experimental evidence for the role of historical contingency in evolution.20 The LTEE has now exceeded 80,000 generations and continues to reveal how natural selection, mutation, and drift interact to shape the trajectory of adaptation over long periods.
Levels of selection
One of the most debated questions in evolutionary biology concerns the level of biological organization at which natural selection acts. Darwin wrote primarily of selection among individual organisms, but the question of whether selection can also operate at the level of genes, cells, groups, or species has generated a rich theoretical and empirical literature.8
The gene-centred view, most prominently associated with George Williams and Richard Dawkins, holds that the gene is the fundamental unit of selection because it is the entity that persists across generations with sufficient fidelity to be selected. Organisms, in this view, are vehicles constructed by genes to promote their own replication. While this perspective has been enormously productive in explaining phenomena such as selfish genetic elements, genomic conflict, and the evolution of sex, it is more accurately a perspective on what counts as a replicator than a claim about what selection directly acts on. Organisms remain the primary targets of selection in most ecological contexts: it is the phenotype of the whole organism that determines whether it survives and reproduces.7, 8
Group selection—the idea that natural selection can favour traits that benefit the group at the expense of the individual—was widely invoked in the mid-twentieth century to explain altruistic behaviours such as alarm calling in birds and reproductive restraint in animal populations. V. C. Wynne-Edwards proposed in 1962 that many animal behaviours evolved to regulate population size for the good of the species. This interpretation was effectively challenged by Williams in 1966 and by Hamilton's theory of kin selection, which showed that apparent altruism could be explained by selection acting on individuals through inclusive fitness—the total fitness of an individual including the effects of its behaviour on the fitness of relatives. The debate has not been entirely resolved: multilevel selection theory, developed by David Sloan Wilson and others, maintains that selection can act simultaneously at multiple levels and that the relative strength of within-group versus between-group selection determines the outcome. Whether multilevel selection is a genuinely distinct process or merely a mathematical repartitioning of individual-level selection remains a matter of ongoing discussion among evolutionary theorists.7, 8
Species selection—differential speciation and extinction rates among lineages that possess different heritable traits—has been documented in the fossil record. Lineages with larger geographic ranges, for instance, tend to have lower extinction rates, and if range size is heritable across speciation events, then a form of selection operating at the species level can produce macroevolutionary trends. Species selection is not a substitute for natural selection operating on organisms; rather, it is an additional process that can influence large-scale evolutionary patterns over geological time scales.7
Natural selection and genetic drift
Natural selection is not the only force that changes allele frequencies in populations. Genetic drift—the random fluctuation of allele frequencies due to the chance sampling of gametes in finite populations—operates alongside selection and can overpower it in small populations or for mutations with very small fitness effects. Understanding the relationship between selection and drift is essential to understanding when and how natural selection shapes evolution.5, 7
The relative importance of selection and drift depends on the product of the effective population size (Ne) and the selection coefficient (s). When Nes is much greater than one, selection dominates and the allele's fate is determined primarily by its fitness effect. When Nes is much less than one, drift dominates and the allele behaves as if it were selectively neutral, increasing or decreasing in frequency by chance regardless of its effect on fitness. For a species like Drosophila melanogaster, with an effective population size on the order of one million, a selection coefficient as small as 0.0001 (one-hundredth of one percent) is effectively visible to selection. For a species with an effective population size of one hundred, only selection coefficients greater than approximately one percent will reliably override drift.5, 7
The neutral theory of molecular evolution, proposed by Motoo Kimura in 1968, argued that the vast majority of molecular evolutionary changes—substitutions at the DNA level—are selectively neutral or nearly so, and are fixed by genetic drift rather than by positive natural selection. The neutral theory does not deny the importance of natural selection for adaptive phenotypic evolution; rather, it asserts that at the molecular level, most substitutions reflect the fixation of neutral variants by drift. The nearly neutral theory, developed by Tomoko Ohta, extended this framework to include weakly deleterious mutations whose fate is determined by the interplay of drift and purifying selection. Both theories emphasize that natural selection is not the only evolutionary force and that its relative importance depends on population size and the distribution of fitness effects of new mutations.7
Constraints and limits on selection
Natural selection is a powerful mechanism, but it does not operate without constraints. Several factors limit the ability of selection to optimize traits and produce perfect adaptation.7
Genetic constraints arise because selection can only act on existing heritable variation. If the mutations necessary for a particular adaptation have not arisen, or if the genetic architecture of a trait limits the range of phenotypes that can be produced, selection cannot move the population toward the optimal phenotype. Pleiotropy—the phenomenon in which a single gene affects multiple traits—means that a mutation that improves one trait may simultaneously worsen another, creating a trade-off that limits the response to selection on either trait individually. Similarly, genetic correlations between traits can constrain the independent evolution of those traits, channelling evolutionary change along the path of least genetic resistance rather than the path of greatest adaptive benefit.7
Historical constraints arise from the path-dependent nature of evolution. Natural selection works by modifying existing structures, not by designing new ones from scratch. The vertebrate eye, the recurrent laryngeal nerve, and the human spine all bear the imprint of their evolutionary history: they function well enough for survival and reproduction, but they contain features that no rational designer would include if building the system de novo. Darwin himself recognized this, writing in the Origin that natural selection "will never produce in a being anything injurious to itself" but can produce structures that are "not absolutely perfect."1
Environmental constraints include the unpredictability of future conditions. Selection adapts organisms to their current environment, not to future environments. A population adapted to a warm climate cannot anticipate and prepare for an impending ice age. When environments change faster than populations can respond through selection, extinction may result. The tempo of environmental change relative to the tempo of evolutionary response is a critical determinant of whether selection can produce adaptation or whether the population simply declines.7, 13
Temporal fluctuations in selection, documented across many populations by Siepielski, DiBattista, and Carlson, further complicate the picture. When the direction of selection reverses from year to year, the net evolutionary change over time may be far less than the magnitude of selection operating in any single year would suggest. This oscillating selection helps explain the long-term morphological stasis observed in many fossil lineages: selection may be strong in the short term but effectively cancelled out over longer periods by reversals in direction.13
Natural selection as the engine of adaptation
Natural selection occupies a unique position among evolutionary forces. Mutation generates new genetic variation, recombination shuffles existing variation into new combinations, gene flow redistributes variation among populations, and genetic drift changes allele frequencies by chance. But only natural selection systematically improves the fit between organisms and their environments. It is the only known process that produces functional complexity—the intricate, interdependent biological machinery that makes organisms work—because it is the only process that is both directional and cumulative: each generation of selection builds on the gains of the previous generation, ratcheting the population toward higher fitness.1, 7
The evidence for natural selection is now vast and multifaceted. It encompasses the comparative anatomy of homologous structures that Darwin used to argue for common descent; the geographical distribution of species on islands and continents that first suggested to both Darwin and Wallace that organisms are shaped by their local environments; the experimental evolution of microorganisms under controlled laboratory conditions; the direct measurement of selection gradients in wild populations across hundreds of species; and the molecular detection of selection's signature in genome sequences, where patterns of nonsynonymous to synonymous substitution rates, selective sweeps, and linkage disequilibrium reveal the ongoing action of positive, purifying, and balancing selection across the genomes of virtually all species examined.7, 9, 22
The central insight of natural selection—that design without a designer is possible, that complexity can emerge from the cumulative action of a simple, mechanical process operating on variation over time—remains among the most consequential ideas in the history of science. It explained, for the first time, how the extraordinary diversity and adaptation of living things could arise through natural causes, and it unified biology under a single theoretical framework that connects molecular genetics to ecology, paleontology to medicine, and the origin of life to the ongoing evolution of every population on Earth.1, 7
References
On the tendency of species to form varieties; and on the perpetuation of varieties and species by natural means of selection
Intense natural selection in a population of Darwin's finches (Geospizinae) in the Galápagos
Evaluation of the rate of evolution in natural populations of guppies (Poecilia reticulata)
Industrial melanism in the peppered moth, Biston betularia: an excellent teaching example of Darwinian evolution in action
Dynamics of adaptation and diversification: a 10,000-generation experiment with bacterial populations
Historical contingency and the evolution of a key innovation in an experimental population of Escherichia coli